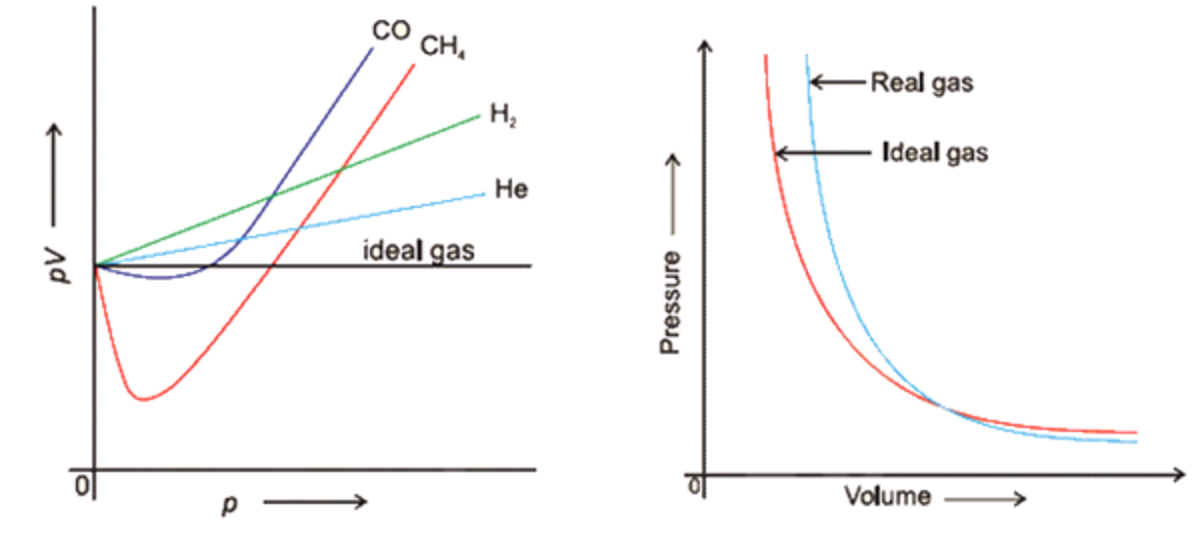
Delineate Gas Law Of Real Gas . The equation of real gas law is : Here, p is the average pressure, v is the fixed volume of the container enclosing n. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The most common relations calculated. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. The equation of state for real gases. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. At high temperatures and low. We also examine liquefaction, a key property of. To recognize the differences between the behavior of an ideal gas and a real gas. To understand how molecular volumes and intermolecular attractions.
from classnotes.org.in
While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. We also examine liquefaction, a key property of. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. The most common relations calculated. At high temperatures and low. To understand how molecular volumes and intermolecular attractions. The equation of state for real gases. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. To recognize the differences between the behavior of an ideal gas and a real gas. Here, p is the average pressure, v is the fixed volume of the container enclosing n.
Real Gases Chemistry, Class 11, States of Matter
Delineate Gas Law Of Real Gas In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. To understand how molecular volumes and intermolecular attractions. To recognize the differences between the behavior of an ideal gas and a real gas. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The equation of state for real gases. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. The equation of real gas law is : Here, p is the average pressure, v is the fixed volume of the container enclosing n. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. The most common relations calculated. At high temperatures and low. We also examine liquefaction, a key property of.
From www.youtube.com
The Ideal Gas Law YouTube Delineate Gas Law Of Real Gas The equation of real gas law is : We also examine liquefaction, a key property of. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. At high temperatures and low. The equation of state for real gases. The most common relations calculated. To understand how. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT 6 Gases PowerPoint Presentation ID352441 Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. In a gas, the potential energy of interactions between molecules is. Delineate Gas Law Of Real Gas.
From energiatoday.com
Ideal Gas vs Real Gas Differences and Concept Delineate Gas Law Of Real Gas To recognize the differences between the behavior of an ideal gas and a real gas. Here, p is the average pressure, v is the fixed volume of the container enclosing n. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. The most common. Delineate Gas Law Of Real Gas.
