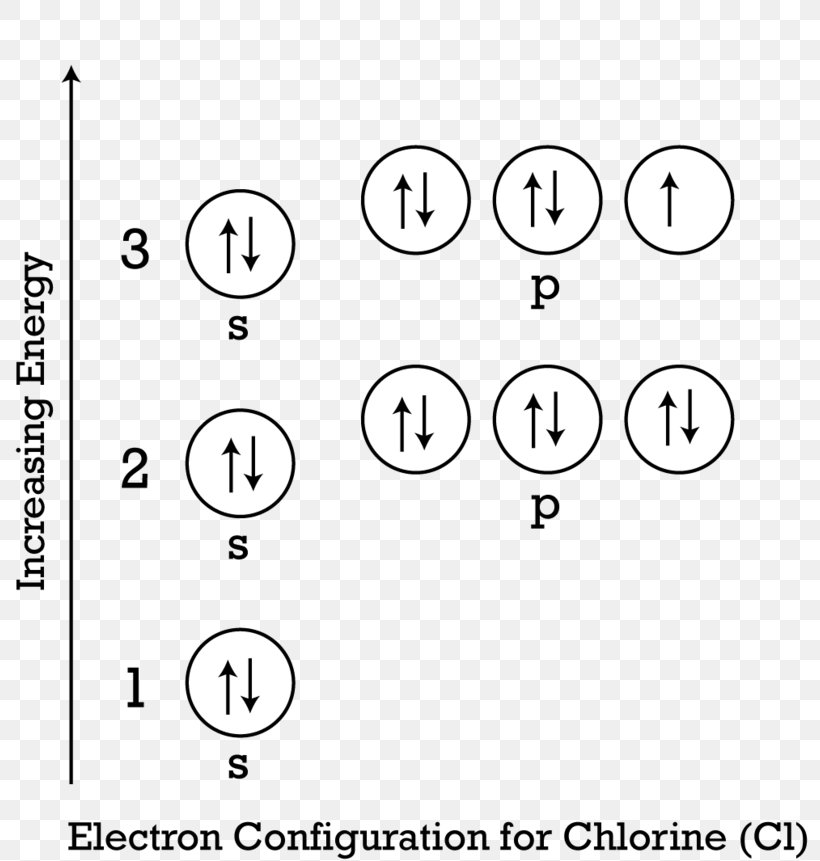
Chlorine Electron Shell Configuration . Why is the electronic configuration of chlorine important? Electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: Chlorine, a halogen, is crucial in many chemical. The shorthand electron configuration (or noble gas configuration) as well as. Chlorine has an atomic number of 17, which means it has 17 protons. Full ground state electron configuration: In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. Chlorine has 17 electrons in total. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 7 valence electrons in its outermost shell (the m shell). The electron configuration of chlorine is [ne] 3s2 3p5. Electron configuration chart of all elements is mentioned in the table below.
from utedzz.blogspot.com
Chlorine has an atomic number of 17, which means it has 17 protons. Chlorine has 17 electrons in total. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 7 valence electrons in its outermost shell (the m shell). Chlorine, a halogen, is crucial in many chemical. Electron configuration of chlorine is [ne] 3s2 3p5. Full ground state electron configuration: It is an extremely reactive element and a strong oxidising agent: The shorthand electron configuration (or noble gas configuration) as well as. The electron configuration of chlorine is [ne] 3s2 3p5.
Periodic Table Electron Configuration Orbital Diagram Periodic Table
Chlorine Electron Shell Configuration It is an extremely reactive element and a strong oxidising agent: Electron configuration of chlorine is [ne] 3s2 3p5. Electron configuration chart of all elements is mentioned in the table below. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Why is the electronic configuration of chlorine important? Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. The electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: Full ground state electron configuration: Chlorine has 17 electrons in total. The shorthand electron configuration (or noble gas configuration) as well as. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 7 valence electrons in its outermost shell (the m shell). Chlorine has an atomic number of 17, which means it has 17 protons. Chlorine, a halogen, is crucial in many chemical.
From egpat.com
Lewis dot structure How to write? Chlorine Electron Shell Configuration Full ground state electron configuration: In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 17 electrons in total. Chlorine, a halogen, is crucial in many chemical. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and. Chlorine Electron Shell Configuration.
