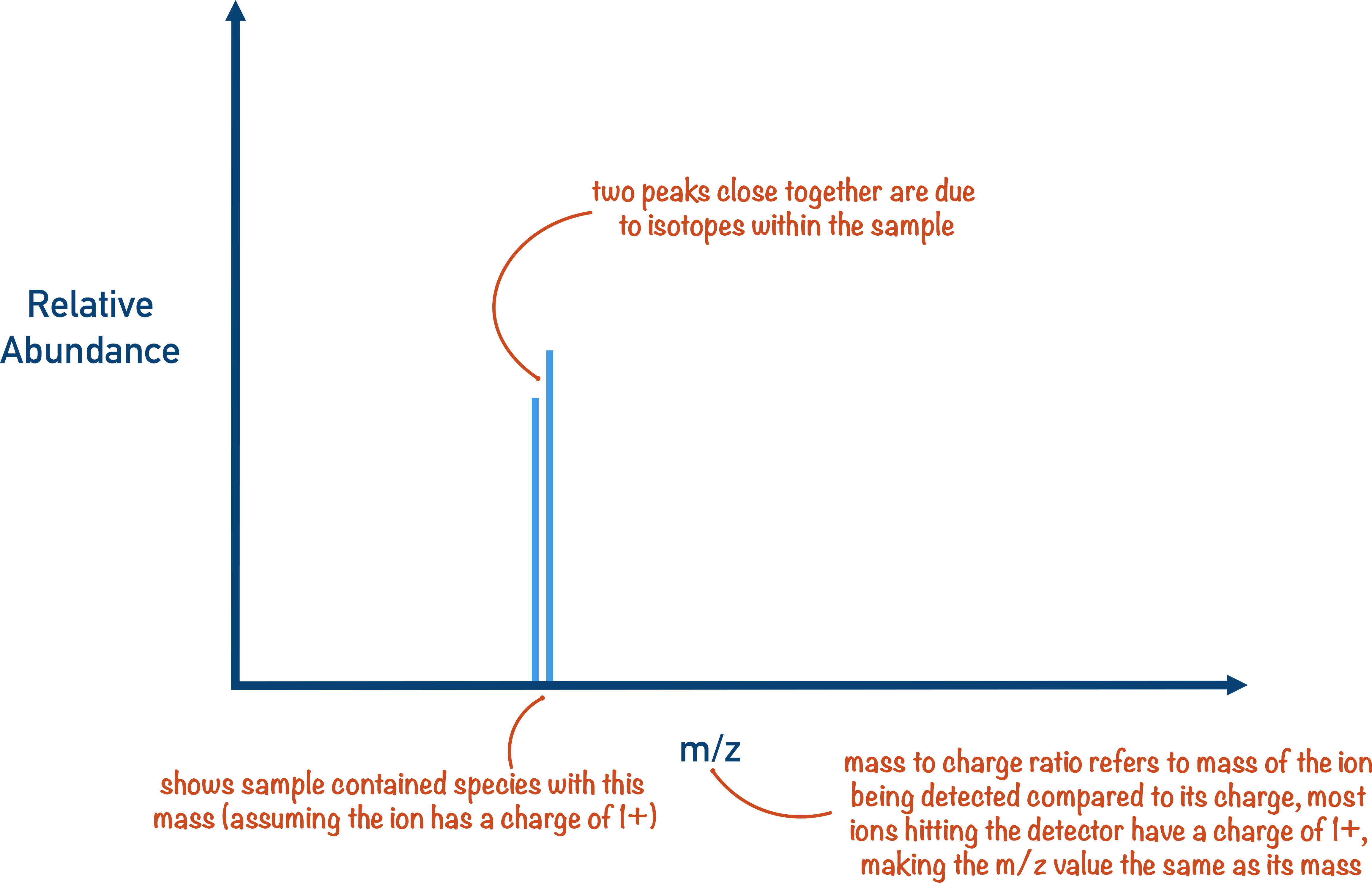
Importance Of Spectrometry In Chemistry . Here are some key reasons. Spectroscopy helps bridge that knowledge gap. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. It is a method of understanding molecules by measuring the interaction of light and matter. In absorption and scattering spectroscopy this energy is supplied by photons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the.
from www.chemistrystudent.com
Spectroscopy helps bridge that knowledge gap. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. In absorption and scattering spectroscopy this energy is supplied by photons. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. Here are some key reasons. It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy.
Mass Spectrometry (ALevel) ChemistryStudent
Importance Of Spectrometry In Chemistry Here are some key reasons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In absorption and scattering spectroscopy this energy is supplied by photons. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Here are some key reasons. All forms of spectroscopy require a source of energy. Spectroscopy helps bridge that knowledge gap. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. It is a method of understanding molecules by measuring the interaction of light and matter. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas.
From chem.libretexts.org
10 Introduction to Spectroscopy Chemistry LibreTexts Importance Of Spectrometry In Chemistry All forms of spectroscopy require a source of energy. Spectroscopy helps bridge that knowledge gap. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. It is a method of understanding molecules by measuring the interaction of light and matter. Absorption. Importance Of Spectrometry In Chemistry.
