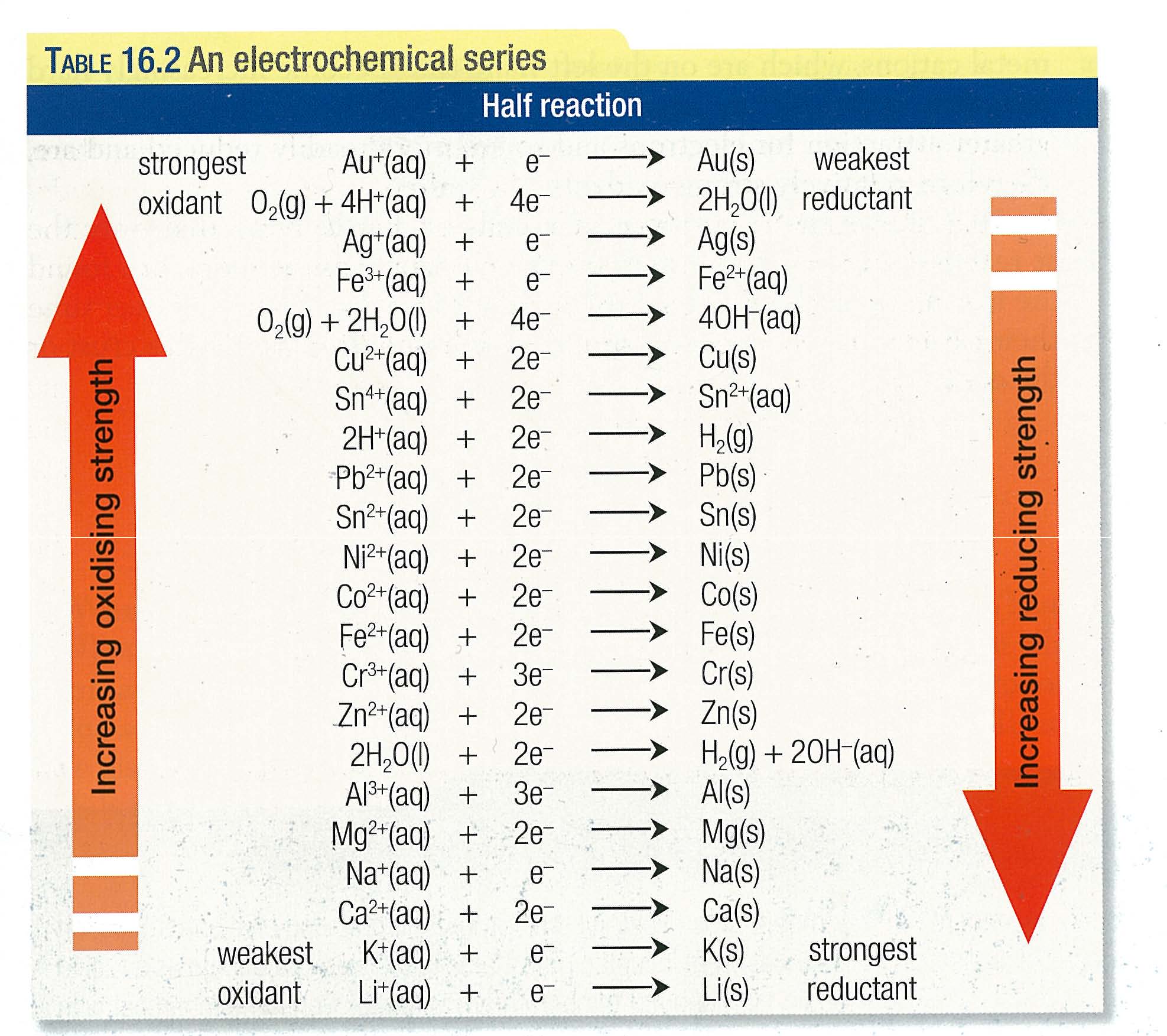
Electrochemical Equivalent Of Cu In The Reaction . Electrochemical equivalence = equivalent mass f = z. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance is the mass of the substance deposited to one of the electrodes when a. Equivalent mass of cu = m olar mass. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox.
from socratic.org
Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f = z. Of a substance is the mass of the substance deposited to one of the electrodes when a. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$.
In which of the following solution metallic ion can be displaced by
Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Equivalent mass of cu = m olar mass. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Electrochemical equivalence = equivalent mass f = z. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance is the mass of the substance deposited to one of the electrodes when a.
From guidemanualcodas.z1.web.core.windows.net
Diagram Of Voltaic Cell Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f = z. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of. Electrochemical Equivalent Of Cu In The Reaction.
