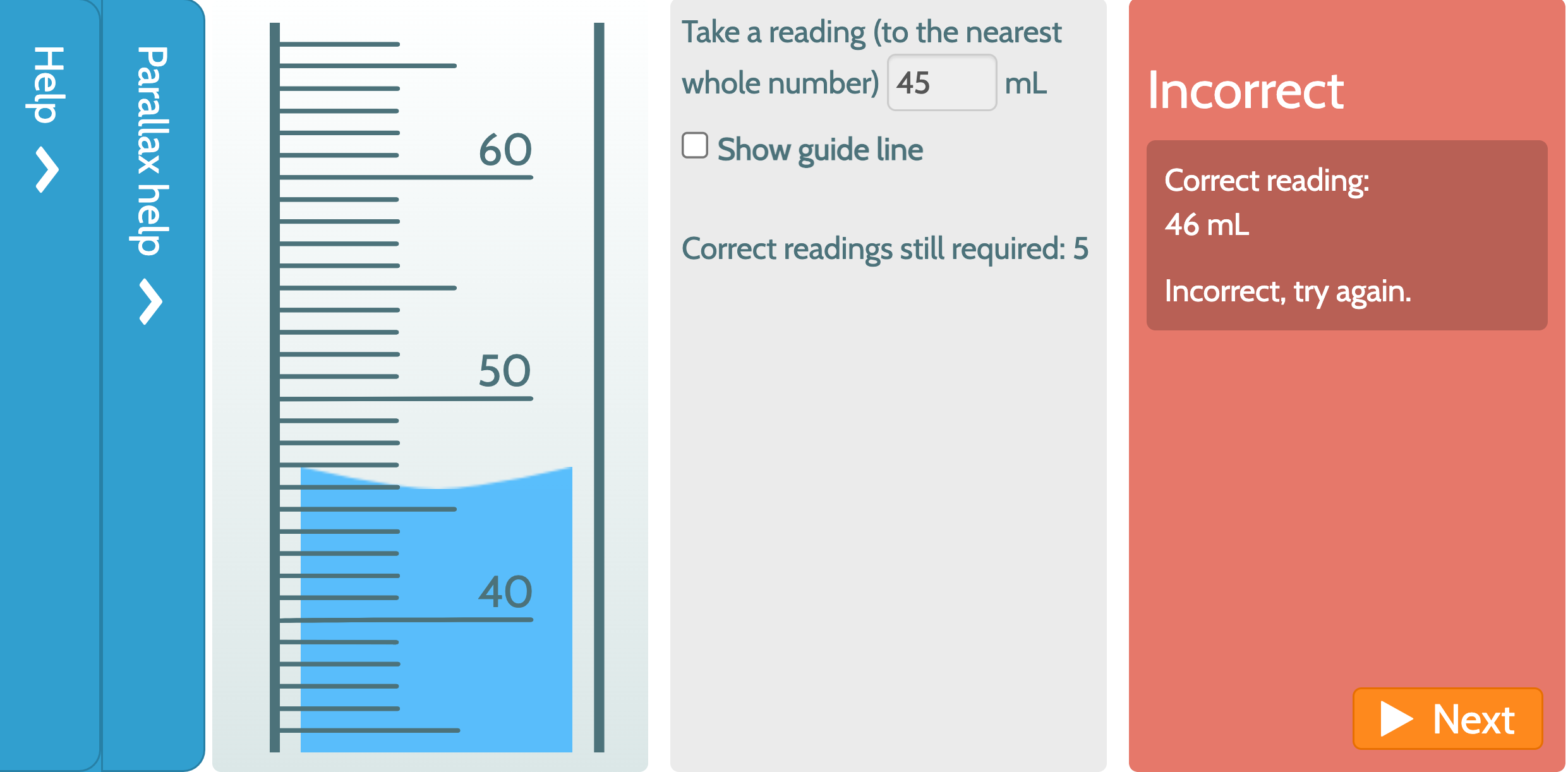
Burette Readings For A Titration Are Shown. What Is The Mean Titre . For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The correct reading is 19.50 ml. During a titration, initial and final burette readings should be taken; Is the volume added (the. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. Burettes are usually marked to a. The steps in a titration are: The first and final reading of the burette should. The steps in a titration. The titre (actual volume delivered) is calculated by difference. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The end point can be. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. The key piece of equipment used in the titration is the burette.
from www.learnsci.com
The titre (actual volume delivered) is calculated by difference. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. Burettes are usually marked to a. The steps in a titration are: For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The end point can be. During a titration, initial and final burette readings should be taken; Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration.
LearnSci LabSim Titration Burette Reading
Burette Readings For A Titration Are Shown. What Is The Mean Titre The steps in a titration. The steps in a titration. The steps in a titration are: The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. The first and final reading of the burette should. The end point can be. The correct reading is 19.50 ml. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The key piece of equipment used in the titration is the burette. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The titre (actual volume delivered) is calculated by difference. For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. Is the volume added (the. Burettes are usually marked to a.