From www.youtube.com
Basic Introduction of Spectroscopy Spectroscopy organic chemistry Importance Of Spectrometry In Chemistry Here are some key reasons. It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Identification of a species involves recording the absorption. Importance Of Spectrometry In Chemistry.
From www.chem.ucla.edu
Illustrated Glossary of Organic Chemistry Spectroscopy Importance Of Spectrometry In Chemistry Spectroscopy helps bridge that knowledge gap. Here are some key reasons. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. It is a method of understanding molecules by measuring the interaction of light and matter. Mass spectrometry. Importance Of Spectrometry In Chemistry.
From www.bronkhorst.com
Mass Spectrometry and Mass Flow Control; A closer ion them Bronkhorst Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Mass spectrometry (ms) is. Importance Of Spectrometry In Chemistry.
From chem.libretexts.org
4.5 Ultraviolet and visible spectroscopy Chemistry LibreTexts Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. All forms of spectroscopy require a source of energy. Spectroscopy helps bridge that knowledge gap. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational,. Importance Of Spectrometry In Chemistry.
From www.vrogue.co
What Is Ir Spectroscopy In Chemistry Definition Types vrogue.co Importance Of Spectrometry In Chemistry All forms of spectroscopy require a source of energy. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. Spectroscopy helps. Importance Of Spectrometry In Chemistry.
From www.aakash.ac.in
NMR Spectroscopy in Chemistry Definition, Types and Importance of NMR Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. All forms of spectroscopy require a source of energy. Identification of a species involves. Importance Of Spectrometry In Chemistry.
From www.vrogue.co
Spectroscopy As Chemistry Teaching Resources vrogue.co Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of. Importance Of Spectrometry In Chemistry.
From www.slideserve.com
PPT Introduction to Mass Spectrometry (MS) PowerPoint Presentation Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. It is a method of understanding molecules by measuring the interaction of light and. Importance Of Spectrometry In Chemistry.
From www.chemistrystudent.com
IR (Infrared Spectroscopy) (ALevel) ChemistryStudent Importance Of Spectrometry In Chemistry Here are some key reasons. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. All forms of spectroscopy require a source of energy. It is a method of understanding molecules by measuring the interaction of light and. Importance Of Spectrometry In Chemistry.
From www.sliderbase.com
Mass Spectrometry Presentation Chemistry Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. It is a method of understanding molecules by measuring the interaction of light and matter. In absorption and scattering spectroscopy this energy is supplied by photons. Here are some key reasons. Spectroscopy helps bridge that knowledge gap. Mass spectrometry (ms) is commonly. Importance Of Spectrometry In Chemistry.
From chem.libretexts.org
10.1 Overview of Spectroscopy Chemistry LibreTexts Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy. Spectroscopy helps bridge that knowledge gap. In absorption and scattering spectroscopy this energy is supplied by photons. Mass. Importance Of Spectrometry In Chemistry.
From www.researchgate.net
14 Schematic diagram of a mass spectrometer. Download Scientific Diagram Importance Of Spectrometry In Chemistry Here are some key reasons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. Spectroscopy helps bridge that knowledge gap. It is a. Importance Of Spectrometry In Chemistry.
From wisc.pb.unizin.org
Isotopes, Atomic Mass, and Mass Spectrometry (M2Q3) UWMadison Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. It is a method of understanding molecules by measuring the interaction of light and matter. Identification of a species. Importance Of Spectrometry In Chemistry.
From www.aakash.ac.in
ir spectroscopy in chemistry Definition, Types and Importance of ir Importance Of Spectrometry In Chemistry In absorption and scattering spectroscopy this energy is supplied by photons. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Here are some. Importance Of Spectrometry In Chemistry.
From www.aakash.ac.in
NMR Spectroscopy in Chemistry Definition, Types and Importance of NMR Importance Of Spectrometry In Chemistry Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. Here are some key reasons. Absorption spectroscopy’s precision and versatility make it a critical. Importance Of Spectrometry In Chemistry.
From www.slideshare.net
Atomic Spectroscopy Basic Principles and Instruments Importance Of Spectrometry In Chemistry Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. In absorption and scattering spectroscopy this energy is supplied by photons. Spectroscopy helps bridge that knowledge gap. Here are some key reasons. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Identification of a species involves recording. Importance Of Spectrometry In Chemistry.
From www.youtube.com
Introduction to Atomic Spectroscopy YouTube Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Mass spectrometry (ms) is commonly regarded as an instrumental technique. Importance Of Spectrometry In Chemistry.
From chemistrymadesimple.net
How Does A Mass Spectrometer Work? Chemistry Made Simple Importance Of Spectrometry In Chemistry Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Spectroscopy helps bridge that knowledge gap. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. In spectroscopy, we use light to determine a. Importance Of Spectrometry In Chemistry.
From microbiologynotes.org
uv vis spectroscopy Microbiology Notes Importance Of Spectrometry In Chemistry Here are some key reasons. It is a method of understanding molecules by measuring the interaction of light and matter. In absorption and scattering spectroscopy this energy is supplied by photons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In spectroscopy, we use light to determine a tremendous range of molecular. Importance Of Spectrometry In Chemistry.
