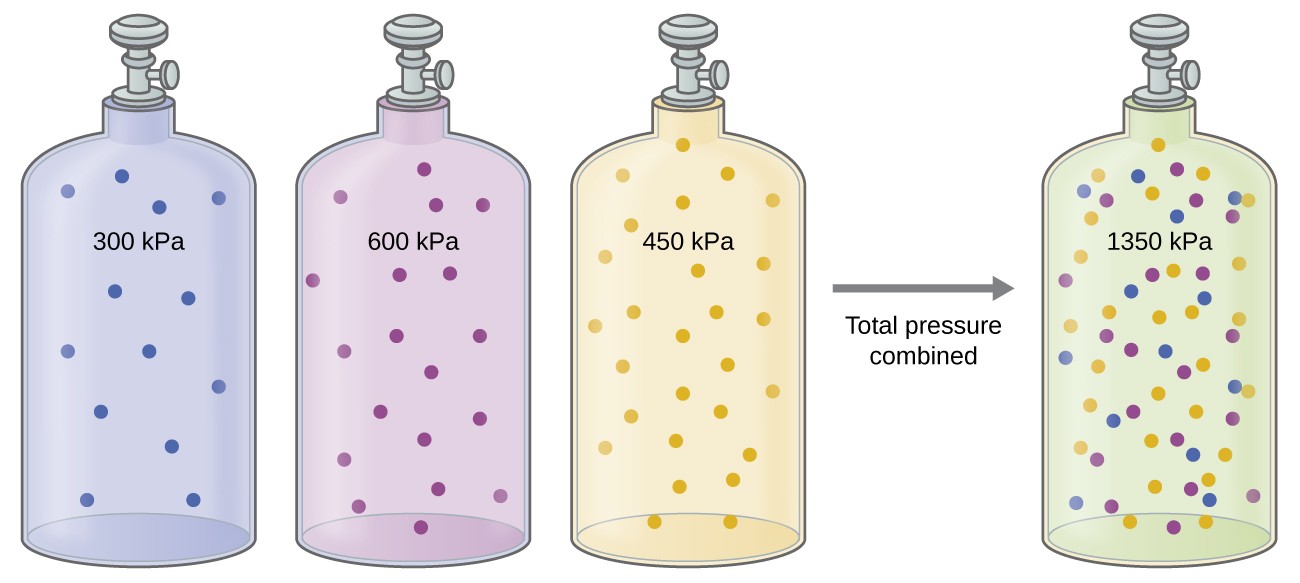
Combined Gas Law Of Partial Pressure . Use the ideal gas law to calculate the partial pressure of each gas. The explanation will assume you understand. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. It involves using dalton's law of partial pressures first, then use of the combined gas law. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components.
from pressbooks.online.ucf.edu
This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. Then add together the partial pressures to obtain the total pressure of. The explanation will assume you understand. Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. It involves using dalton's law of partial pressures first, then use of the combined gas law.
9.4 Mixtures of Gases and Partial Pressures Chemistry Fundamentals
Combined Gas Law Of Partial Pressure Use the ideal gas law to calculate the partial pressure of each gas. The explanation will assume you understand. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Use the ideal gas law to calculate the partial pressure of each gas. It involves using dalton's law of partial pressures first, then use of the combined gas law.
From worksheets.decoomo.com
30++ Dalton's Law Of Partial Pressure Worksheet Worksheets Decoomo Combined Gas Law Of Partial Pressure This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Use the ideal gas law to calculate the partial pressure of each gas. The explanation will assume you understand. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Behavior of Gases PowerPoint Presentation, free download ID3099616 Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. The explanation will assume you understand. It involves using dalton's law of partial pressures first, then use of the combined gas law. Then add together the partial. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Use the ideal gas law to calculate the partial pressure of each gas. It involves using dalton's law of partial pressures first, then use of the combined gas law. The explanation will assume you understand. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the. Combined Gas Law Of Partial Pressure.
From www.worksheeto.com
11 Chemistry Gas Laws Worksheet / Combined Gas Law Of Partial Pressure This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Use the ideal gas law to calculate the partial pressure of each gas. It involves using dalton's law of partial pressures first,. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Basic Gas Laws and Partial Pressures Laws) PowerPoint Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Gas Laws and Nature of Gases PowerPoint Presentation, free Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of. Combined Gas Law Of Partial Pressure.
From slidetodoc.com
The Combined Gas Law The Combined Gas Law Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. It. Combined Gas Law Of Partial Pressure.
From studylib.net
I. Dalton’s Law Combined Gas Law Of Partial Pressure This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressure is an ideal gas. Combined Gas Law Of Partial Pressure.
From pressbooks.online.ucf.edu
9.4 Mixtures of Gases and Partial Pressures Chemistry Fundamentals Combined Gas Law Of Partial Pressure Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the. Combined Gas Law Of Partial Pressure.
From www.britannica.com
perfect gas law chemistry and physics Britannica Combined Gas Law Of Partial Pressure The explanation will assume you understand. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Use the ideal gas law to calculate the partial pressure of each gas. Then add together the partial pressures to obtain the total pressure of. It involves using dalton's law of partial pressures first, then use of the combined gas law. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the. Combined Gas Law Of Partial Pressure.