From www.showme.com
How to read a buret Science, Chemistry, Laboratory Skills ShowMe Burette Readings For A Titration Are Shown. What Is The Mean Titre Burettes are usually marked to a. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The steps in a titration. The titre (actual volume delivered) is calculated by difference. Measuring a known volume. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.alamy.com
Titration experiment. Student taking a volume reading from a burette Burette Readings For A Titration Are Shown. What Is The Mean Titre Burettes are usually marked to a. During a titration, initial and final burette readings should be taken; Due to parallax error, the reading can be 19.62 ml or 19.42 ml. Is the volume added (the. The first and final reading of the burette should. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED The Figure shows parts of the burette before and after Burette Readings For A Titration Are Shown. What Is The Mean Titre For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The first and final reading of the burette should. The titre (actual volume delivered) is calculated by difference. The steps. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.microlit.us
Explore What is a Burette, Its Uses, Functions, and Diagrams Burette Readings For A Titration Are Shown. What Is The Mean Titre The key piece of equipment used in the titration is the burette. The end point can be. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. For example,. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.coursehero.com
[Solved] . Burette readings for a titration are shown. Burette readings Burette Readings For A Titration Are Shown. What Is The Mean Titre The key piece of equipment used in the titration is the burette. The steps in a titration. The first and final reading of the burette should. The titre (actual volume delivered) is calculated by difference. The correct reading is 19.50 ml. Burettes are usually marked to a. The steps in a titration are: Due to parallax error, the reading can. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From slidetodoc.com
10 2 Neutralization and AcidBase Titrations Learning Goals Burette Readings For A Titration Are Shown. What Is The Mean Titre The steps in a titration are: Is the volume added (the. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The first and final reading of the burette should. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. The correct reading is 19.50 ml. Titrations are. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.learnsci.com
LearnSci LabSim Titration Burette Reading Burette Readings For A Titration Are Shown. What Is The Mean Titre The correct reading is 19.50 ml. For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The steps in a titration. During a titration, initial and final burette readings should be taken; The titre (actual volume delivered) is calculated by difference. Measuring a. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED The student repeated the titration experiment three times. The Burette Readings For A Titration Are Shown. What Is The Mean Titre The titre (actual volume delivered) is calculated by difference. Is the volume added (the. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The titre close titre volume. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.chegg.com
Solved Burette readings for a titration are shown.What is Burette Readings For A Titration Are Shown. What Is The Mean Titre The titre (actual volume delivered) is calculated by difference. Burettes are usually marked to a. The steps in a titration. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The first and final. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
Titration 1 Filling and reading a burette ScienceWaikato YouTube Burette Readings For A Titration Are Shown. What Is The Mean Titre Due to parallax error, the reading can be 19.62 ml or 19.42 ml. During a titration, initial and final burette readings should be taken; Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. Is the volume added (the. In a titration the liquid in the burette is allowed to slowly run. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED Part B Acid solution pipetted 5.00 mL; Burette readings (NaOH Burette Readings For A Titration Are Shown. What Is The Mean Titre The steps in a titration are: The key piece of equipment used in the titration is the burette. The correct reading is 19.50 ml. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The titre close titre volume of one reactant needed to react completely with the other reactant in a. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.alamy.com
Reading a burette, close up. When reading the burette at the end of a Burette Readings For A Titration Are Shown. What Is The Mean Titre For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. During a titration, initial and final burette readings should be. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From learningschoolbrady88.z13.web.core.windows.net
Reading A Burrette Practice Worksheet Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. The steps in a titration. Burettes are usually marked to a. The key piece of equipment used in the titration is the burette. The correct reading is 19.50 ml. Is the volume added (the. The titre (actual volume delivered) is calculated by difference. Due to parallax error, the reading can be 19.62 ml or 19.42. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
How to read a burette YouTube Burette Readings For A Titration Are Shown. What Is The Mean Titre Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The first and final reading of the burette should. During a titration, initial and final burette readings should be taken; The end point can be. The titre (actual volume delivered) is calculated by difference. The titre close titre volume of one reactant needed to react completely with. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.learnsci.com
LearnSci LabSim Titration Burette Reading Burette Readings For A Titration Are Shown. What Is The Mean Titre The titre (actual volume delivered) is calculated by difference. The first and final reading of the burette should. The key piece of equipment used in the titration is the burette. During a titration, initial and final burette readings should be taken; The steps in a titration. Titrations are done often to find out the concentration of one substance by reacting. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
Year 12 Chemistry Revision Titrating Using the Burette to make an Burette Readings For A Titration Are Shown. What Is The Mean Titre In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The first and final reading of the burette should. The steps in a titration are: For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED The diagram of a burette shown below shows the initial and Burette Readings For A Titration Are Shown. What Is The Mean Titre For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The first and final reading of the burette should. The correct reading is 19.50 ml. In a titration the liquid in the burette is allowed to slowly run into the conical flask until. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.microlit.us
