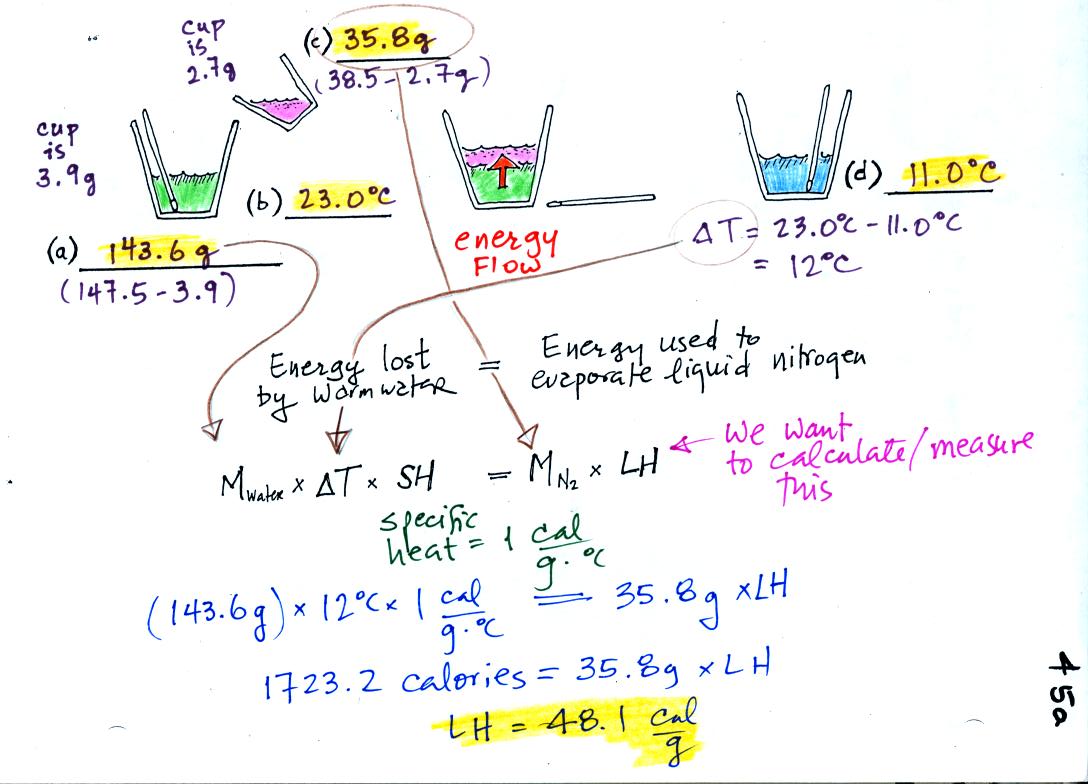
Heat Of Vaporization Nitrogen . Heat imparts energy into the system. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Temperature enthalpy, internal energy and. The input energy required to change the state from liquid to. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the.
from democracyunlimited.web.fc2.com
In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Heat imparts energy into the system. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The input energy required to change the state from liquid to. Temperature enthalpy, internal energy and. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,.
heat of vaporization of liquid nitrogen
Heat Of Vaporization Nitrogen Temperature enthalpy, internal energy and. The input energy required to change the state from liquid to. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Heat imparts energy into the system. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Temperature enthalpy, internal energy and.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Temperature enthalpy, internal energy and. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Heat imparts energy into the system. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen. Heat Of Vaporization Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The input energy required to change the state from liquid to. Heat imparts energy into the system. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Temperature enthalpy, internal energy and.. Heat Of Vaporization Nitrogen.
From www.numerade.com
SOLVED The table displays the vapor pressure of nitrogen at several Heat Of Vaporization Nitrogen The input energy required to change the state from liquid to. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Temperature enthalpy, internal energy and. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to. Heat Of Vaporization Nitrogen.
From www.researchgate.net
(PDF) Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporization Nitrogen The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or. Heat Of Vaporization Nitrogen.
From studylib.net
Latent Heat of Vaporization of Nitrogen Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Heat imparts energy into the system. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The input. Heat Of Vaporization Nitrogen.
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The input energy required to change the state from liquid to. Latent heat of vaporization for fluids like. Heat Of Vaporization Nitrogen.
From golearning5.blogspot.com
The boiling temperature of nitrogen at atmospheric pressure at sea Heat Of Vaporization Nitrogen As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Temperature enthalpy, internal energy and. In thermodynamics, the enthalpy. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved The vapor pressure of nitrogen at several different Heat Of Vaporization Nitrogen The input energy required to change the state from liquid to. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. In this experiment you are to determine the latent heat of. Heat Of Vaporization Nitrogen.
From www.periodic-table.org
Nitrogen Latent Heat of Vaporization Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Heat imparts energy into the system. The input energy required to change the state from liquid to. As a system exchanges thermal energy with its surroundings, the. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved The Boiling Temperature Of Oxygen At Atmospheric P... Heat Of Vaporization Nitrogen In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. The input energy required to change the state from liquid to. Temperature enthalpy, internal energy and. Heat imparts energy into the system. Latent heat of vaporization for fluids like alcohol, ether,. Heat Of Vaporization Nitrogen.