From peoi.org
Chapter 6 Section E The Ideal Gas Law and Some Applications Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Then add together the partial pressures to obtain the total pressure of. The explanation will assume you understand. It involves using dalton's law of partial pressures first,. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Gas Law and Gas Behavior PowerPoint Presentation ID3183351 Combined Gas Law Of Partial Pressure Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. The explanation will assume you understand. Use the ideal gas law to calculate the partial pressure of each gas. Then add together the partial pressures. Combined Gas Law Of Partial Pressure.
From inspiritvr.com
Ideal Gas Law Study Guide Inspirit Learning Inc Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. It involves using dalton's law of partial pressures first, then use of the combined gas law. Dalton’s law of partial pressures is a gas law which states. Combined Gas Law Of Partial Pressure.
From slideplayer.com
Basic Gas Laws and Partial Pressures Laws) ppt download Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. The explanation will assume you understand. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum. Combined Gas Law Of Partial Pressure.
From millingschem.com
Chemistry Resources 3 MillingsChem Combined Gas Law Of Partial Pressure The explanation will assume you understand. Use the ideal gas law to calculate the partial pressure of each gas. Then add together the partial pressures to obtain the total pressure of. It involves using dalton's law of partial pressures first, then use of the combined gas law. This law states that in a mixture of two or more gases, the. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Introduction to the Gas Laws PowerPoint Presentation, free Combined Gas Law Of Partial Pressure The explanation will assume you understand. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Law PowerPoint Presentation, free download ID5864985 Combined Gas Law Of Partial Pressure Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of. Combined Gas Law Of Partial Pressure.
From www.sliderbase.com
Gas Laws Presentation Chemistry Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Dalton’s law of partial pressures is a gas law which states. Combined Gas Law Of Partial Pressure.
From www.chegg.com
Solved ulvise 11 Gas Laws Name Lab day , time __1 Gas Laws Combined Gas Law Of Partial Pressure This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Dalton’s. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT The Combined Gas Law PowerPoint Presentation, free download ID Combined Gas Law Of Partial Pressure The explanation will assume you understand. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure The explanation will assume you understand. Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. This law states that in a mixture of two. Combined Gas Law Of Partial Pressure.
From owlcation.com
The Theories and Behavior of Gas Owlcation Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Law Avogadro’s Principle PowerPoint Presentation Combined Gas Law Of Partial Pressure Use the ideal gas law to calculate the partial pressure of each gas. Then add together the partial pressures to obtain the total pressure of. It involves using dalton's law of partial pressures first, then use of the combined gas law. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure The explanation will assume you understand. Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. This law states that in a. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. Use the ideal gas law to calculate the partial pressure of each gas. Then add together the partial pressures to obtain the total pressure of. The explanation will assume you understand. This law states that in a mixture of two or more gases, the. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. This law states that in a mixture of two or more gases, the total. Combined Gas Law Of Partial Pressure.
From www.youtube.com
Gas Laws Boyle's Law, Charles's Law, GayLussac’s Law and Avogadro's Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. Then add together the partial pressures to obtain the total pressure of. The explanation will assume you understand. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum. Combined Gas Law Of Partial Pressure.
From sciencenotes.org
Combined Gas Law Definition, Formula, Examples Combined Gas Law Of Partial Pressure Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. Dalton’s law of partial pressures is a gas law which states that the total pressure. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual gas. The explanation will assume you understand. Then add together the partial pressures to obtain the total pressure of. This law states that in a mixture. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Combined Gas Laws and Dalton’s Law of Partial Pressures Combined Gas Law Of Partial Pressure Then add together the partial pressures to obtain the total pressure of. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the sum of the partial pressures of each gas. This law states that in a mixture of two or more gases, the total pressure. Combined Gas Law Of Partial Pressure.
From www.slideserve.com
PPT Basic Gas Laws and Partial Pressures Laws) PowerPoint Combined Gas Law Of Partial Pressure This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Dalton’s law of partial pressures is a gas law which states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures exerted by each individual. Combined Gas Law Of Partial Pressure.
From www.youtube.com
Combined Gas Law Pressure, Volume and Temperature Straight Science Combined Gas Law Of Partial Pressure It involves using dalton's law of partial pressures first, then use of the combined gas law. The explanation will assume you understand. Use the ideal gas law to calculate the partial pressure of each gas. Dalton’s law of partial pressure is an ideal gas law that states that the total pressure of a mixture of gases is equal to the. Combined Gas Law Of Partial Pressure.
From sciencenotes.org
Dalton's Law of Partial Pressure Definition and Examples Combined Gas Law Of Partial Pressure Then add together the partial pressures to obtain the total pressure of. This law states that in a mixture of two or more gases, the total pressure is the sum of the partial pressures of all the components. Use the ideal gas law to calculate the partial pressure of each gas. The explanation will assume you understand. Dalton’s law of. Combined Gas Law Of Partial Pressure.