From www.researchgate.net
The mechanism for CO reduction on Cu that shows how it splits at Electrochemical Equivalent Of Cu In The Reaction to find the electrochemical equivalent (z) of copper (cu) in the reaction: we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Equivalent mass of cu = m olar mass. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Of. Electrochemical Equivalent Of Cu In The Reaction.
From www.researchgate.net
Electrochemical properties of Cu/C at different loading a, FE of Electrochemical Equivalent Of Cu In The Reaction Of a substance is the mass of the substance deposited to one of the electrodes when a. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have to know that copper is a substance component with the. Electrochemical Equivalent Of Cu In The Reaction.
From www.youtube.com
Emf of daniel cell from nernst equation(Electrochemistry part 28 for Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f = z. Of a substance is the mass of the substance deposited to one of the electrodes when a. Cu 2 + + 2 e − → cu ( s ) we will follow these. Electrochemical Equivalent Of Cu In The Reaction.
From leverageedu.com
Electrochemical Series Notes Chemistry Class 11 & 12 Leverage Edu Electrochemical Equivalent Of Cu In The Reaction Equivalent mass of cu = m olar mass. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance. Electrochemical Equivalent Of Cu In The Reaction.
From www.youtube.com
Photoelectrochemical reduction of CO2 using Cu2O YouTube Electrochemical Equivalent Of Cu In The Reaction to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance is the mass of the substance deposited to one of the electrodes when a. Equivalent mass of cu = m olar mass. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Cu 2 +. Electrochemical Equivalent Of Cu In The Reaction.
From www.chemicals.co.uk
A Level Chemistry Electrodes & Electrochemical Cells Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical. Electrochemical Equivalent Of Cu In The Reaction.
From www.dynamicscience.com.au
redox reactions electrolysis The difference between electrochemical Electrochemical Equivalent Of Cu In The Reaction Of a substance is the mass of the substance deposited to one of the electrodes when a. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Electrochemical equivalence = equivalent mass f = z. the electrochemical. Electrochemical Equivalent Of Cu In The Reaction.
From byjus.com
Electrochemical equivalent of Cu in the reaction Cu2+(aq) + 2e > Cu is Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Equivalent mass of cu = m olar mass. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of. Electrochemical Equivalent Of Cu In The Reaction.
From www.mdpi.com
Catalysts Free FullText DFT Study of CO2 Reduction Reaction to Electrochemical Equivalent Of Cu In The Reaction to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Of a substance is the mass of the substance deposited to one of the electrodes when a. we have. Electrochemical Equivalent Of Cu In The Reaction.
From alevelchemistry.co.uk
Electrochemical Cells Definition, Description & Types Electrochemical Equivalent Of Cu In The Reaction to find the electrochemical equivalent (z) of copper (cu) in the reaction: Electrochemical equivalence = equivalent mass f = z. Of a substance is the mass of the substance deposited to one of the electrodes when a. Equivalent mass of cu = m olar mass. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of. Electrochemical Equivalent Of Cu In The Reaction.
From www.mdpi.com
Processes Free FullText Progress in Electroreduction of CO2 to Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Electrochemical equivalence = equivalent mass f = z. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Of. Electrochemical Equivalent Of Cu In The Reaction.
From askfilo.com
Electrochemical equivalent of Cu in the reaction Filo Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Equivalent mass of cu = m olar mass. Of a substance. Electrochemical Equivalent Of Cu In The Reaction.
From www.meritnation.com
electrochemical equivalent of cu in the given reaction Chemistry Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Electrochemical equivalence = equivalent mass f = z. Equivalent mass of cu = m olar mass. Of a substance is the mass of the substance deposited to one. Electrochemical Equivalent Of Cu In The Reaction.
From exomnwzaf.blob.core.windows.net
What Is The Electrochemical Equivalent at Marcus Evenson blog Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Of a substance is the mass of the substance deposited to one of the electrodes when a. the electrochemical equivalent is a crucial. Electrochemical Equivalent Of Cu In The Reaction.
From www.mdpi.com
ChemEngineering Free FullText Electrochemical Carbon Dioxide Electrochemical Equivalent Of Cu In The Reaction Of a substance is the mass of the substance deposited to one of the electrodes when a. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Equivalent mass of cu = m olar mass. Electrochemical equivalence = equivalent mass. Electrochemical Equivalent Of Cu In The Reaction.
From chemistry-europe.onlinelibrary.wiley.com
Influence of Different Dissolved Gases on Electrocatalytic Nitrate Electrochemical Equivalent Of Cu In The Reaction Equivalent mass of cu = m olar mass. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance. Electrochemical Equivalent Of Cu In The Reaction.
From www.researchgate.net
(a) CNTbased electrochemical sensor for the detection of bisphenolA Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Of a substance is the mass of the substance deposited to one of the electrodes when a. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. we have. Electrochemical Equivalent Of Cu In The Reaction.
