Category 8 In-Vitro Diagnostic Medical Devices . (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. Definition of in vitro diagnostic medical devices regulation for registration of medical devices:
from laegemiddelstyrelsen.dk
(article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,.
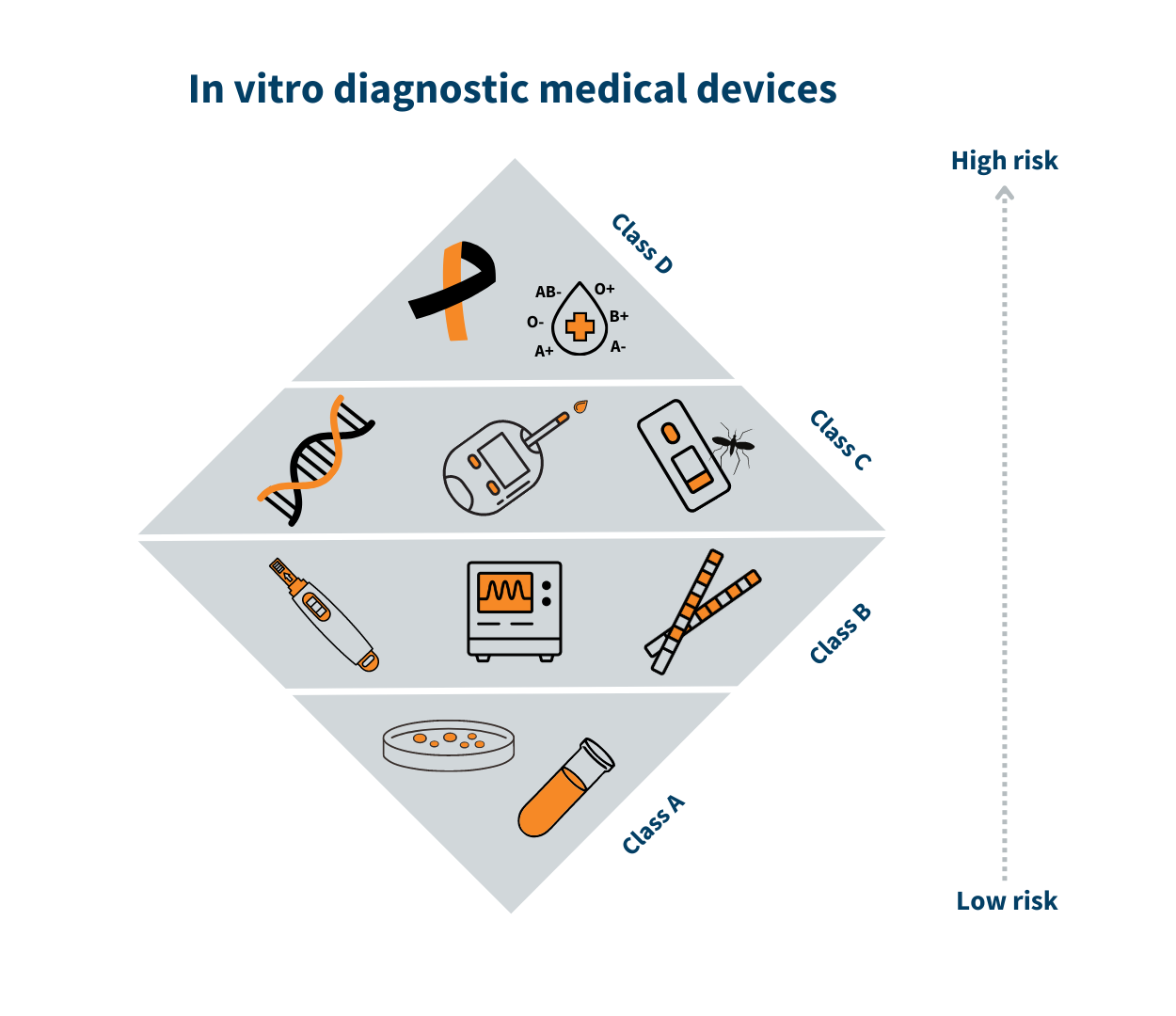
Classification of in vitro diagnostic medical devices (IVD)
Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Definition of in vitro diagnostic medical devices regulation for registration of medical devices:
From vantagemedtech.com
Deciphering In Vitro Diagnostics (IVD) Medical Device Regulations Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017. Category 8 In-Vitro Diagnostic Medical Devices.
