Definition Of Bombarded In Chemistry . Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. The effect is known as ion mixing. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. Step by step radioactive decay and bombardment. Because of this, neutrons do not. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The “attractive” nuclear force between protons and neutrons is.
from www.chemistrylearner.com
The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. The effect is known as ion mixing. The “attractive” nuclear force between protons and neutrons is. Because of this, neutrons do not. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Step by step radioactive decay and bombardment. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron!
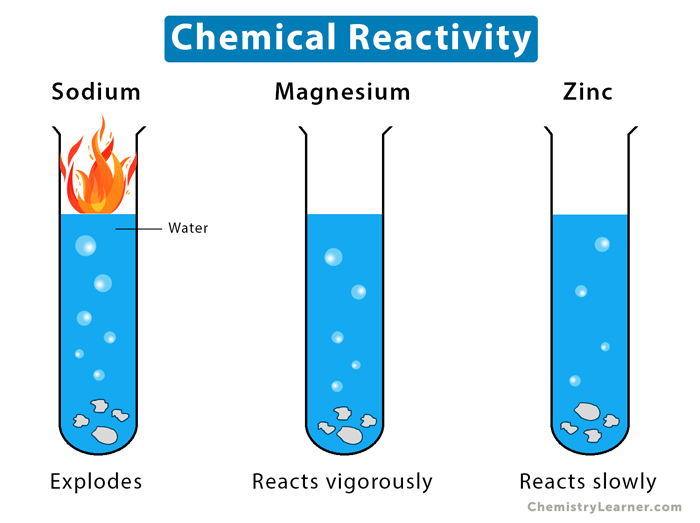
Reactivity Series Definition and Chart
Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. The “attractive” nuclear force between protons and neutrons is. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Because of this, neutrons do not. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The effect is known as ion mixing. Step by step radioactive decay and bombardment. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions.
From sciencenotes.org
Precipitation Reaction Definition and Examples in Chemistry Definition Of Bombarded In Chemistry Because of this, neutrons do not. Step by step radioactive decay and bombardment. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The “attractive” nuclear force between protons and neutrons. Definition Of Bombarded In Chemistry.
From socratic.org
What part of an atom is involved in nuclear reactions? Socratic Definition Of Bombarded In Chemistry The “attractive” nuclear force between protons and neutrons is. Because of this, neutrons do not. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. Step by step radioactive decay and. Definition Of Bombarded In Chemistry.
From www.youtube.com
What is chemistry \ Define chemistry YouTube Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has.. Definition Of Bombarded In Chemistry.
From exoglnfqg.blob.core.windows.net
Bombarded Meaning In Malayalam at David Noonan blog Definition Of Bombarded In Chemistry The effect is known as ion mixing. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. Step by step radioactive decay and bombardment. The “attractive” nuclear. Definition Of Bombarded In Chemistry.
From www.vedantu.com
Chemicals Learn Definition, Properties, Types and Examples Definition Of Bombarded In Chemistry There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The effect is known as ion mixing. The “attractive” nuclear force between protons and neutrons is. Because of this, neutrons do not. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of. Definition Of Bombarded In Chemistry.
From www.tpsearchtool.com
Chemistry Basics Overview Images Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. Because of this, neutrons do not. The “attractive” nuclear force between protons and neutrons is. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single. Definition Of Bombarded In Chemistry.
From www.thoughtco.com
10 Amazing Chemical Reactions Definition Of Bombarded In Chemistry Because of this, neutrons do not. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. The “attractive” nuclear force between protons and neutrons. Definition Of Bombarded In Chemistry.
From exowfrlrq.blob.core.windows.net
Bombarded Meaning In Chemistry at Thomas Carroll blog Definition Of Bombarded In Chemistry Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances. Definition Of Bombarded In Chemistry.
From chem.libretexts.org
20.7 The Discovery of Fission The Atomic Bomb and Nuclear Power Definition Of Bombarded In Chemistry Because of this, neutrons do not. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The effect is known as ion mixing. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The penetrating ion loses energy both as a. Definition Of Bombarded In Chemistry.
From www.aakash.ac.in
Discovery of Protons and Neutrons in Chemistry Definition, Types and Definition Of Bombarded In Chemistry Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The effect is known as ion mixing. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The “attractive” nuclear force between protons and neutrons is. Fast atom bombardment (fab) and. Definition Of Bombarded In Chemistry.
From www.worksheetsplanet.com
What Is A Chemical Reaction Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei. Definition Of Bombarded In Chemistry.
From giohksjnu.blob.core.windows.net
Another Word To Say Bombarded at David Espinosa blog Definition Of Bombarded In Chemistry The “attractive” nuclear force between protons and neutrons is. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to. Definition Of Bombarded In Chemistry.
From sciencenotes.org
What Is a Mixture in Chemistry? Definition and Examples Definition Of Bombarded In Chemistry Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Because of this, neutrons do not. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. There is another type of nuclear emission that results in a very unusual particle, a positron. Definition Of Bombarded In Chemistry.
From sciencenotes.org
Alpha Particle Definition, Symbol and Charge Definition Of Bombarded In Chemistry The effect is known as ion mixing. Step by step radioactive decay and bombardment. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Since a neutron. Definition Of Bombarded In Chemistry.
From www.slideserve.com
PPT Nuclear Chemistry PowerPoint Presentation, free download ID1964800 Definition Of Bombarded In Chemistry There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The effect is known as ion mixing. Since a neutron has no charge, it is not electrostatically repelled by the nucleus. Definition Of Bombarded In Chemistry.
