How To Find Delta S Surroundings From Delta S System . We have seen that the energy given off (or absorbed) by a reaction,. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). If this equation is replaced in the. To calculate entropy changes for a chemical reaction. What matters is the total. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The reverse is true for an endothermic change.
from www.chegg.com
An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). We have seen that the energy given off (or absorbed) by a reaction,. The reverse is true for an endothermic change. To calculate entropy changes for a chemical reaction. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. If this equation is replaced in the. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation.
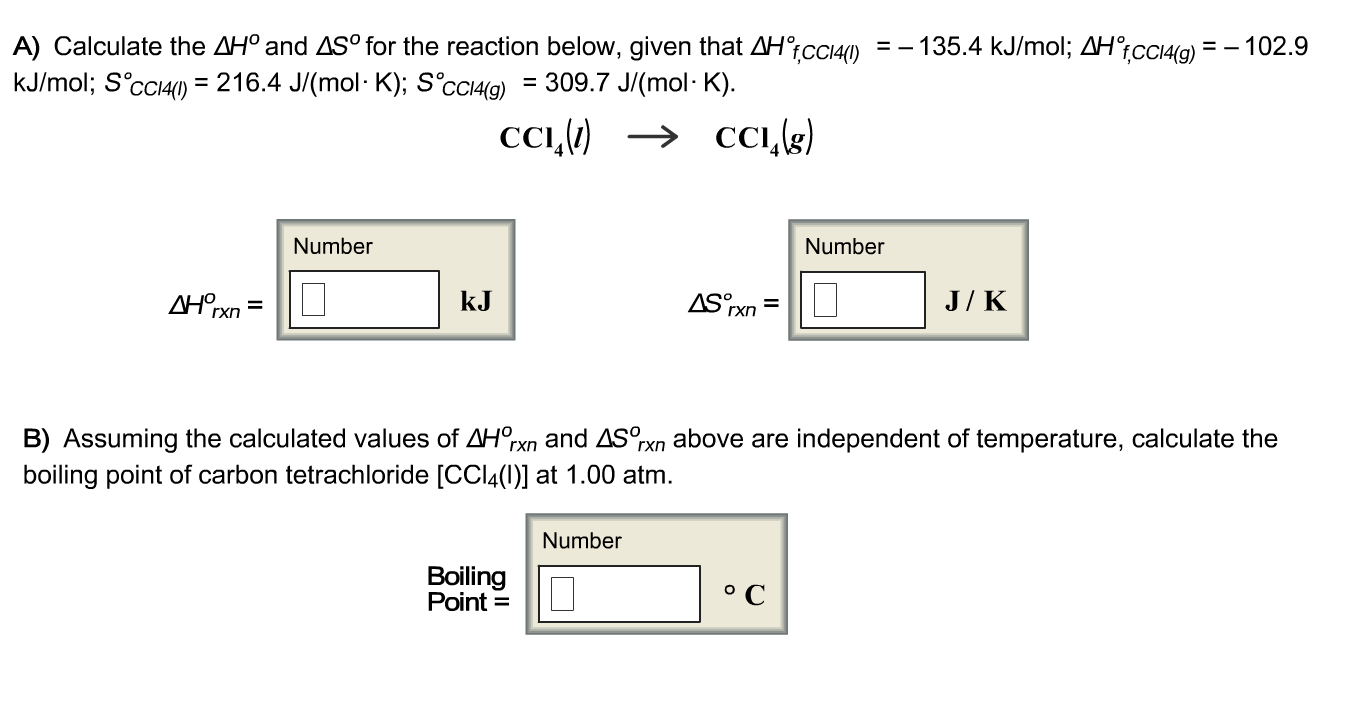
Solved Calculate the delta H degree and delta s^degree for
How To Find Delta S Surroundings From Delta S System The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). Chad continues the chapter on thermodynamics with a lesson on how to calculate. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. If this equation is replaced in the. To calculate entropy changes for a chemical reaction. We have seen that the energy given off (or absorbed) by a reaction,. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. What matters is the total. The reverse is true for an endothermic change.
From www.solutionspile.com
[Solved] According to the second law which of the following How To Find Delta S Surroundings From Delta S System [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. We have seen that the energy given off (or absorbed) by a reaction,. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). To calculate entropy changes for. How To Find Delta S Surroundings From Delta S System.
From www.youtube.com
Thermodynamics Lecture 25 , Chemistry IIT JEE ( Delta S of Universe How To Find Delta S Surroundings From Delta S System \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. If this equation is replaced in the. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation.. How To Find Delta S Surroundings From Delta S System.
From www.chemistrystudent.com
Total Entropy (ALevel) ChemistryStudent How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. The reverse is true for an endothermic change. What matters is the total. If this equation is replaced in the. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved In a balanced threephase YDelta system, the source How To Find Delta S Surroundings From Delta S System The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The reverse is true for an endothermic change.. How To Find Delta S Surroundings From Delta S System.