From www.kplc.or.kr
CEIVDR(In Vitro Diagnostic Medical Devices Regulation) 한국PL센터 Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april. Category 8 In-Vitro Diagnostic Medical Devices.
From www.psi-software.com
In Vitro Diagnostics (IVD) Device Precision Systems, Inc. Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices. Category 8 In-Vitro Diagnostic Medical Devices.
From apacmed.org
What Is In Vitro Diagnostics (IVD) Types, Benefits & Regulations Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Regulation (eu) 2017/745 of the european parliament and of. Category 8 In-Vitro Diagnostic Medical Devices.
From qbdgroup.com
IVDR classification of invitro diagnostic medical devices a brief guide Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. On may 26, 2022, after a transitional period of 5 years,. Category 8 In-Vitro Diagnostic Medical Devices.
From lsacademy.com
Performance Evaluation of InVitroDiagnostic Devices (IVDs) LS Academy Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: ‘in vitro diagnostic medical. Category 8 In-Vitro Diagnostic Medical Devices.
From www.clinicalresearchassociatecra.com
IVD Clinical Trials & Development Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. (article 9) in vitro diagnostic device (hereafter. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic. Category 8 In-Vitro Diagnostic Medical Devices.
From www.vrogue.co
Eu Ivd Approval Process For Medical Devices vrogue.co Category 8 In-Vitro Diagnostic Medical Devices This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices. Category 8 In-Vitro Diagnostic Medical Devices.
From www.en-standard.eu
BS EN ISO 67172021 In vitro diagnostic medical devices. Singleuse Category 8 In-Vitro Diagnostic Medical Devices (article 9) in vitro diagnostic device (hereafter. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu). Category 8 In-Vitro Diagnostic Medical Devices.
From qbdgroup.com
IVDR classification of invitro diagnostic medical devices a brief guide Category 8 In-Vitro Diagnostic Medical Devices (article 9) in vitro diagnostic device (hereafter. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Regulation (eu) 2017/746 of the european parliament and of the council of. Category 8 In-Vitro Diagnostic Medical Devices.
From www.joharidigital.com
In Vitro Diagnostic (IVD) Device Manufacturer Medical Device Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic. Category 8 In-Vitro Diagnostic Medical Devices.
From www.slideserve.com
PPT Best In Vitro Diagnostic & Point Of Care Testing Medical Device Category 8 In-Vitro Diagnostic Medical Devices ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/745 of the european parliament and of the council of. Category 8 In-Vitro Diagnostic Medical Devices.
From operonstrategist.com
Guide to In Vitro Diagnostic Medical Device Regulation (IVDR) IVD Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu) 2017/745 of the. Category 8 In-Vitro Diagnostic Medical Devices.
From exoiuwfph.blob.core.windows.net
In Vitro Diagnostic Medical Device Performance Evaluation at Marion Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. Regulation (eu) 2017/746 of the. Category 8 In-Vitro Diagnostic Medical Devices.
From www.slideshare.net
Regulation of In Vitro Diagnostic Medical Devices Transition to the… Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. ‘in vitro. Category 8 In-Vitro Diagnostic Medical Devices.
From www.tuvsud.com
Infographic The In Vitro Diagnostic Medical Device Regulation TÜV Category 8 In-Vitro Diagnostic Medical Devices This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017. Category 8 In-Vitro Diagnostic Medical Devices.
From www.qvccert.com
InVitro Diagnostic Medical Devices CE Marking CE Marking, Ce Mark Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Definition of in vitro diagnostic medical devices regulation for registration of medical devices: Regulation (eu) 2017/745 of the european. Category 8 In-Vitro Diagnostic Medical Devices.
From g-medtech.com
InVitro Diagnostics GMedTech Marketplace Category 8 In-Vitro Diagnostic Medical Devices (article 9) in vitro diagnostic device (hereafter. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in. Category 8 In-Vitro Diagnostic Medical Devices.
From imgbin.com
In Vitro Diagnostics Medical Device Medical Diagnosis Medical Equipment Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and. Category 8 In-Vitro Diagnostic Medical Devices.