From studylib.net
12.2 Ideal Gasses and the Ideal Gas Law Delineate Gas Law Of Real Gas To recognize the differences between the behavior of an ideal gas and a real gas. We also examine liquefaction, a key property of. The most common relations calculated. The equation of state for real gases. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. While. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT Gases PowerPoint Presentation, free download ID4387987 Delineate Gas Law Of Real Gas To recognize the differences between the behavior of an ideal gas and a real gas. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. To understand. Delineate Gas Law Of Real Gas.
From www.chegg.com
Solved Technical Report (5 marks) Delineate Gas laws of Delineate Gas Law Of Real Gas Here, p is the average pressure, v is the fixed volume of the container enclosing n. At high temperatures and low. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. To understand how molecular volumes. Delineate Gas Law Of Real Gas.
From owlcation.com
The Theories and Behavior of Gas Owlcation Delineate Gas Law Of Real Gas At high temperatures and low. The equation of real gas law is : While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. In this section, we consider the properties of real gases and how and why they differ from the predictions of the. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Real Gas vs Ideal Gas Delineate Gas Law Of Real Gas We also examine liquefaction, a key property of. The most common relations calculated. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Combined Gas Law Definition, Formula, Examples Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. Here, p is the average pressure, v is the fixed volume of the container enclosing n. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. To recognize the. Delineate Gas Law Of Real Gas.
From www.sliderbase.com
Gas Laws Presentation Chemistry Delineate Gas Law Of Real Gas While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. To understand how molecular volumes and intermolecular attractions. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. At high. Delineate Gas Law Of Real Gas.
From www.sliderbase.com
The Ideal Gas Law Presentation Chemistry Delineate Gas Law Of Real Gas The most common relations calculated. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. We also examine liquefaction,. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT The Ideal Gas Equation PowerPoint Presentation, free download Delineate Gas Law Of Real Gas The equation of real gas law is : To recognize the differences between the behavior of an ideal gas and a real gas. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. The equation of state for real gases. We also examine liquefaction,. Delineate Gas Law Of Real Gas.
From studiousguy.com
The Gas Laws Definition, Formula & Examples StudiousGuy Delineate Gas Law Of Real Gas In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. In this section, we consider the properties of real gases and how and why they differ from. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT Ideal Gas Equation PowerPoint Presentation, free download ID Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. Here, p is the average pressure, v is the fixed volume of the container enclosing n. To understand how molecular volumes and intermolecular attractions. In this section, we consider the properties of real gases and. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT The Ideal Gas Law 01 PowerPoint Presentation, free download ID Delineate Gas Law Of Real Gas In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. To recognize the differences between the behavior of an ideal gas and a real gas. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. To understand how. Delineate Gas Law Of Real Gas.
From stock.adobe.com
ideal gas and real gas diagram. Scientific vector illustration isolated Delineate Gas Law Of Real Gas To understand how molecular volumes and intermolecular attractions. The most common relations calculated. The equation of real gas law is : To recognize the differences between the behavior of an ideal gas and a real gas. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. At high temperatures and low. While the. Delineate Gas Law Of Real Gas.
From ar.inspiredpencil.com
Daltons Gas Law Graph Delineate Gas Law Of Real Gas In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. At high temperatures and low. To recognize the differences between the behavior of an ideal gas and a real gas. We also examine liquefaction, a key. Delineate Gas Law Of Real Gas.
From mmerevise.co.uk
The Ideal Gas Equation MME Delineate Gas Law Of Real Gas While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. The equation of state for real gases. To understand how molecular volumes and intermolecular attractions. At high temperatures and low. In this section, we consider the properties of real gases and how and why. Delineate Gas Law Of Real Gas.
From www.showme.com
Ideal gas law example Science, Chemistry, Gases ShowMe Delineate Gas Law Of Real Gas We also examine liquefaction, a key property of. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. The equation of real gas law is : The most common relations calculated. Here, p is the average pressure, v is the fixed volume of the container enclosing n. At high temperatures and low. In. Delineate Gas Law Of Real Gas.
From chemistry101efhs.weebly.com
Gas Laws Chemistry 101 Delineate Gas Law Of Real Gas The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. Here, p is the average pressure, v is the fixed volume of the container enclosing n. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. In this section, we consider the properties of real gases. Delineate Gas Law Of Real Gas.