From sciencenotes.org
Catenation Definition and Examples in Chemistry Definition Of Bombarded In Chemistry The “attractive” nuclear force between protons and neutrons is. The effect is known as ion mixing. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Because of this, neutrons do not.. Definition Of Bombarded In Chemistry.
From sdrc.stanford.edu
We are bombarded by thousands of diverse species and chemicals Definition Of Bombarded In Chemistry The effect is known as ion mixing. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. Step by step radioactive decay and bombardment. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The penetrating ion. Definition Of Bombarded In Chemistry.
From www.worksheetsplanet.com
What is Chemistry Definition of Chemistry Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The penetrating ion loses energy both as. Definition Of Bombarded In Chemistry.
From www.theengineeringprojects.com
What is Chemistry? Definition, Branches, Books and Scientists The Definition Of Bombarded In Chemistry Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. The “attractive” nuclear force between protons and neutrons is. The effect is known as ion mixing. There is another type of. Definition Of Bombarded In Chemistry.
From www.youtube.com
Define of Chemical Hazards What is Chemical Hazards Types of Definition Of Bombarded In Chemistry Because of this, neutrons do not. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. The effect is known as ion mixing. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use. Definition Of Bombarded In Chemistry.
From www.tessshebaylo.com
Uranium 234 Alpha Decay Equation Tessshebaylo Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. The effect is known as ion mixing. The “attractive” nuclear force between protons and neutrons is. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Step by step radioactive decay and. Definition Of Bombarded In Chemistry.
From ar.inspiredpencil.com
Basic Atomic Structure Definition Definition Of Bombarded In Chemistry The effect is known as ion mixing. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. The “attractive” nuclear force between protons and neutrons is. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. There is another type of nuclear. Definition Of Bombarded In Chemistry.
From www.chemistrylearner.com
Reactivity Series Definition and Chart Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The “attractive” nuclear force between protons and neutrons is. Because of this, neutrons do not. Since a neutron has no charge,. Definition Of Bombarded In Chemistry.
From www.youtube.com
How to Say Bombard in English? How Does Bombard Look? What is Definition Of Bombarded In Chemistry Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The effect is known as ion mixing. The penetrating ion loses energy both as a consequence of. Definition Of Bombarded In Chemistry.
From www.studocu.com
CH.1.1 Chemistry and Chemicals 1 Chemistry and Chemicals Learning Definition Of Bombarded In Chemistry As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The “attractive” nuclear force between protons and neutrons is. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and. Definition Of Bombarded In Chemistry.
From sciencenotes.org
What Is a Salt in Chemistry? Definition and Examples Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Fast. Definition Of Bombarded In Chemistry.
From giohksjnu.blob.core.windows.net
Another Word To Say Bombarded at David Espinosa blog Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! The effect is known as ion mixing. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step.. Definition Of Bombarded In Chemistry.
From www.slideshare.net
Nuclear Chemistry Powerpoint 2003 Definition Of Bombarded In Chemistry The effect is known as ion mixing. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. Because of this, neutrons do not. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy atoms to sputter and ionize the sample in a single step. The. Definition Of Bombarded In Chemistry.
From sciencenotes.org
What Is a Chemical Reaction? Definition and Examples Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. The “attractive” nuclear force between protons and neutrons is. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Since a. Definition Of Bombarded In Chemistry.
From giohksjnu.blob.core.windows.net
Another Word To Say Bombarded at David Espinosa blog Definition Of Bombarded In Chemistry The “attractive” nuclear force between protons and neutrons is. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. Step by step radioactive decay and bombardment. Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. As an example, consider thin film a on substrate. Definition Of Bombarded In Chemistry.
From www.thoughtco.com
Aqueous Solution Definition in Chemistry Definition Of Bombarded In Chemistry Step by step radioactive decay and bombardment. Because of this, neutrons do not. Since a neutron has no charge, it is not electrostatically repelled by the nucleus it is bombarding. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Fast atom bombardment (fab) and secondary ion mass spectrometry. Definition Of Bombarded In Chemistry.
From exoigesah.blob.core.windows.net
Bombarded By In A Sentence at Joy Irby blog Definition Of Bombarded In Chemistry The effect is known as ion mixing. The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The “attractive” nuclear force between protons and neutrons is. Fast atom bombardment (fab) and. Definition Of Bombarded In Chemistry.
From www.worksheetsplanet.com
What is a pure substance Definition Definition Of Bombarded In Chemistry The “attractive” nuclear force between protons and neutrons is. Step by step radioactive decay and bombardment. There is another type of nuclear emission that results in a very unusual particle, a positron or a positively charged electron! Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. The penetrating ion loses energy. Definition Of Bombarded In Chemistry.
From saylordotorg.github.io
Nuclear Chemistry Definition Of Bombarded In Chemistry The penetrating ion loses energy both as a consequence of elastic collisions with target nuclei and of excitations and ionizations of. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. The effect is known as ion mixing. Fast atom bombardment (fab) and secondary ion mass spectrometry (sims) both use high energy. Definition Of Bombarded In Chemistry.
From dictionary.langeek.co
Definition & Meaning of "Chemical science" LanGeek Definition Of Bombarded In Chemistry Particle collisions play a fundamental role in the field of chemistry, where the concept of bombarding substances has. As an example, consider thin film a on substrate b bombarded by a beam of inert gas ions. Step by step radioactive decay and bombardment. The “attractive” nuclear force between protons and neutrons is. Because of this, neutrons do not. Since a. Definition Of Bombarded In Chemistry.