Uses of Burettes in Pharmaceutical Industry Diagrams & Functions Burette Readings For A Titration Are Shown. What Is The Mean Titre In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The titre (actual volume delivered) is calculated by difference. Burettes are usually marked to a. The steps in a titration. The steps in a titration are: For example, if the liquid in your burette rests with the. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED Three titrations were done. Parts of the burette with liquid Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. The steps in a titration are: Is the volume added (the. The key piece of equipment used in the titration is the burette. The steps in a titration. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. During a titration, initial and final burette readings should. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.chegg.com
Solved Read the buret (burette) volume and report your Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. Burettes are usually marked to a. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. In a titration the liquid in the burette is allowed to slowly run into the conical flask until. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From brunofuga.adv.br
Titration Burette Best Online brunofuga.adv.br Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. During a titration, initial and final burette readings should be taken; The steps in a titration. Is the volume added (the. The key piece of equipment used in the titration is the burette. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. Due to parallax error,. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
How to Read a Buret in Chemistry YouTube Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. The steps in. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.numerade.com
SOLVED The student repeated the titration experiment three times. The Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. Is the volume added (the. The titre (actual volume delivered) is calculated by difference. The steps in a titration. In a titration the liquid in the burette is allowed to slowly run into the conical flask. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From mmerevise.co.uk
Titrations and Uncertainties MME Burette Readings For A Titration Are Shown. What Is The Mean Titre The steps in a titration are: The correct reading is 19.50 ml. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. During a titration, initial and final burette readings should be taken; The titre close titre volume of one reactant needed to react completely with the other reactant. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.chegg.com
Solved In a titration the initial volume of titrant in a Burette Readings For A Titration Are Shown. What Is The Mean Titre During a titration, initial and final burette readings should be taken; The steps in a titration. In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The key piece of equipment used in the. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
How to read a burette YouTube Burette Readings For A Titration Are Shown. What Is The Mean Titre In a titration the liquid in the burette is allowed to slowly run into the conical flask until an end point is reached. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. The key piece of equipment used in the titration is the burette. During a titration, initial and final. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.youtube.com
Titration Step 2 Preparing the burette YouTube Burette Readings For A Titration Are Shown. What Is The Mean Titre For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The key piece of equipment used in the titration is the burette. Is the volume added (the. The titre (actual volume delivered) is calculated by difference. Measuring a known volume (usually 20 or. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.gauthmath.com
Solved (ii) The diagram shows the burette readings in one titration Burette Readings For A Titration Are Shown. What Is The Mean Titre The titre (actual volume delivered) is calculated by difference. Is the volume added (the. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. The first and final reading of the burette should. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The correct reading. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From worksheetlistwu.z13.web.core.windows.net
Burette Reading Worksheet Burette Readings For A Titration Are Shown. What Is The Mean Titre Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The first and final reading of the burette should. For example, if the liquid in your burette rests with the 15 ml mark above it and the 20 ml mark below it, the reading is something. The titre (actual volume delivered) is. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.alamy.com
Reading the level of potassium permanganate in a burette. When Burette Readings For A Titration Are Shown. What Is The Mean Titre The correct reading is 19.50 ml. The first and final reading of the burette should. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. Burettes are usually marked to. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From chemistrymadesimple.net
What is Titration and How is it Done? Chemistry Made Simple Burette Readings For A Titration Are Shown. What Is The Mean Titre The correct reading is 19.50 ml. Due to parallax error, the reading can be 19.62 ml or 19.42 ml. The titre (actual volume delivered) is calculated by difference. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. During a titration, initial and final burette readings should be taken;. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.chegg.com
Table 2 Burette readings for titration. (xvi) Using Burette Readings For A Titration Are Shown. What Is The Mean Titre Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The key piece of equipment used in the titration is the burette. The titre close titre volume of one reactant needed to react completely with the other reactant in a titration. The correct reading is 19.50 ml. The steps in a titration.. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From techblog.ctgclean.com
Chemistry What is Titration? CTG Clean Burette Readings For A Titration Are Shown. What Is The Mean Titre Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The titre (actual volume delivered) is calculated by difference. The titre close titre volume of one reactant needed to react completely. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From www.slideserve.com
PPT TITRATION PowerPoint Presentation, free download ID9336460 Burette Readings For A Titration Are Shown. What Is The Mean Titre The end point can be. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a volumetric. The correct reading is 19.50 ml. Titrations are done often to find out the concentration of one substance by reacting it with another substance of known concentration. The steps in a titration. The steps in a titration. Burette Readings For A Titration Are Shown. What Is The Mean Titre.
From theedge.com.hk
Chemistry How To Titration The Edge Burette Readings For A Titration Are Shown. What Is The Mean Titre The key piece of equipment used in the titration is the burette. During a titration, initial and final burette readings should be taken; The end point can be. The steps in a titration are: The titre (actual volume delivered) is calculated by difference. Measuring a known volume (usually 20 or 25 cm 3) of one of the solutions with a. Burette Readings For A Titration Are Shown. What Is The Mean Titre.