From www.numerade.com
SOLVED 2. Under normal atmospheric pressure, nitrogen boils (and Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Heat imparts energy into the system. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in. Heat Of Vaporization Nitrogen.
From studylib.net
Heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Temperature enthalpy, internal energy and. Heat imparts energy into the system. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. The heat of vaporization (also called the. Heat Of Vaporization Nitrogen.
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporization Nitrogen In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase. Heat Of Vaporization Nitrogen.
From www.numerade.com
The latent heat of vaporization of liquid nitrogen is about 200 . kJ Heat Of Vaporization Nitrogen Temperature enthalpy, internal energy and. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved Part A Use the data to determine the heat of Heat Of Vaporization Nitrogen In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. In. Heat Of Vaporization Nitrogen.
From www.transtutors.com
(Solved) Latent Heat Of Vaporization Of Liquid Nitrogen Introduction Heat Of Vaporization Nitrogen In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. The input energy required to change the state from liquid to. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Temperature enthalpy, internal energy and. In this. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved Liquid nitrogen can be used to cool down objects. The Heat Of Vaporization Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. Temperature enthalpy, internal energy and. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Latent heat of. Heat Of Vaporization Nitrogen.
From www.youtube.com
Virtual Lab Heat of Vaporization of Liquid Nitrogen YouTube Heat Of Vaporization Nitrogen The input energy required to change the state from liquid to. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of. Heat Of Vaporization Nitrogen.
From www.numerade.com
⏩SOLVEDThis table displays the vapor pressure of nitrogen at… Numerade Heat Of Vaporization Nitrogen In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Temperature enthalpy, internal energy and. The input energy required to change the state from liquid to. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. As a system exchanges thermal energy with its surroundings, the temperature of the system. Heat Of Vaporization Nitrogen.
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved Question 1 The boiling temperature of nitrogen at Heat Of Vaporization Nitrogen In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. Heat imparts energy into the system. The heat of vaporization (also called the enthalpy of vaporization) is. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved 2. The boiling temperature of nitrogen at atmospheric Heat Of Vaporization Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Temperature enthalpy, internal energy and. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. In case of liquid to gas phase change, this amount of energy is. Heat Of Vaporization Nitrogen.
From www.youtube.com
Heat of vaporization of liquid nitrogen data collection YouTube Heat Of Vaporization Nitrogen In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Temperature enthalpy, internal energy and. Heat imparts energy into the system. As a system exchanges thermal energy with. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporization Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The input energy required to change the state from liquid to. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Temperature enthalpy, internal energy and. Heat imparts energy into the system. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat. Heat Of Vaporization Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen Heat imparts energy into the system. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The input energy required to change the state from liquid to. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the. Heat Of Vaporization Nitrogen.
From brainly.com
The vapor pressure of nitrogen at several different temperatures is Heat Of Vaporization Nitrogen In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. The input energy required to change the state from liquid to. Heat imparts energy into the system. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine. Heat Of Vaporization Nitrogen.
From www.periodic-table.org
Nitrogen Latent Heat of Vaporization Heat Of Vaporization Nitrogen Temperature enthalpy, internal energy and. Heat imparts energy into the system. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. The input energy required to change the state from liquid to. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Latent heat of vaporization for fluids like alcohol,. Heat Of Vaporization Nitrogen.
From classes.oc.edu
THE LATENT HEAT OF VAPORIZATION OF NITROGEN Heat Of Vaporization Nitrogen In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Heat imparts energy into the system. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as. Heat Of Vaporization Nitrogen.
From www.chegg.com
Solved The boiling temperature of nitrogen at atmospheric Heat Of Vaporization Nitrogen In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization or heat of evaporation, is the. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this. Heat Of Vaporization Nitrogen.
From www.chegg.com
Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporization Nitrogen The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The input energy required to change the state from liquid to. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In thermodynamics, the enthalpy of vaporization (symbol ∆h vap), also known as the (latent) heat of vaporization. Heat Of Vaporization Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from room. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The input energy required to change the state from liquid to. As a system exchanges thermal energy with. Heat Of Vaporization Nitrogen.
From learningfullmaurer.z1.web.core.windows.net
Heat Of Vaporization Biology Heat Of Vaporization Nitrogen Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. The input energy required to change the state from liquid to. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Temperature enthalpy, internal energy and. As a system exchanges thermal energy with its. Heat Of Vaporization Nitrogen.
From studylib.net
HEAT OF VAPORIZATION OF NITROGEN ©By Lee Marek Heat Of Vaporization Nitrogen The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The input energy required to change the state from liquid to. Temperature enthalpy, internal energy and. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and. Heat Of Vaporization Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporization Nitrogen As a system exchanges thermal energy with its surroundings, the temperature of the system will usually increase or decrease,. Latent heat of vaporization for fluids like alcohol, ether, nitrogen, water and more. In this experiment you are to determine the latent heat of vaporization of nitrogen by observing the amount of nitrogen vaporized in cooling a sample of aluminum from. Heat Of Vaporization Nitrogen.