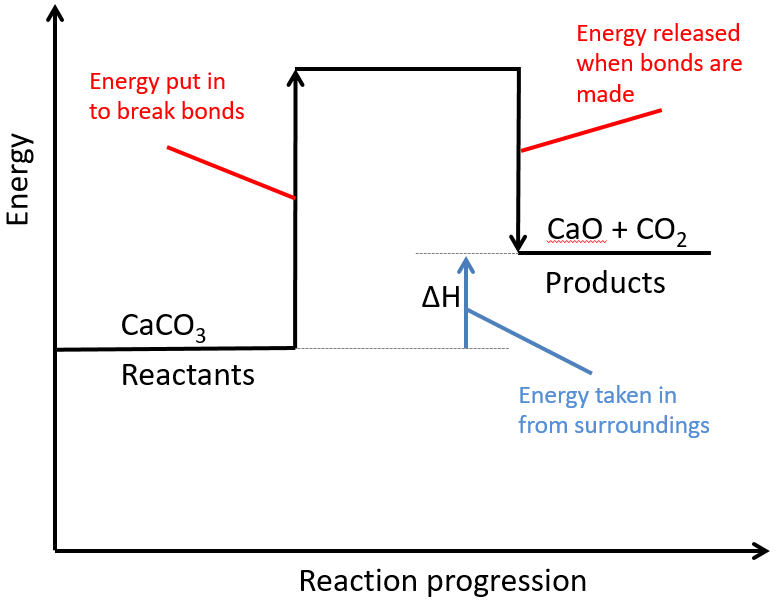
Endothermic Reaction Of Activation Energy . The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Some examples of endothermic reactions are: The reaction between ethanoic acid and sodium carbonate. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. In endothermic reactions, products have higher energy than reactants. Of calcium carbonate in a blast furnace. Because heat is absorbed, endothermic reactions feel cold. For an endothermic reaction, the value of h is positive and the arrow point upwards. The initial increase in energy, from the reactants to the. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. Δh is a positive value for endothermic reactions. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings.
from www.tutormyself.com
An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. The initial increase in energy, from the reactants to the. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. In endothermic reactions, products have higher energy than reactants. Because heat is absorbed, endothermic reactions feel cold. Of calcium carbonate in a blast furnace. Δh is a positive value for endothermic reactions.
314 (Triple only) draw and explain reaction profile diagrams showing
Endothermic Reaction Of Activation Energy Some examples of endothermic reactions are: Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Because heat is absorbed, endothermic reactions feel cold. Some examples of endothermic reactions are: The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. The initial increase in energy, from the reactants to the. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. The reaction between ethanoic acid and sodium carbonate. For an endothermic reaction, the value of h is positive and the arrow point upwards. Of calcium carbonate in a blast furnace. In endothermic reactions, products have higher energy than reactants. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Δh is a positive value for endothermic reactions.
From byjus.com
24. What is the Minimum activation energy required for endothermic and Endothermic Reaction Of Activation Energy The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Some examples of endothermic reactions are: For an endothermic reaction, the value of h is positive and the arrow point upwards. Because heat. Endothermic Reaction Of Activation Energy.
From www.studypool.com
SOLUTION Energy changes in chemical reactions activation energy Endothermic Reaction Of Activation Energy The initial increase in energy, from the reactants to the. Of calcium carbonate in a blast furnace. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. Because heat is absorbed, endothermic reactions feel cold. The. Endothermic Reaction Of Activation Energy.
From www.workmanagementsolutions.com.au
An Analogy The Activation Energy and Catalysis in Business Operations Endothermic Reaction Of Activation Energy The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. Of calcium carbonate in a blast furnace. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Some examples of endothermic reactions are: The transfer of thermal energy during a. Endothermic Reaction Of Activation Energy.
From studylib.net
Lesson 7 Reaction profiles Endothermic Reaction Of Activation Energy The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Of calcium carbonate in a blast furnace. Some examples of endothermic reactions are: When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. Δh is a positive value for endothermic reactions. For an. Endothermic Reaction Of Activation Energy.
From byjus.com
Activation Energy Definition, Formula, SI Units, Examples, Calculation Endothermic Reaction Of Activation Energy Of calcium carbonate in a blast furnace. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. Δh is a positive value for endothermic reactions. The reaction between ethanoic acid and sodium carbonate. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. The activation energy. Endothermic Reaction Of Activation Energy.
From www.slideserve.com
PPT Endothermic Vs. Exothermic Reaction Graphs PowerPoint Endothermic Reaction Of Activation Energy For an endothermic reaction, the value of h is positive and the arrow point upwards. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Δh is a positive value for endothermic reactions. In endothermic reactions, products have higher energy than reactants. The initial increase in energy, from the reactants. Endothermic Reaction Of Activation Energy.
From telegra.ph
Endothermic energy profile diagram activation Telegraph Endothermic Reaction Of Activation Energy Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. Of calcium carbonate in a blast furnace. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. Δh is a positive value for endothermic reactions. The reaction between ethanoic acid and sodium carbonate. The activation energy (\ (e_a\)), labeled \ (\delta {g^. Endothermic Reaction Of Activation Energy.