From interhospi.com
EU sets out new regulations on in vitro diagnostic medical devices Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic. Category 8 In-Vitro Diagnostic Medical Devices.
From www.castoredc.com
InVitro Diagnostic Regulation (IVDR) From oversight to overhead Castor Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending. Category 8 In-Vitro Diagnostic Medical Devices.
From studylib.net
In Vitro Diagnostic Device Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. (article 9) in vitro diagnostic device (hereafter. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Definition of in vitro diagnostic medical devices regulation for registration of. Category 8 In-Vitro Diagnostic Medical Devices.
From www.healthcareradius.in
Invitro diagnostics (IVD) trends 2023 and beyond Healthcare Radius Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. (article 9) in vitro diagnostic device (hereafter. This guidance, relating to the application of regulation (eu) 2017/746. Category 8 In-Vitro Diagnostic Medical Devices.
From www.mdpi.com
A Systematic Database Approach to Identify Companion Diagnostic Testing Category 8 In-Vitro Diagnostic Medical Devices ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic. Category 8 In-Vitro Diagnostic Medical Devices.
From gsap.co.il
In Vitro Diagnostic Medical Device (IVD) in the EU Gsap Category 8 In-Vitro Diagnostic Medical Devices This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. (article 9) in vitro diagnostic device (hereafter. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/745 of the european parliament and of the council of. Category 8 In-Vitro Diagnostic Medical Devices.
From www.slideserve.com
PPT IVD and Point of care testing PowerPoint Presentation, free Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. Regulation (eu) 2017/745 of the european parliament and. Category 8 In-Vitro Diagnostic Medical Devices.
From pacificcert.com
ISO 236402011In vitro diagnostic medical devices Pacific Certifications Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. Regulation (eu) 2017/746 of the european parliament and of. Category 8 In-Vitro Diagnostic Medical Devices.
From www.greenlight.guru
IVDR for In Vitro Diagnostic Medical Device Companies Ultimate Guide Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic. Category 8 In-Vitro Diagnostic Medical Devices.
From www.investindia.gov.in
In Vitro Diagnostics The Silent Champion of Medical Devices Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in. Category 8 In-Vitro Diagnostic Medical Devices.
From www.nsf.org
In Vitro Diagnostic Medical Device Regulation… NSF International Category 8 In-Vitro Diagnostic Medical Devices This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses the. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices.. Category 8 In-Vitro Diagnostic Medical Devices.
From stock.adobe.com
In vitro diagnostic medical device symbol. Stock Vector Adobe Stock Category 8 In-Vitro Diagnostic Medical Devices Definition of in vitro diagnostic medical devices regulation for registration of medical devices: On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices,. Category 8 In-Vitro Diagnostic Medical Devices.
From laegemiddelstyrelsen.dk
Classification of in vitro diagnostic medical devices (IVD) Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. (article 9) in vitro diagnostic device (hereafter. Regulation (eu) 2017/745 of the european parliament and of the council. Category 8 In-Vitro Diagnostic Medical Devices.
From www.thema-med.com
In Vitro Diagnostic Medical Devices proposed extension of IVDR 2017/746 Category 8 In-Vitro Diagnostic Medical Devices Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. Regulation (eu) 2017/746 of the european parliament and of the council of 5 april 2017 on in vitro diagnostic medical devices and repealing. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr). Category 8 In-Vitro Diagnostic Medical Devices.
From dicentra.com
EU In Vitro Diagnostic Medical Device Regulation dicentra Category 8 In-Vitro Diagnostic Medical Devices (article 9) in vitro diagnostic device (hereafter. ‘in vitro diagnostic medical device’ means any medical device which uses reagents and their products, calibrators, control materials, kits,. On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Definition of in vitro diagnostic medical devices regulation for registration of medical. Category 8 In-Vitro Diagnostic Medical Devices.
From www.pewtrusts.org
What Are In Vitro Diagnostic Tests, and How Are They Regulated? The Category 8 In-Vitro Diagnostic Medical Devices On may 26, 2022, after a transitional period of 5 years, the new regulation (eu) 2017/746 on in vitro diagnostic medical devices. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive. This guidance, relating to the application of regulation (eu) 2017/746 on in vitro diagnostic medical devices (ivdr) addresses. Category 8 In-Vitro Diagnostic Medical Devices.