What Is A Buffer State Used For . A buffer is an aqueous solution that has a highly stable ph. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. Ions are atoms or molecules that have lost or gained one or more electrons. If you add an acid or a base to a buffered solution, its ph will not change significantly. Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. What is buffer in chemistry? Buffers do so by being composed of certain pairs of solutes: The mechanism involves a buffer, a solution that resists dramatic changes in ph. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. It is able to neutralize small.
from chem.libretexts.org
A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. A buffer is an aqueous solution that has a highly stable ph. Ions are atoms or molecules that have lost or gained one or more electrons. Buffers do so by being composed of certain pairs of solutes: What is buffer in chemistry?
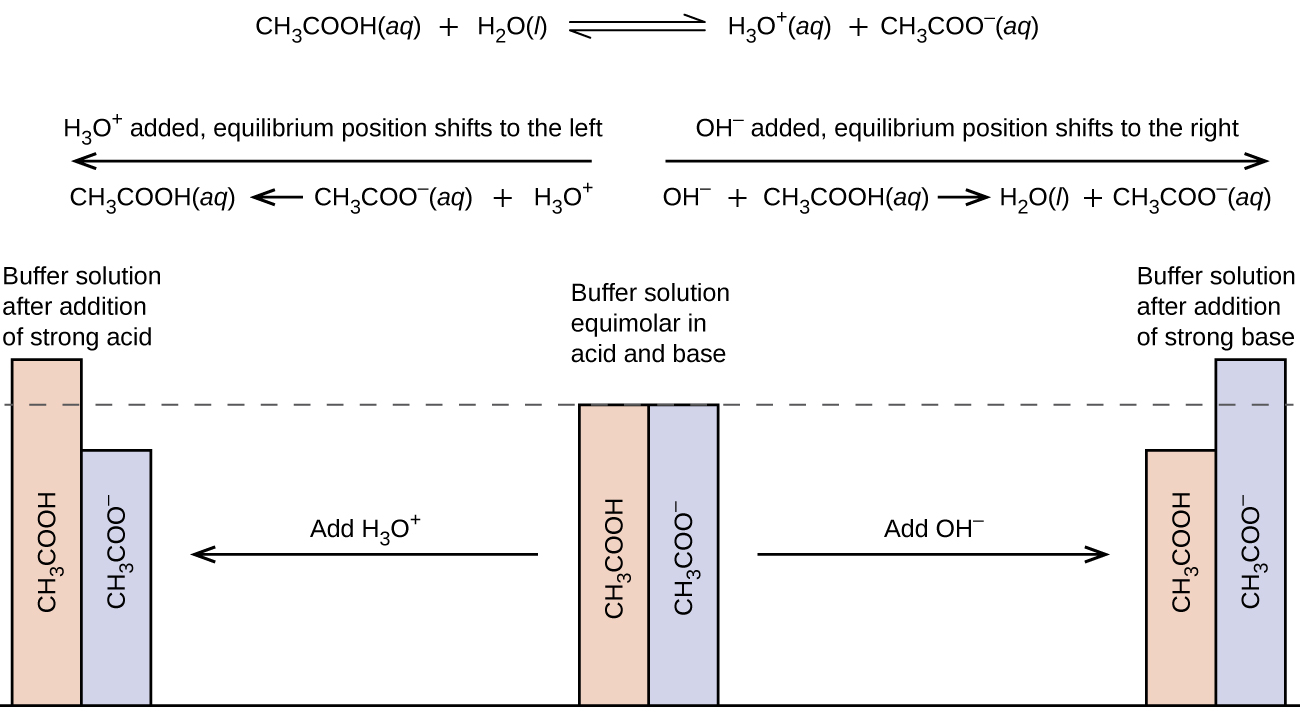
14.6 Buffers Chemistry LibreTexts
What Is A Buffer State Used For Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. A buffer is an aqueous solution that has a highly stable ph. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. The mechanism involves a buffer, a solution that resists dramatic changes in ph. Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. What is buffer in chemistry? A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. If you add an acid or a base to a buffered solution, its ph will not change significantly. This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. It is able to neutralize small. Ions are atoms or molecules that have lost or gained one or more electrons. Buffers do so by being composed of certain pairs of solutes:
From www.researchgate.net
Buffer principle and state diagram Download Scientific Diagram What Is A Buffer State Used For An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. What is buffer in chemistry? A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. Either a weak acid plus a salt derived from that weak acid or a weak. What Is A Buffer State Used For.
From www.slideshare.net
Types of states What Is A Buffer State Used For A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. It is. What Is A Buffer State Used For.
From www.chemistrylearner.com
Buffer Solution Definition, Examples, and Preparation What Is A Buffer State Used For A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. Ions are atoms or molecules that have lost or gained one or more electrons. It is able to neutralize small. This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. Buffer, in chemistry,. What Is A Buffer State Used For.