From educationcajole.z4.web.core.windows.net
How To Calculate Delta S System How To Find Delta S Surroundings From Delta S System [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. We have seen that the energy given off (or absorbed) by a reaction,. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The reverse is true for. How To Find Delta S Surroundings From Delta S System.
From www.youtube.com
Calculate the entropy change in the surroundings YouTube How To Find Delta S Surroundings From Delta S System The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). If this equation is replaced in the. The reverse is true for an endothermic change. What matters is the total. To calculate entropy changes for a chemical reaction. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}.. How To Find Delta S Surroundings From Delta S System.
From www.slideshare.net
Deltas types, formation, case studies How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. What matters is the total. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). We have seen that the energy given off (or absorbed) by a reaction,. The reverse is true for an endothermic change. If this equation is replaced in the. To. How To Find Delta S Surroundings From Delta S System.
From deltas-memories.tumblr.com
delta's system How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. What matters is the total. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding. How To Find Delta S Surroundings From Delta S System.
From www.numerade.com
SOLVED how to determine delta s and delta h from a graph of delta G vs T How To Find Delta S Surroundings From Delta S System The reverse is true for an endothermic change. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. What matters is the total. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. We have seen that the energy given off (or absorbed) by a reaction,. Chad continues the chapter on thermodynamics with a lesson on how to calculate. [latex]\delta s_{univ}. How To Find Delta S Surroundings From Delta S System.
From www.numerade.com
SOLVEDExplain the terms ΔSuniverse , ΔSsystem , and ΔSsurroundings How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. To calculate entropy changes for a chemical reaction. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. We have seen that the energy given. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Calculate Delta S fortheprocess, Delta G forthepr How To Find Delta S Surroundings From Delta S System The reverse is true for an endothermic change. We have seen that the energy given off (or absorbed) by a reaction,. If this equation is replaced in the. To calculate entropy changes for a chemical reaction. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the.. How To Find Delta S Surroundings From Delta S System.
From www.doubtnut.com
Delta S (system) decreases but Delta S (surroundings) increases How To Find Delta S Surroundings From Delta S System [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The formula for the entropy change in the surroundings is \(\delta. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved can some one help with the following How To Find Delta S Surroundings From Delta S System \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. What matters is the total. We have seen that the energy given off (or absorbed) by a reaction,. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. If this equation is replaced in the. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. To. How To Find Delta S Surroundings From Delta S System.
From educationcajole.z4.web.core.windows.net
How To Calculate Delta S Of A Reaction How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. What matters is the total. The reverse is true for an endothermic change. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. An endothermic. How To Find Delta S Surroundings From Delta S System.
From www.slideserve.com
PPT Deltas PowerPoint Presentation, free download ID2358661 How To Find Delta S Surroundings From Delta S System The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. If this equation is replaced in the. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. [latex]\delta s_{univ} = \delta s_{sys} +. How To Find Delta S Surroundings From Delta S System.
From general.chemistrysteps.com
Entropy Changes in the Surroundings Chemistry Steps How To Find Delta S Surroundings From Delta S System If this equation is replaced in the. To calculate entropy changes for a chemical reaction. What matters is the total. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The reverse is true for an endothermic change. We have seen that the energy given off (or absorbed) by a reaction,.. How To Find Delta S Surroundings From Delta S System.
From www.doubtnut.com
Delta S (system) decreases but Delta S (surroundings) increases How To Find Delta S Surroundings From Delta S System What matters is the total. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). We have seen that the energy given off (or absorbed) by a reaction,. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. If this equation is replaced in the. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. [latex]\delta s_{univ} =. How To Find Delta S Surroundings From Delta S System.
From www.youtube.com
How to calculate delta S of vaporization and fusion Gibbs Free Energy How To Find Delta S Surroundings From Delta S System The reverse is true for an endothermic change. If this equation is replaced in the. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. \nonumber. How To Find Delta S Surroundings From Delta S System.
From quizrelativity.z4.web.core.windows.net
How To Determine Delta S Of A Reaction How To Find Delta S Surroundings From Delta S System \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. To calculate entropy changes for a chemical reaction. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. We have seen that the energy given off (or absorbed) by a reaction,. If this equation is replaced in the.. How To Find Delta S Surroundings From Delta S System.
From general.chemistrysteps.com
Entropy Changes in the Surroundings Chemistry Steps How To Find Delta S Surroundings From Delta S System [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. The formula for the entropy. How To Find Delta S Surroundings From Delta S System.