From chemistnotes.com
NMR spectroscopy principle, Instrumentation, Application, Chemical Importance Of Spectrometry In Chemistry Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In absorption and scattering spectroscopy this energy is supplied by photons.. Importance Of Spectrometry In Chemistry.
From www.youtube.com
Mass spectrometry Atomic structure and properties AP Chemistry Importance Of Spectrometry In Chemistry In absorption and scattering spectroscopy this energy is supplied by photons. All forms of spectroscopy require a source of energy. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Here are some key reasons. It is a. Importance Of Spectrometry In Chemistry.
From atascientific.com.au
Understanding Spectrometry and Spectroscopy ATA Scientific Importance Of Spectrometry In Chemistry Spectroscopy helps bridge that knowledge gap. All forms of spectroscopy require a source of energy. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. It is a method of understanding molecules by measuring the interaction of light. Importance Of Spectrometry In Chemistry.
From microbiologynotes.org
Spectroscopy Introduction, Principles, Types and Applications Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Spectroscopy helps bridge that knowledge gap. In absorption and scattering spectroscopy this energy is supplied by photons. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. All forms of spectroscopy require a source of. Importance Of Spectrometry In Chemistry.
From www.youtube.com
Mass Spectrometry in Organic Chemistry // HSC Chemistry YouTube Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. In spectroscopy, we use light to determine a tremendous range of molecular properties,. Importance Of Spectrometry In Chemistry.
From www.chemistrystudent.com
Mass Spectrometry (ALevel) ChemistryStudent Importance Of Spectrometry In Chemistry Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. Spectroscopy helps bridge that knowledge gap. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. In absorption and scattering spectroscopy this energy is supplied by photons. Identification of a species involves recording the absorption. Importance Of Spectrometry In Chemistry.
From www.tes.com
Spectroscopy (AS Chemistry) Teaching Resources Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. Here are some key reasons. In absorption and scattering spectroscopy. Importance Of Spectrometry In Chemistry.
From www.vrogue.co
Molecular Spectroscopy Chemistry vrogue.co Importance Of Spectrometry In Chemistry In absorption and scattering spectroscopy this energy is supplied by photons. It is a method of understanding molecules by measuring the interaction of light and matter. Here are some key reasons. Spectroscopy helps bridge that knowledge gap. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. In spectroscopy, we use light to. Importance Of Spectrometry In Chemistry.
From www.aakash.ac.in
ir spectroscopy in chemistry Definition, Types and Importance of ir Importance Of Spectrometry In Chemistry It is a method of understanding molecules by measuring the interaction of light and matter. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. All forms of spectroscopy require a source of energy. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Mass spectrometry (ms) is. Importance Of Spectrometry In Chemistry.
From www.rachidscience.com
INTRODUCTION IN CHEMISTRY MASS SPECTROMETRY Science Importance Of Spectrometry In Chemistry In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. In absorption and scattering spectroscopy this energy is supplied by. Importance Of Spectrometry In Chemistry.
From www.chemistrysteps.com
NMR spectroscopy An Easy Introduction Chemistry Steps Importance Of Spectrometry In Chemistry Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. It is a method of understanding molecules by measuring the interaction of light and matter. All forms of spectroscopy require a source of energy. Spectroscopy helps bridge that knowledge gap. Absorption spectroscopy’s precision and versatility make it a critical tool in many research. Importance Of Spectrometry In Chemistry.
From www.chemistrystudent.com
Mass Spectrometry (ALevel) ChemistryStudent Importance Of Spectrometry In Chemistry All forms of spectroscopy require a source of energy. In spectroscopy, we use light to determine a tremendous range of molecular properties, including electronic, vibrational, rotational, and. It is a method of understanding molecules by measuring the interaction of light and matter. Here are some key reasons. In absorption and scattering spectroscopy this energy is supplied by photons. Identification of. Importance Of Spectrometry In Chemistry.
From www.chem.ucla.edu
Illustrated Glossary of Organic Chemistry Ultraviolet spectroscopy Importance Of Spectrometry In Chemistry Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. In absorption and scattering spectroscopy this energy is supplied by photons. Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. Spectroscopy helps bridge. Importance Of Spectrometry In Chemistry.
From organicchemistoncall.com
Most Commonly Used IR Spectroscopy Values In Organic Chemistry The Importance Of Spectrometry In Chemistry Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the. In absorption and scattering spectroscopy this energy is supplied by photons. Absorption spectroscopy’s precision and versatility make it a critical tool in many research areas. Here are some. Importance Of Spectrometry In Chemistry.
From www.slideserve.com
PPT Chromatography and Spectroscopy PowerPoint Presentation, free Importance Of Spectrometry In Chemistry All forms of spectroscopy require a source of energy. Mass spectrometry (ms) is commonly regarded as an instrumental technique for separation of electrically charged species in. Here are some key reasons. It is a method of understanding molecules by measuring the interaction of light and matter. Spectroscopy helps bridge that knowledge gap. Identification of a species involves recording the absorption. Importance Of Spectrometry In Chemistry.