From sciencenotes.org
Chlorine Facts Chlorine Electron Shell Configuration The shorthand electron configuration (or noble gas configuration) as well as. It is an extremely reactive element and a strong oxidising agent: Chlorine, a halogen, is crucial in many chemical. Chlorine has an atomic number of 17, which means it has 17 protons. The electron configuration of chlorine is [ne] 3s2 3p5. Chlorine has 7 valence electrons in its outermost. Chlorine Electron Shell Configuration.
From www.askiitians.com
Chlorine Study Material for IIT JEE askIITians Chlorine Electron Shell Configuration The electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. In order to write the chlorine electron configuration we. Chlorine Electron Shell Configuration.
From utedzz.blogspot.com
Periodic Table Chlorine Bohr Model Periodic Table Timeline Chlorine Electron Shell Configuration Chlorine has 17 electrons in total. Electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: Chlorine has 7 valence electrons in its outermost shell (the m shell). The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. The electron. Chlorine Electron Shell Configuration.
From www.youtube.com
Atomic Structure (Bohr Model) for Chlorine (Cl) YouTube Chlorine Electron Shell Configuration The shorthand electron configuration (or noble gas configuration) as well as. Electron configuration chart of all elements is mentioned in the table below. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. Why is the electronic configuration of. Chlorine Electron Shell Configuration.
From www.webelements.com
Elements Periodic Table » Chlorine » properties of free atoms Chlorine Electron Shell Configuration Chlorine has an atomic number of 17, which means it has 17 protons. Chlorine has 7 valence electrons in its outermost shell (the m shell). Electron configuration of chlorine is [ne] 3s2 3p5. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Electron configuration. Chlorine Electron Shell Configuration.
From www.vectorstock.com
Diagram representation of the element chlorine Vector Image Chlorine Electron Shell Configuration Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. Chlorine, a halogen, is crucial in many chemical. Electron configuration chart of all elements is mentioned in the table below. Why is the electronic configuration of chlorine important? Chlorine. Chlorine Electron Shell Configuration.
From www.youtube.com
Chlorine Electron Configuration YouTube Chlorine Electron Shell Configuration Chlorine has an atomic number of 17, which means it has 17 protons. The electron configuration of chlorine is [ne] 3s2 3p5. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine, a halogen, is crucial in many chemical. Electron configuration of chlorine is [ne] 3s2 3p5. The. Chlorine Electron Shell Configuration.
From periodictable.me
Chlorine Electron Configuration (Cl) with Orbital Diagram Chlorine Electron Shell Configuration Full ground state electron configuration: Electron configuration chart of all elements is mentioned in the table below. Chlorine has 17 electrons in total. Why is the electronic configuration of chlorine important? Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules,. Chlorine Electron Shell Configuration.
From www.shutterstock.com
Atom Chlorine This Diagram Shows Electron Stock Vector 328668782 Chlorine Electron Shell Configuration Chlorine, a halogen, is crucial in many chemical. Chlorine has an atomic number of 17, which means it has 17 protons. Full ground state electron configuration: Chlorine has 17 electrons in total. Electron configuration of chlorine is [ne] 3s2 3p5. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this. Chlorine Electron Shell Configuration.
From www.dreamstime.com
Diagram Representation Element Chlorine Stock Illustrations 1 Diagram Chlorine Electron Shell Configuration Chlorine, a halogen, is crucial in many chemical. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 7 valence electrons in its outermost shell (the m shell). The electron configuration of chlorine is [ne] 3s2 3p5. Chlorine has an atomic number of. Chlorine Electron Shell Configuration.
From commons.wikimedia.org
FileElectron shell 017 chlorine.png Wikimedia Commons Chlorine Electron Shell Configuration Electron configuration chart of all elements is mentioned in the table below. Chlorine has an atomic number of 17, which means it has 17 protons. It is an extremely reactive element and a strong oxidising agent: Full ground state electron configuration: Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill. Chlorine Electron Shell Configuration.
From www.shutterstock.com
3.722 Atom structure of chlorine Görseli, Stok Fotoğraflar ve Vektörler Chlorine Electron Shell Configuration Electron configuration of chlorine is [ne] 3s2 3p5. Why is the electronic configuration of chlorine important? Chlorine has an atomic number of 17, which means it has 17 protons. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. It is an extremely reactive element and a strong oxidising. Chlorine Electron Shell Configuration.