From www.youtube.com
How to Find Delta S of the Universe! (AP Chemistry Tutoring) YouTube How To Find Delta S Surroundings From Delta S System \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. To calculate entropy changes for a chemical reaction. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. What matters is the total.. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved Which of the following situations represent a How To Find Delta S Surroundings From Delta S System To calculate entropy changes for a chemical reaction. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. What matters is the total. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). If this equation is replaced in the. We have seen that the energy. How To Find Delta S Surroundings From Delta S System.
From www.youtube.com
A Level Physical Geography Deltas YouTube How To Find Delta S Surroundings From Delta S System What matters is the total. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The reverse is true for an endothermic change. To calculate entropy changes for a chemical reaction. The formula for the entropy. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved 1calculate delta s of surroundings 2 calculate delta How To Find Delta S Surroundings From Delta S System To calculate entropy changes for a chemical reaction. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. The reverse is true for an endothermic change. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides. How To Find Delta S Surroundings From Delta S System.
From angelinediazs.blogspot.com
How To Solve For Delta S Universe Angeline Diaz's Math Problems How To Find Delta S Surroundings From Delta S System \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. What matters is the total. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The reverse is true for an endothermic change. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases.. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved Calculate the delta H degree and delta s^degree for How To Find Delta S Surroundings From Delta S System We have seen that the energy given off (or absorbed) by a reaction,. What matters is the total. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. To calculate entropy changes for a chemical reaction. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). An endothermic reaction will cool the surroundings, and so the. How To Find Delta S Surroundings From Delta S System.
From www.toppr.com
Calculate ΔS univ (in J/K) for the chemical reaction C(graphite) + 2H2 How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. If this equation is replaced in the. The formula for the entropy change in the surroundings is \(\delta. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved Identify whether Delta S of the system, the How To Find Delta S Surroundings From Delta S System \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. If this equation is replaced in the. Chad continues the chapter on thermodynamics with a lesson on how to calculate. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). What matters is the total. We have seen that the energy given off (or absorbed) by a reaction,. \nonumber. How To Find Delta S Surroundings From Delta S System.
From educationcajole.z4.web.core.windows.net
How To Calculate Delta S System How To Find Delta S Surroundings From Delta S System To calculate entropy changes for a chemical reaction. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation. What matters is the total. Chad continues the. How To Find Delta S Surroundings From Delta S System.
From www.animalia-life.club
Delta Geography How To Find Delta S Surroundings From Delta S System To calculate entropy changes for a chemical reaction. If this equation is replaced in the. The reverse is true for an endothermic change. What matters is the total. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. \[\delta s_{univ} = \delta s_{sys} +. How To Find Delta S Surroundings From Delta S System.
From www.slideshare.net
Chapter 19 Lecture Thermodynamics How To Find Delta S Surroundings From Delta S System Chad continues the chapter on thermodynamics with a lesson on how to calculate. \[\delta s_{univ} = \delta s_{sys} + \delta s_{surr}. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases. We have seen that the energy given off (or absorbed) by a reaction,. What matters is the total. If this equation is replaced in. How To Find Delta S Surroundings From Delta S System.
From www.chegg.com
Solved For reactions carried out under standardstate How To Find Delta S Surroundings From Delta S System We have seen that the energy given off (or absorbed) by a reaction,. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). Chad continues the chapter on thermodynamics with a lesson on how to calculate. [latex]\delta s_{univ} = \delta s_{sys} + \delta s_{surr} = 0 [/latex] these results lead to a profound statement regarding the relation.. How To Find Delta S Surroundings From Delta S System.
From courses.lumenlearning.com
Entropy Boundless Physics How To Find Delta S Surroundings From Delta S System If this equation is replaced in the. To calculate entropy changes for a chemical reaction. Chad continues the chapter on thermodynamics with a lesson on how to calculate. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. We have seen that the energy given off (or absorbed) by a reaction,. What matters is the total. \[\delta s_{univ} =. How To Find Delta S Surroundings From Delta S System.
From www.animalia-life.club
Delta Geography How To Find Delta S Surroundings From Delta S System We have seen that the energy given off (or absorbed) by a reaction,. If this equation is replaced in the. The reverse is true for an endothermic change. What matters is the total. The formula for the entropy change in the surroundings is \(\delta s_{surr}=\delta h_{sys}/t\). To calculate entropy changes for a chemical reaction. [latex]\delta s_{univ} = \delta s_{sys} +. How To Find Delta S Surroundings From Delta S System.
From angelinediazs.blogspot.com
How To Solve For Delta S Universe Angeline Diaz's Math Problems How To Find Delta S Surroundings From Delta S System We have seen that the energy given off (or absorbed) by a reaction,. What matters is the total. To calculate entropy changes for a chemical reaction. \nonumber \] this calculation is important as \(\delta s_{univ}\) provides the. The reverse is true for an endothermic change. An endothermic reaction will cool the surroundings, and so the entropy of the surroundings decreases.. How To Find Delta S Surroundings From Delta S System.