From www.doubtnut.com
For an endothermic reaction energy of activation is E(a) and enthlpy o Endothermic Reaction Of Activation Energy An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. The initial increase in energy, from the reactants to the. The reaction between ethanoic acid and sodium carbonate. In endothermic reactions, products have higher energy than. Endothermic Reaction Of Activation Energy.
From www.varsitytutors.com
Reaction Coordinate Diagrams College Chemistry Endothermic Reaction Of Activation Energy Some examples of endothermic reactions are: The reaction between ethanoic acid and sodium carbonate. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. For an endothermic reaction, the value of h is positive and the. Endothermic Reaction Of Activation Energy.
From www.dreamstime.com
Activation Energy Endothermic Reaction 2d Illustration Stock Endothermic Reaction Of Activation Energy Of calcium carbonate in a blast furnace. Because heat is absorbed, endothermic reactions feel cold. In endothermic reactions, products have higher energy than reactants. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. Some examples of endothermic reactions are: An endothermic reaction is a chemical reaction that absorbs thermal. Endothermic Reaction Of Activation Energy.
From www.chemistrylearner.com
Endothermic Reaction Definition, Equation, Graph & Examples Endothermic Reaction Of Activation Energy In endothermic reactions, products have higher energy than reactants. The initial increase in energy, from the reactants to the. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. The. Endothermic Reaction Of Activation Energy.
From manualpartsynaxis123.z13.web.core.windows.net
Exothermic And Endothermic Energy Diagrams Endothermic Reaction Of Activation Energy In endothermic reactions, products have higher energy than reactants. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. The reaction between ethanoic acid and sodium carbonate. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. Δh is a positive value for endothermic reactions. The transfer of thermal energy during a. Endothermic Reaction Of Activation Energy.
From stock.adobe.com
Activation energy in endothermic and exothermic reactions. Stock Endothermic Reaction Of Activation Energy Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. In endothermic reactions, products have higher energy than reactants. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Some examples of endothermic reactions are: The reaction between ethanoic acid and sodium carbonate. For an endothermic. Endothermic Reaction Of Activation Energy.
From sciencenotes.org
Endothermic Reactions Definition and Examples Endothermic Reaction Of Activation Energy The initial increase in energy, from the reactants to the. Because heat is absorbed, endothermic reactions feel cold. For an endothermic reaction, the value of h is positive and the arrow point upwards. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. In endothermic reactions, products have higher energy than reactants. The activation energy (\. Endothermic Reaction Of Activation Energy.
From blogs.glowscotland.org.uk
Activation Energy Higher Chemistry Unit 1 Endothermic Reaction Of Activation Energy Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Of calcium carbonate in a blast furnace. Δh is. Endothermic Reaction Of Activation Energy.
From wiringdatabaseinfo.blogspot.com
Energy Diagram For Endothermic Reaction Wiring Site Resource Endothermic Reaction Of Activation Energy When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. For an endothermic reaction, the value of h is positive and the arrow point upwards. The activation energy (\ (e_a\)),. Endothermic Reaction Of Activation Energy.
From schematron.org
Endothermic Reaction Coordinate Diagram Wiring Diagram Pictures Endothermic Reaction Of Activation Energy Some examples of endothermic reactions are: For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Because heat is absorbed, endothermic reactions feel cold. The reaction between ethanoic acid and sodium carbonate. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. For an endothermic reaction,. Endothermic Reaction Of Activation Energy.
From www.tutormyself.com
314 (Triple only) draw and explain reaction profile diagrams showing Endothermic Reaction Of Activation Energy The initial increase in energy, from the reactants to the. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. For an endothermic reaction, the value of h is positive and the arrow. Endothermic Reaction Of Activation Energy.
From socratic.org
What are activation energies? Socratic Endothermic Reaction Of Activation Energy For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Because heat is absorbed, endothermic reactions feel cold. For an endothermic reaction, the value of h is positive and the arrow point upwards. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference. Endothermic Reaction Of Activation Energy.
From www.revisechemistry.uk
Energetics OCR Gateway C3 revisechemistry.uk Endothermic Reaction Of Activation Energy When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. Of calcium carbonate in a blast furnace. Δh is a positive value for endothermic reactions. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. The reaction between ethanoic acid. Endothermic Reaction Of Activation Energy.
From www.istockphoto.com
Vector Graphs Of Endothermic And Exothermic Reactions Activation Energy Endothermic Reaction Of Activation Energy The reaction between ethanoic acid and sodium carbonate. Some examples of endothermic reactions are: In endothermic reactions, products have higher energy than reactants. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. For an endothermic chemical reaction to. Endothermic Reaction Of Activation Energy.
From www.slideserve.com
PPT Endothermic Vs. Exothermic Reaction Graphs PowerPoint Endothermic Reaction Of Activation Energy In endothermic reactions, products have higher energy than reactants. Δh is a positive value for endothermic reactions. The reaction between ethanoic acid and sodium carbonate. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. For an endothermic reaction,. Endothermic Reaction Of Activation Energy.