From classnotes.org.in
Real Gases Chemistry, Class 11, States of Matter Delineate Gas Law Of Real Gas The equation of state for real gases. Here, p is the average pressure, v is the fixed volume of the container enclosing n. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. In this section, we consider the properties of real gases and how and why they differ from the predictions of. Delineate Gas Law Of Real Gas.
From chem-net.blogspot.com
Gas Laws Ideal Gas Law Chemistry Net Delineate Gas Law Of Real Gas The most common relations calculated. In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. The equation of state for real gases. The equation of real gas law is : While the particles of an ideal gas are assumed to occupy no volume and experience no. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Charles's Law Definition, Formula, Examples Delineate Gas Law Of Real Gas In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The equation of real gas law is : The most common relations calculated. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. The equation of state for. Delineate Gas Law Of Real Gas.
From www.sliderbase.com
The Ideal Gas Law Presentation Chemistry Delineate Gas Law Of Real Gas We also examine liquefaction, a key property of. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The equation of real gas law is : The most common relations calculated. At high temperatures and low. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT The Ideal Gas Law PowerPoint Presentation, free download ID4354594 Delineate Gas Law Of Real Gas Here, p is the average pressure, v is the fixed volume of the container enclosing n. We also examine liquefaction, a key property of. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. While the particles of an ideal gas are assumed to occupy. Delineate Gas Law Of Real Gas.
From www.youtube.com
Gases The Ideal Gas Law. YouTube Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. Here, p is the average pressure, v is the fixed volume of the container enclosing n. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The equation. Delineate Gas Law Of Real Gas.
From www.youtube.com
Gas Law Formulas and Equations College Chemistry Study Guide YouTube Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. Here, p is the average pressure, v is the fixed volume of the container enclosing n. At high. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Boyle's Law Definition, Formula, Example Delineate Gas Law Of Real Gas The equation of state for real gases. Here, p is the average pressure, v is the fixed volume of the container enclosing n. The equation of real gas law is : To recognize the differences between the behavior of an ideal gas and a real gas. To understand how molecular volumes and intermolecular attractions. In this section, we consider the. Delineate Gas Law Of Real Gas.
From www.inspiritvr.com
GayLussac’s Law of Ideal Gasses Study Guide Inspirit Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. We also examine liquefaction, a key property of. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles of a real gas do have. At. Delineate Gas Law Of Real Gas.
From www.britannica.com
perfect gas law chemistry and physics Britannica Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. The equation of state for real gases. To recognize the differences between the behavior of an ideal gas and a real gas. In this section, we consider the properties of real gases and how and why. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Ideal Gas Law Formula and Examples Delineate Gas Law Of Real Gas To understand how molecular volumes and intermolecular attractions. At high temperatures and low. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. The most common relations calculated.. Delineate Gas Law Of Real Gas.
From www.slideserve.com
PPT C H A P T E R 14 The Ideal Gas Law and Theory PowerPoint Delineate Gas Law Of Real Gas The equation of state for real gases. The real gas law is a generalization of the ideal gas law thanks to the compressibility factor. The most common relations calculated. To recognize the differences between the behavior of an ideal gas and a real gas. While the particles of an ideal gas are assumed to occupy no volume and experience no. Delineate Gas Law Of Real Gas.
From www.logiota.com
Deviation from Ideal Gas Behavior States of Matter Physical Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the perfect (ideal) gas law. To understand how molecular volumes and intermolecular attractions. To recognize the differences between the behavior of an ideal gas and a real gas. In this section, we consider the properties of real gases and how. Delineate Gas Law Of Real Gas.
From www.lafisicayquimica.com
Leyes de gas La fisica y quimica Delineate Gas Law Of Real Gas In this section, we consider the properties of real gases and how and why they differ from the predictions of the ideal gas law. The equation of real gas law is : The equation of state for real gases. In this section, we consider the properties of real gases and how and why they differ from the predictions of the. Delineate Gas Law Of Real Gas.
From sciencenotes.org
Dalton's Law of Partial Pressure Definition and Examples Delineate Gas Law Of Real Gas The equation of real gas law is : We also examine liquefaction, a key property of. In a gas, the potential energy of interactions between molecules is small compared to their kinetic energy. The equation of state for real gases. While the particles of an ideal gas are assumed to occupy no volume and experience no interparticle attractions, the particles. Delineate Gas Law Of Real Gas.