From animalia-life.club
Lewis Dot Structure For Chlorine Chlorine Electron Shell Configuration It is an extremely reactive element and a strong oxidising agent: Chlorine has an atomic number of 17, which means it has 17 protons. Why is the electronic configuration of chlorine important? Chlorine has 17 electrons in total. Electron configuration chart of all elements is mentioned in the table below. Chlorine has 7 valence electrons in its outermost shell (the. Chlorine Electron Shell Configuration.
From imgbin.com
Electron Configuration Atomic Orbital Chlorine Chemistry PNG, Clipart Chlorine Electron Shell Configuration Chlorine has 7 valence electrons in its outermost shell (the m shell). The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 17 electrons in total. Electron configuration of chlorine is [ne] 3s2 3p5. The shorthand electron configuration (or noble gas configuration) as well as. In order. Chlorine Electron Shell Configuration.
From www.youtube.com
Cl Electron Configuration (Chloride Ion) YouTube Chlorine Electron Shell Configuration Electron configuration of chlorine is [ne] 3s2 3p5. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 7 valence electrons in its outermost shell (the m shell). Why is the electronic configuration of chlorine important? The electron configuration of chlorine is [ne]. Chlorine Electron Shell Configuration.
From ar.inspiredpencil.com
Electron Configuration For Chlorine Chlorine Electron Shell Configuration Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. The shorthand electron configuration (or noble gas configuration) as well as. Why is the electronic configuration of chlorine important? The electron configuration of chlorine is [ne] 3s2 3p5. Electron. Chlorine Electron Shell Configuration.
From ar.inspiredpencil.com
Chlorine Atomic Structure Chlorine Electron Shell Configuration Full ground state electron configuration: In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 7 valence electrons in its outermost shell (the m shell). Electron configuration of chlorine is [ne] 3s2 3p5. The electron configuration of chlorine is [ne] 3s2 3p5. It. Chlorine Electron Shell Configuration.
From www.britannica.com
Halogen Elements, Examples, Properties, Uses, & Facts Britannica Chlorine Electron Shell Configuration Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 7 valence electrons in its outermost. Chlorine Electron Shell Configuration.
From www.animalia-life.club
Electron Configuration For Chlorine Chlorine Electron Shell Configuration In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). It is an extremely reactive element and a strong oxidising agent: Chlorine has 7 valence electrons in its outermost shell (the m shell). Why is the electronic configuration of chlorine important? Full ground state electron. Chlorine Electron Shell Configuration.
From www.embibe.com
Draw the atomic structure of the Chlorine atom and chlorine ion Chlorine Electron Shell Configuration Chlorine has 17 electrons in total. In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 7 valence electrons in its outermost shell (the m shell). The electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a. Chlorine Electron Shell Configuration.
From periodictable.me
Chlorine Electron Configuration (Cl) with Orbital Diagram Chlorine Electron Shell Configuration The electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: In order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there are 17 electrons). Chlorine has 17 electrons in total. Why is the electronic configuration of chlorine important? Group. Chlorine Electron Shell Configuration.
From utedzz.blogspot.com
Periodic Table Electron Configuration Orbital Diagram Periodic Table Chlorine Electron Shell Configuration The electron configuration of chlorine is [ne] 3s2 3p5. The shorthand electron configuration (or noble gas configuration) as well as. Chlorine has an atomic number of 17, which means it has 17 protons. Electron configuration of chlorine is [ne] 3s2 3p5. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill. Chlorine Electron Shell Configuration.
From chemtech-us.com
15 Interesting Facts About Chlorine Chlorine Electron Shell Configuration Full ground state electron configuration: Electron configuration chart of all elements is mentioned in the table below. The shorthand electron configuration (or noble gas configuration) as well as. Chlorine has 7 valence electrons in its outermost shell (the m shell). Chlorine, a halogen, is crucial in many chemical. In order to write the chlorine electron configuration we first need to. Chlorine Electron Shell Configuration.
From favpng.com
Atom Bohr Model Electron Configuration Chlorine, PNG, 1000x1000px, Atom Chlorine Electron Shell Configuration Chlorine has an atomic number of 17, which means it has 17 protons. The shorthand electron configuration (or noble gas configuration) as well as. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. It is an extremely reactive. Chlorine Electron Shell Configuration.