From www.slideserve.com
PPT PART 4 Salt Hydrolysis and Buffer Solutions PowerPoint What Is A Buffer State Used For Ions are atoms or molecules that have lost or gained one or more electrons. Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. It is able to neutralize small. A buffer is an aqueous solution that has a highly stable ph. If you add an acid. What Is A Buffer State Used For.
From www.slideserve.com
PPT pH and Buffers PowerPoint Presentation ID2948821 What Is A Buffer State Used For The mechanism involves a buffer, a solution that resists dramatic changes in ph. If you add an acid or a base to a buffered solution, its ph will not change significantly. A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. It is able to neutralize small. Ions are atoms or. What Is A Buffer State Used For.
From www.slideserve.com
PPT Digital Logic Design PowerPoint Presentation, free download ID What Is A Buffer State Used For Buffers do so by being composed of certain pairs of solutes: What is buffer in chemistry? Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. It is able to neutralize small. The mechanism involves a buffer, a solution that resists dramatic changes in ph. Ions are. What Is A Buffer State Used For.
From www.slideserve.com
PPT PART 4 Salt Hydrolysis and Buffer Solutions PowerPoint What Is A Buffer State Used For A buffer is an aqueous solution that has a highly stable ph. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. Either a weak acid plus a salt derived from that weak. What Is A Buffer State Used For.
From www.slideserve.com
PPT Unit 9 PowerPoint Presentation, free download ID3733324 What Is A Buffer State Used For A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. Buffers do so by being composed of certain pairs of solutes:. What Is A Buffer State Used For.
From www.slideserve.com
PPT Threestate buffers PowerPoint Presentation, free download ID What Is A Buffer State Used For A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. What is buffer in chemistry? Ions are atoms or molecules that have lost. What Is A Buffer State Used For.
From www.youtube.com
18.2.1 Describe the composition of a buffer solution and explain its What Is A Buffer State Used For What is buffer in chemistry? A buffer is an aqueous solution that has a highly stable ph. Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. Buffers do so by being composed of certain pairs of solutes: A solution whose ph is not altered to any. What Is A Buffer State Used For.
From www.worldatlas.com
What Is The Definition Of A Buffer State? WorldAtlas What Is A Buffer State Used For Ions are atoms or molecules that have lost or gained one or more electrons. It is able to neutralize small. Buffers do so by being composed of certain pairs of solutes: A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. If you add an acid or a base to a. What Is A Buffer State Used For.
From www.slideserve.com
PPT Renal Physiology PowerPoint Presentation, free download ID5632772 What Is A Buffer State Used For This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. It is able to neutralize small. A buffer is an aqueous solution that has a highly stable ph. Ions are atoms or molecules that have lost or gained one or more electrons. Buffers do so by being composed of certain pairs of solutes: Either a. What Is A Buffer State Used For.
From www.pinterest.es
Understanding Buffer Solution Chemistry A Complete Guide What Is A Buffer State Used For If you add an acid or a base to a buffered solution, its ph will not change significantly. A buffer is an aqueous solution that has a highly stable ph. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. An example of a common. What Is A Buffer State Used For.
From www.youtube.com
NCEA L3 Chem Buffers, Buffer Region & Preparation of Buffer Solutions What Is A Buffer State Used For Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. It is able to neutralize small. Ions are atoms or molecules that have lost or gained one or more electrons. A solution whose ph is not altered to any great extent by the addition of small quantities. What Is A Buffer State Used For.
From www.chemistrystudent.com
Buffer Solutions (ALevel) ChemistryStudent What Is A Buffer State Used For Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. The mechanism involves a buffer, a solution that resists dramatic changes in ph. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. This characteristic makes buffers important in biological and chemical. What Is A Buffer State Used For.
From wiringfixarrishes.z21.web.core.windows.net
Tri State Buffer Circuit Diagram What Is A Buffer State Used For Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. What is buffer in chemistry? This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. It is able to neutralize small. The mechanism involves a buffer, a solution that resists dramatic changes. What Is A Buffer State Used For.
From study.com
Buffer System in Chemistry Definition, Function & Examples Lesson What Is A Buffer State Used For This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. The mechanism involves a buffer, a solution that resists dramatic changes in ph. A buffer is an aqueous solution that has a highly stable ph. Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant. What Is A Buffer State Used For.
From www.slideshare.net
Buffer system What Is A Buffer State Used For This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. If you add an acid or a base to a buffered solution, its ph will not change significantly. Ions are atoms or. What Is A Buffer State Used For.
From www.slideserve.com
PPT Unit 9 PowerPoint Presentation, free download ID3733324 What Is A Buffer State Used For Buffers do so by being composed of certain pairs of solutes: A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. If you add an acid or a base to a buffered solution, its ph will not change significantly. Ions are atoms or molecules that. What Is A Buffer State Used For.
