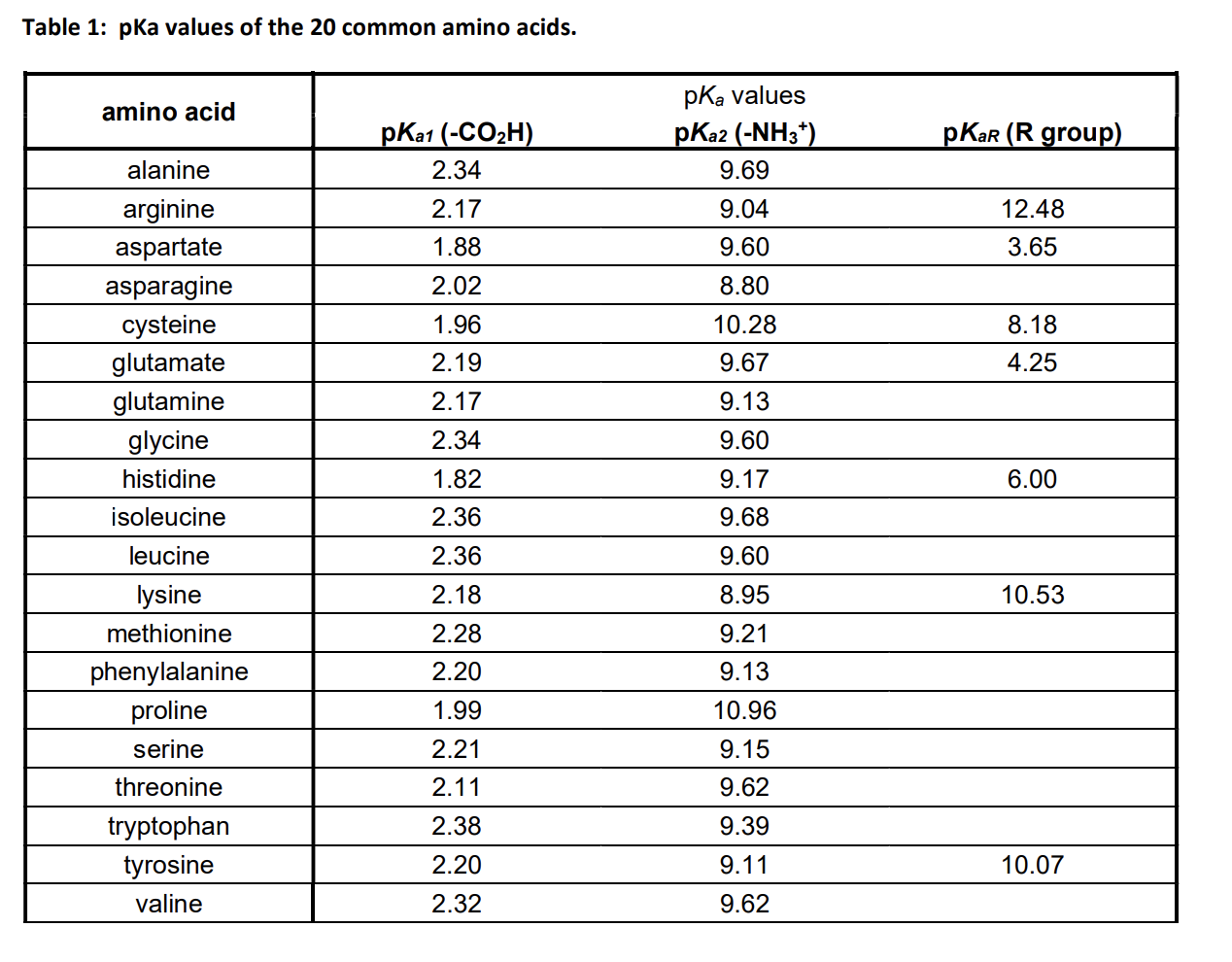
What Is The Ph Of Amino Acids . At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At the “center” of each amino acid is a carbon called the α carbon. At ph values above or below the. All amino acids have the same basic structure, which is shown in figure 2.1. At a ph greater than 10, the amine exists as a neutral. Each amino acid has its own pi value based on the properties of the amino acid. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. At the isoelectric point, the amino. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. A 1.0 −m 1.0 − m solution of a simple. Acidic side chains, such as.
from briannaobie.blogspot.com
Each amino acid has its own pi value based on the properties of the amino acid. At a ph greater than 10, the amine exists as a neutral. Acidic side chains, such as. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At the “center” of each amino acid is a carbon called the α carbon. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. A 1.0 −m 1.0 − m solution of a simple. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. At ph values above or below the.
BriannaObie
What Is The Ph Of Amino Acids All amino acids have the same basic structure, which is shown in figure 2.1. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Acidic side chains, such as. Each amino acid has its own pi value based on the properties of the amino acid. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. A 1.0 −m 1.0 − m solution of a simple. At a ph greater than 10, the amine exists as a neutral. At the “center” of each amino acid is a carbon called the α carbon. All amino acids have the same basic structure, which is shown in figure 2.1. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At the isoelectric point, the amino. At ph values above or below the. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and.