From dxocnyazy.blob.core.windows.net
Electrolysis Oil And Gas at Victor Serrano blog Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Equivalent mass of cu = m olar mass. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Electrochemical equivalence =. Electrochemical Equivalent Of Cu In The Reaction.
From dxontbmym.blob.core.windows.net
Electrode Meaning Chemistry at Christy Rogers blog Electrochemical Equivalent Of Cu In The Reaction Equivalent mass of cu = m olar mass. Of a substance is the mass of the substance deposited to one of the electrodes when a. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Electrochemical equivalence = equivalent mass f = z. to find the electrochemical equivalent (z) of. Electrochemical Equivalent Of Cu In The Reaction.
From english.whut.edu.cn
Wuhan University of Technology Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Of a substance is the mass of the substance deposited to one of the electrodes when a. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f =. Electrochemical Equivalent Of Cu In The Reaction.
From monomole.com
Pourbaix diagram Mono Mole Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. the electrochemical. Electrochemical Equivalent Of Cu In The Reaction.
From socratic.org
In which of the following solution metallic ion can be displaced by Electrochemical Equivalent Of Cu In The Reaction Equivalent mass of cu = m olar mass. Of a substance is the mass of the substance deposited to one of the electrodes when a. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: to find the electrochemical equivalent (z) of copper (cu) in the reaction: Electrochemical equivalence = equivalent mass. Electrochemical Equivalent Of Cu In The Reaction.
From chemistry.stackexchange.com
electrochemistry Why does the anode solution contain Sn2+ in a SnCu Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f = z. Equivalent mass of cu = m olar mass. to find the electrochemical equivalent (z) of. Electrochemical Equivalent Of Cu In The Reaction.
From quizdbmonarchist.z21.web.core.windows.net
How To Calculate E Cell Chemistry Electrochemical Equivalent Of Cu In The Reaction Of a substance is the mass of the substance deposited to one of the electrodes when a. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical. Electrochemical Equivalent Of Cu In The Reaction.
From scitechdaily.com
Catalytic Hydrogenation of CO2 to Methanol Low Temperature and High Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have. Electrochemical Equivalent Of Cu In The Reaction.
From peacecommission.kdsg.gov.ng
Conversion Chart Template Google Sheets, Google Slides, Excel Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Of a substance is the mass of the substance deposited to. Electrochemical Equivalent Of Cu In The Reaction.
From www.researchgate.net
A) Electrochemical equivalent circuit used for modeling the EIS data Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Of a substance is the mass of the substance. Electrochemical Equivalent Of Cu In The Reaction.
From schoolbag.info
Electrochemical Cells Electrochemistry Training MCAT General Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Electrochemical equivalence = equivalent mass f = z. we have to know that copper is a substance component with the symbol $cu$ and. Electrochemical Equivalent Of Cu In The Reaction.
From www.chegg.com
Solved List the electrochemical series you developed from Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu. Electrochemical Equivalent Of Cu In The Reaction.
From www.sarthaks.com
In a Cuvoltameter, mass deposited in 30 s is m gm. If the timecurrent Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: to find the electrochemical. Electrochemical Equivalent Of Cu In The Reaction.
From schematicfixcinnamon.z5.web.core.windows.net
Electron Configuration Cu 1 Orbital Diagram Electrochemical Equivalent Of Cu In The Reaction to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. Cu 2 + + 2 e − → cu ( s ) we will follow these steps: we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Of a substance. Electrochemical Equivalent Of Cu In The Reaction.
From pubs.acs.org
Electrocatalytic Reduction of Nitrate to Ammonia via a Au/Cu Single Electrochemical Equivalent Of Cu In The Reaction Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Electrochemical equivalence = equivalent mass f = z. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. Equivalent mass of cu = m olar mass. Of a substance is the mass of the. Electrochemical Equivalent Of Cu In The Reaction.
From 2012books.lardbucket.org
Electrochemistry Electrochemical Equivalent Of Cu In The Reaction we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Cu 2 + + 2 e − → cu ( s ) we will follow these steps: Electrochemical equivalence = equivalent mass f = z. Equivalent mass of. Electrochemical Equivalent Of Cu In The Reaction.
From physicsworld.com
Oxygen and carbon monoxide electrocatalysis for renewableenergy Electrochemical Equivalent Of Cu In The Reaction the electrochemical equivalent is a crucial concept in electrochemistry, quantifying the amount of substance involved in a redox. we have to know that copper is a substance component with the symbol $cu$ and nuclear number $29$. to find the electrochemical equivalent (z) of copper (cu) in the reaction: Equivalent mass of cu = m olar mass. Cu. Electrochemical Equivalent Of Cu In The Reaction.