From topblogtenz.com
Chlorine Orbital diagram, Electron configuration, and Valence electrons Chlorine Electron Shell Configuration Chlorine has an atomic number of 17, which means it has 17 protons. Electron configuration chart of all elements is mentioned in the table below. The electron configuration of chlorine is [ne] 3s2 3p5. It is an extremely reactive element and a strong oxidising agent: Chlorine has 7 valence electrons in its outermost shell (the m shell). In order to. Chlorine Electron Shell Configuration.
From www.youtube.com
Chlorine Electron Configuration YouTube Chlorine Electron Shell Configuration Why is the electronic configuration of chlorine important? Electron configuration of chlorine is [ne] 3s2 3p5. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. The shorthand electron configuration (or noble gas configuration) as well as. The electronic. Chlorine Electron Shell Configuration.
From www.thoughtco.com
Atoms Diagrams Electron Configurations of Elements Chlorine Electron Shell Configuration The electron configuration of chlorine is [ne] 3s2 3p5. Chlorine has 7 valence electrons in its outermost shell (the m shell). The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has an atomic number of 17, which means it has 17 protons. Group 17 elements, including fluorine. Chlorine Electron Shell Configuration.
From www.alamy.com
Chlorine (Cl). Diagram of the nuclear composition and electron Chlorine Electron Shell Configuration Why is the electronic configuration of chlorine important? The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 17 electrons in total. The shorthand electron configuration (or noble gas configuration) as well as. Chlorine has an atomic number of 17, which means it has 17 protons. Electron. Chlorine Electron Shell Configuration.
From www.sciencephoto.com
Chlorine, atomic structure Stock Image C018/3698 Science Photo Library Chlorine Electron Shell Configuration Electron configuration of chlorine is [ne] 3s2 3p5. Chlorine has an atomic number of 17, which means it has 17 protons. Electron configuration chart of all elements is mentioned in the table below. Chlorine has 17 electrons in total. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell. Chlorine Electron Shell Configuration.
From circuitdataboattrains.z14.web.core.windows.net
Chlorine Atom Diagram Chlorine Electron Shell Configuration Chlorine has an atomic number of 17, which means it has 17 protons. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. Chlorine has 7 valence electrons in its outermost shell (the m shell). The electron configuration of chlorine is [ne] 3s2 3p5. Electron configuration chart of all. Chlorine Electron Shell Configuration.
From www.sciencephoto.com
Chlorine electron configuration Stock Image C029/5025 Science Chlorine Electron Shell Configuration It is an extremely reactive element and a strong oxidising agent: Electron configuration chart of all elements is mentioned in the table below. Chlorine has an atomic number of 17, which means it has 17 protons. The shorthand electron configuration (or noble gas configuration) as well as. In order to write the chlorine electron configuration we first need to know. Chlorine Electron Shell Configuration.
From www.alamy.com
Chlorine (Cl). Diagram of the nuclear composition and electron Chlorine Electron Shell Configuration Electron configuration of chlorine is [ne] 3s2 3p5. Group 17 elements, including fluorine and chlorine, have seven electrons in their outmost shells, so they tend to fill this shell with an electron from other atoms or molecules, making them. Chlorine has an atomic number of 17, which means it has 17 protons. Chlorine has 7 valence electrons in its outermost. Chlorine Electron Shell Configuration.
From www.newtondesk.com
Chlorine Cl (Element 17) of Periodic Table Newton Desk Chlorine Electron Shell Configuration Why is the electronic configuration of chlorine important? Electron configuration chart of all elements is mentioned in the table below. Full ground state electron configuration: The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. It is an extremely reactive element and a strong oxidising agent: Electron configuration of. Chlorine Electron Shell Configuration.
From womackthille.blogspot.com
Expanded Electron Configuration of Chlorine Womack Thille Chlorine Electron Shell Configuration Electron configuration of chlorine is [ne] 3s2 3p5. Electron configuration chart of all elements is mentioned in the table below. Chlorine has 17 electrons in total. The electronic configuration of chlorine determines its chemical properties and behavior, such as its reactivity and ability to form chemical bonds. The electron configuration of chlorine is [ne] 3s2 3p5. The shorthand electron configuration. Chlorine Electron Shell Configuration.