From www.slideserve.com
PPT Sea Empire PowerPoint Presentation ID5080078 What Is A Buffer State Used For It is able to neutralize small. What is buffer in chemistry? Buffers do so by being composed of certain pairs of solutes: Ions are atoms or molecules that have lost or gained one or more electrons. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. Buffer, in chemistry, solution usually containing an acid and. What Is A Buffer State Used For.
From manualfixstarks.z13.web.core.windows.net
Tri State Buffer Schematic What Is A Buffer State Used For Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. It is able to neutralize small. Either a weak acid plus a salt derived from that weak acid or a weak base. What Is A Buffer State Used For.
From www.researchgate.net
Visualizing the buffer states. Download Scientific Diagram What Is A Buffer State Used For A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. Buffers do so by being composed of certain pairs of solutes: An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. The mechanism involves a buffer, a solution that resists dramatic changes in. What Is A Buffer State Used For.
From nationalinterest.org
Buffer States Are Worth a Second Look The National Interest What Is A Buffer State Used For Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. An example of a common buffer is a solution of acetic acid. What Is A Buffer State Used For.
From www.slideserve.com
PPT Unit 9 PowerPoint Presentation, free download ID3733324 What Is A Buffer State Used For What is buffer in chemistry? Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. A buffer is an aqueous solution that has. What Is A Buffer State Used For.
From www.slideshare.net
Buffers in chemical analysis, types of buffers What Is A Buffer State Used For A solution whose ph is not altered to any great extent by the addition of small quantities of either an acid or base is. It is able to neutralize small. A buffer is an aqueous solution that has a highly stable ph. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. A buffering agent. What Is A Buffer State Used For.
From www.youtube.com
What is Buffer ? Why Buffer and TriState Buffers are used in Digital What Is A Buffer State Used For This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. Buffers do so by being composed of certain pairs of solutes: If you add an acid or a base to a buffered solution, its ph will not change significantly. A. What Is A Buffer State Used For.
From www.researchgate.net
Buffer principle and state diagram Download Scientific Diagram What Is A Buffer State Used For A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. An example of a common buffer is a solution of acetic acid (ch3cooh) and sodium. Buffers do so by being composed of certain pairs of solutes: If you add an acid or a base to a buffered solution, its ph will. What Is A Buffer State Used For.
From www.slideserve.com
PPT Digital Logic Design PowerPoint Presentation, free download ID What Is A Buffer State Used For This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. The mechanism involves a buffer, a solution that resists dramatic changes in ph. What is buffer in chemistry? Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. Buffer, in chemistry, solution. What Is A Buffer State Used For.
From www.youtube.com
Tristate buffer what is tristate buffer inverting non inverting What Is A Buffer State Used For What is buffer in chemistry? A buffer is an aqueous solution that has a highly stable ph. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. It is able to neutralize small. A buffer is a solution that can resist ph change upon the addition of an. What Is A Buffer State Used For.
From chem.libretexts.org
14.6 Buffers Chemistry LibreTexts What Is A Buffer State Used For Buffers do so by being composed of certain pairs of solutes: Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. Ions. What Is A Buffer State Used For.
From www.slideserve.com
PPT Chapter 3 Processes PowerPoint Presentation, free download ID What Is A Buffer State Used For Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. A buffer is a solution that can resist ph change upon the addition of an. What Is A Buffer State Used For.
From www.slideserve.com
PPT Buffers PowerPoint Presentation, free download ID5687114 What Is A Buffer State Used For A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. The mechanism involves a buffer, a solution that resists dramatic changes in ph. This characteristic makes buffers important in biological and chemical applications where ph stability is crucial. Buffer, in chemistry, solution usually containing an acid and a base, or a. What Is A Buffer State Used For.
From www.youtube.com
CONSTRUCTION OF BUS SYSTEM WITH THREE STATE BUFFERS YouTube What Is A Buffer State Used For A buffer is an aqueous solution that has a highly stable ph. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. Buffer, in chemistry, solution usually containing an acid and a base, or a salt, that tends to maintain a constant hydrogen ion concentration. This characteristic makes. What Is A Buffer State Used For.
From slidetodoc.com
Introduction to Buffers These solutions contain relatively high What Is A Buffer State Used For Either a weak acid plus a salt derived from that weak acid or a weak base plus a salt of that weak base. A buffer is a solution that maintains the stability of a system’s ph level when adding small quantities of acids or bases. A buffering agent is a weak acid or weak base that helps maintain the ph. What Is A Buffer State Used For.
From sciencenotes.org
Buffer Definition and Examples in Chemistry What Is A Buffer State Used For A buffering agent is a weak acid or weak base that helps maintain the ph of an aqueous solution after adding another acid or base. It is able to neutralize small. The mechanism involves a buffer, a solution that resists dramatic changes in ph. Buffers do so by being composed of certain pairs of solutes: A buffer is a solution. What Is A Buffer State Used For.