From bceweb.org
Amino Acid Ph Chart A Visual Reference of Charts Chart Master What Is The Ph Of Amino Acids A 1.0 −m 1.0 − m solution of a simple. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Each amino acid has its own pi value based on the properties of the amino acid. Acidic side chains, such as. At the “center” of each amino acid is a carbon called the α carbon.. What Is The Ph Of Amino Acids.
From keywordsuggest.org
Image Gallery hydrochloric acid ph What Is The Ph Of Amino Acids The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At the “center” of each amino acid is a carbon called the α carbon. Acidic side chains, such as. At ph values above or below the. All amino acids have the same basic structure, which is. What Is The Ph Of Amino Acids.
From www.sampletemplates.com
16+ Amino Acid Chart Templates Sample Templates What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. Acidic side chains, such as. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. At ph values above or below the. All amino acids have the same basic structure, which is shown in figure 2.1. At the isoelectric. What Is The Ph Of Amino Acids.
From www.pinterest.co.kr
amino acid chart Google Search A exam images Pinterest 화학, 과학 What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. All amino acids have the same basic structure, which is shown in figure 2.1. A 1.0 −m 1.0 − m solution of a simple. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Amino acids are distinguished by. What Is The Ph Of Amino Acids.
From onlinesciencenotes.com
Amino acids General properties and classification Online Science Notes What Is The Ph Of Amino Acids A 1.0 −m 1.0 − m solution of a simple. At the “center” of each amino acid is a carbon called the α carbon. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Each amino acid has its own pi value based on the properties of the amino acid. At ph values above or. What Is The Ph Of Amino Acids.
From bio.libretexts.org
3.1 Amino Acids Biology LibreTexts What Is The Ph Of Amino Acids Acidic side chains, such as. Each amino acid has its own pi value based on the properties of the amino acid. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. Amino acids are distinguished by their side chains, which can be categorized as acidic or. What Is The Ph Of Amino Acids.
From chemistry.com.pk
A Brief Introduction of Amino Acids The Building Blocks of Proteins What Is The Ph Of Amino Acids Acidic side chains, such as. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At ph values above or below. What Is The Ph Of Amino Acids.
From www.vrogue.co
68 Structure Of Acidic Amino Acids At Various Ph Yout vrogue.co What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. Acidic side chains, such as. At a ph lower than 2, both the carboxylate and amine functions are protonated, so. What Is The Ph Of Amino Acids.
From alevelbiology.co.uk
Structure And Properties Of 20 Standard Amino Acids A Level Notes What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. A 1.0 −m 1.0 − m solution of a simple. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in. What Is The Ph Of Amino Acids.
From www.reddit.com
Help with a pH problem finding charges on amino acids at acidic and What Is The Ph Of Amino Acids The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Each amino acid has its own pi value based on the properties of the amino acid. Amino acids are distinguished by. What Is The Ph Of Amino Acids.
From slidesharenow.blogspot.com
Ph Of Amino Acids Chart slideshare What Is The Ph Of Amino Acids Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At the isoelectric point, the amino. Acidic side chains, such as. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. A 1.0 −m 1.0 − m solution of a. What Is The Ph Of Amino Acids.
From slidesharenow.blogspot.com
Ph Of Amino Acids Chart slideshare What Is The Ph Of Amino Acids The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. All amino acids have the same basic structure, which is shown in figure 2.1. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive. What Is The Ph Of Amino Acids.
From microbiologynotes.org
Amino acids physical, chemical properties and peptide bond What Is The Ph Of Amino Acids Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. A 1.0 −m 1.0 − m solution of a simple. At ph values above or below the. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. All amino acids have. What Is The Ph Of Amino Acids.
From www.masterorganicchemistry.com
Isoelectric Points of Amino Acids (and How To Calculate Them) Master What Is The Ph Of Amino Acids At ph values above or below the. Acidic side chains, such as. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At the isoelectric point, the amino. The isoelectric point. What Is The Ph Of Amino Acids.
From www.animalia-life.club
Printable Amino Acid Chart What Is The Ph Of Amino Acids At the “center” of each amino acid is a carbon called the α carbon. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. Acidic side chains, such as. At ph values above or below the. Amino acids are distinguished by their side chains, which can be. What Is The Ph Of Amino Acids.
From mavink.com
Amino Acid Charge Chart What Is The Ph Of Amino Acids At the isoelectric point, the amino. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. All amino acids have the same basic. What Is The Ph Of Amino Acids.
From www.inf.fu-berlin.de
Amino acids What Is The Ph Of Amino Acids At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. At ph values above or below the. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. Acidic side chains, such as. Amino. What Is The Ph Of Amino Acids.
From www.pinterest.com
Pin on Biochemistry 2018 What Is The Ph Of Amino Acids The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Acidic side chains, such as. At ph values above or below the. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. We saw in section 20.3 and section 24.5 that. What Is The Ph Of Amino Acids.
From briannaobie.blogspot.com
BriannaObie What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. At the “center” of each amino acid is a carbon called the α. What Is The Ph Of Amino Acids.