From www.doubtnut.com
An endothermic reaction with high activation energy for the forward re Endothermic Reaction Of Activation Energy Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. The initial increase in energy, from the reactants to the. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the. Endothermic Reaction Of Activation Energy.
From www.expii.com
Energy Diagram — Overview & Parts Expii Endothermic Reaction Of Activation Energy The initial increase in energy, from the reactants to the. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. The reaction between ethanoic acid and sodium carbonate. Because heat. Endothermic Reaction Of Activation Energy.
From ar.inspiredpencil.com
Exothermic Reaction Graph Endothermic Reaction Of Activation Energy For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Of calcium carbonate in a blast furnace. The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. For an endothermic reaction, the value of h is positive and the arrow. Endothermic Reaction Of Activation Energy.
From byjus.com
Activation Energy Definition, Formula, SI Units, Examples, Calculation Endothermic Reaction Of Activation Energy When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. The reaction between ethanoic acid and sodium carbonate. Δh is a positive value for endothermic reactions. For an endothermic reaction, the value of h is positive and the arrow point upwards. In endothermic reactions, products have higher energy than reactants.. Endothermic Reaction Of Activation Energy.
From elecdiags.com
The Significance and Use of Endothermic Reaction Profile Diagrams in Endothermic Reaction Of Activation Energy In endothermic reactions, products have higher energy than reactants. For an endothermic reaction, the value of h is positive and the arrow point upwards. The reaction between ethanoic acid and sodium carbonate. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Exothermic reactions transfer energy to the surroundings and the temperature of. Endothermic Reaction Of Activation Energy.
From www.studyorgo.com
How to Interpret Thermodynamics of Reactions Endothermic Reaction Of Activation Energy Of calcium carbonate in a blast furnace. The reaction between ethanoic acid and sodium carbonate. Because heat is absorbed, endothermic reactions feel cold. The initial increase in energy, from the reactants to the. For an endothermic reaction, the value of h is positive and the arrow point upwards. Some examples of endothermic reactions are: Δh is a positive value for. Endothermic Reaction Of Activation Energy.
From www.youtube.com
6.1 Activation energy (SL) YouTube Endothermic Reaction Of Activation Energy The initial increase in energy, from the reactants to the. For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. For an endothermic reaction, the value of h is positive and the arrow point upwards. Δh is a positive value for endothermic reactions. In endothermic reactions, products have higher energy. Endothermic Reaction Of Activation Energy.
From www.youtube.com
Endothermic and Exothermic Reactions With Potential Energy Diagrams Endothermic Reaction Of Activation Energy For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. For an endothermic reaction, the value of h is positive and the arrow point upwards. The transfer of thermal energy. Endothermic Reaction Of Activation Energy.
From www.doubtnut.com
An endothermic reaction with high activation energy for the forward re Endothermic Reaction Of Activation Energy For an endothermic chemical reaction to proceed, the reactants must absorb energy from their environment to be converted to products. Δh is a positive value for endothermic reactions. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. The reaction between ethanoic acid and sodium carbonate. An endothermic reaction is a chemical reaction that absorbs thermal. Endothermic Reaction Of Activation Energy.
From professional.patrickneyman.com
Activation Energy Equation Endothermic Reaction Of Activation Energy The reaction between ethanoic acid and sodium carbonate. Of calcium carbonate in a blast furnace. Some examples of endothermic reactions are: An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. Δh is a positive value for endothermic reactions. Because heat is absorbed, endothermic reactions feel cold. For an endothermic reaction, the value of h is. Endothermic Reaction Of Activation Energy.
From stock.adobe.com
activation energy endothermic reaction diagram vector de Stock Adobe Endothermic Reaction Of Activation Energy The reaction between ethanoic acid and sodium carbonate. Of calcium carbonate in a blast furnace. In endothermic reactions, products have higher energy than reactants. The transfer of thermal energy during a reaction is called the enthalpy change, δh, of the reaction. Because heat is absorbed, endothermic reactions feel cold. For an endothermic reaction, the value of h is positive and. Endothermic Reaction Of Activation Energy.
From www.slideserve.com
PPT Chapter 7 PowerPoint Presentation ID239015 Endothermic Reaction Of Activation Energy Of calcium carbonate in a blast furnace. When we draw a reaction profile diagram for an endothermic reaction, we find that the products are at a higher. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. Some examples of endothermic reactions are: The transfer of thermal energy during a reaction is called the enthalpy change,. Endothermic Reaction Of Activation Energy.
From revisechemistry.uk
Exothermic and Endothermic Reactions AQA C5 revisechemistry.uk Endothermic Reaction Of Activation Energy The activation energy (\ (e_a\)), labeled \ (\delta {g^ {\ddagger}}\) in figure 2, is the energy difference between the reactants. An endothermic reaction is a chemical reaction that absorbs thermal energy from its surroundings. Exothermic reactions transfer energy to the surroundings and the temperature of the surroundings increases. In endothermic reactions, products have higher energy than reactants. The initial increase. Endothermic Reaction Of Activation Energy.