From www.chegg.com
Using The table, list the amino acids that will carry What Is The Ph Of Amino Acids The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At ph values above or below the. At a ph greater than 10, the amine exists as a neutral. Each amino acid has its own pi value based on the properties of the amino acid. At. What Is The Ph Of Amino Acids.
From www.numerade.com
SOLVED Draw any tetrapeptide composed of naturally occurring amino What Is The Ph Of Amino Acids At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. The isoelectric point (pi). What Is The Ph Of Amino Acids.
From chem.libretexts.org
13.1 Amino Acids Chemistry LibreTexts What Is The Ph Of Amino Acids All amino acids have the same basic structure, which is shown in figure 2.1. Acidic side chains, such as. At the isoelectric point, the amino. At the “center” of each amino acid is a carbon called the α carbon. Each amino acid has its own pi value based on the properties of the amino acid. We saw in section 20.3. What Is The Ph Of Amino Acids.
From www.chegg.com
Solved Given the following four amino acids at pH = 7.4, What Is The Ph Of Amino Acids The zwitterion of an amino acid exists at a ph equal to the isoelectric point. All amino acids have the same basic structure, which is shown in figure 2.1. Each amino acid has its own pi value based on the properties of the amino acid. At a ph greater than 10, the amine exists as a neutral. We saw in. What Is The Ph Of Amino Acids.
From leah4sci.com
Amino Acid Charge in Zwitterions and Isoelectric Point MCAT Tutorial What Is The Ph Of Amino Acids The zwitterion of an amino acid exists at a ph equal to the isoelectric point. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. All amino acids have the same basic structure, which is shown in. What Is The Ph Of Amino Acids.
From freeskill.pk
Structure Of Amino Acids Pdf Chart FreeSkill What Is The Ph Of Amino Acids Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. All amino acids have the same basic structure, which is shown in figure 2.1. At ph values above or below. What Is The Ph Of Amino Acids.
From www.researchgate.net
List of the 20 most common amino acids Download Table What Is The Ph Of Amino Acids At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Each amino acid has its own. What Is The Ph Of Amino Acids.
From www.youtube.com
Amino Acids (Part 3) pH and Pka Biochemistry for MCAT, DAT, NEET What Is The Ph Of Amino Acids At the isoelectric point, the amino. We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. At the “center” of each amino acid is a carbon called the α carbon. At a ph greater than 10, the. What Is The Ph Of Amino Acids.
From mungfali.com
Amino Acid Chain Structure What Is The Ph Of Amino Acids The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Each amino acid has its own pi value based on the properties of the amino acid. Acidic side chains, such. What Is The Ph Of Amino Acids.
From socratic.org
How does pH affect amino acid structure? + Example What Is The Ph Of Amino Acids Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. All amino acids have the same basic structure, which is shown in figure 2.1. At the “center” of each amino acid is a carbon called the α carbon. At a ph greater than 10, the amine exists as a neutral. At a ph lower. What Is The Ph Of Amino Acids.
From chem.libretexts.org
13.1 Amino Acids Chemistry LibreTexts What Is The Ph Of Amino Acids Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At ph values above or below the. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. Each amino acid has its own pi value based on the properties of the amino acid. At a ph greater than 10,. What Is The Ph Of Amino Acids.
From slidesharenow.blogspot.com
Ph Values Of Amino Acids slideshare What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. All amino acids have the same basic structure, which is shown in figure 2.1. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At ph values above or below. What Is The Ph Of Amino Acids.
From biologydictionary.net
[LS16] Sugar to Carbon Molecules Biology Dictionary What Is The Ph Of Amino Acids Acidic side chains, such as. All amino acids have the same basic structure, which is shown in figure 2.1. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. The zwitterion of an amino acid exists at a ph equal to the isoelectric point. A 1.0 −m. What Is The Ph Of Amino Acids.
From amit1b.wordpress.com
Amino Acids Chemistry, Biochemistry & Nutrition Amit Kessel Ph.D What Is The Ph Of Amino Acids Each amino acid has its own pi value based on the properties of the amino acid. At the isoelectric point, the amino. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. At a ph lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive. What Is The Ph Of Amino Acids.
From www.sexizpix.com
Isoelectric Points Of Amino Acids And How To Calculate Them Master What Is The Ph Of Amino Acids The zwitterion of an amino acid exists at a ph equal to the isoelectric point. The isoelectric point (pi) of an amino acid refers to the ph at which the amino acid exists in its neutral, or zwitterionic, form. At a ph greater than 10, the amine exists as a neutral. We saw in section 20.3 and section 24.5 that. What Is The Ph Of Amino Acids.
From www.scienceabc.com
What Is Gastric Acid (Stomach Acid)? Is It Diluted When We Drink Water? What Is The Ph Of Amino Acids We saw in section 20.3 and section 24.5 that a carboxyl group is deprotonated and exists as the carboxylate anion at a physiological ph of 7.3, while an amino group is protonated and. At the isoelectric point, the amino. At a ph greater than 10, the amine exists as a neutral. At ph values above or below the. At the. What Is The Ph Of Amino Acids.