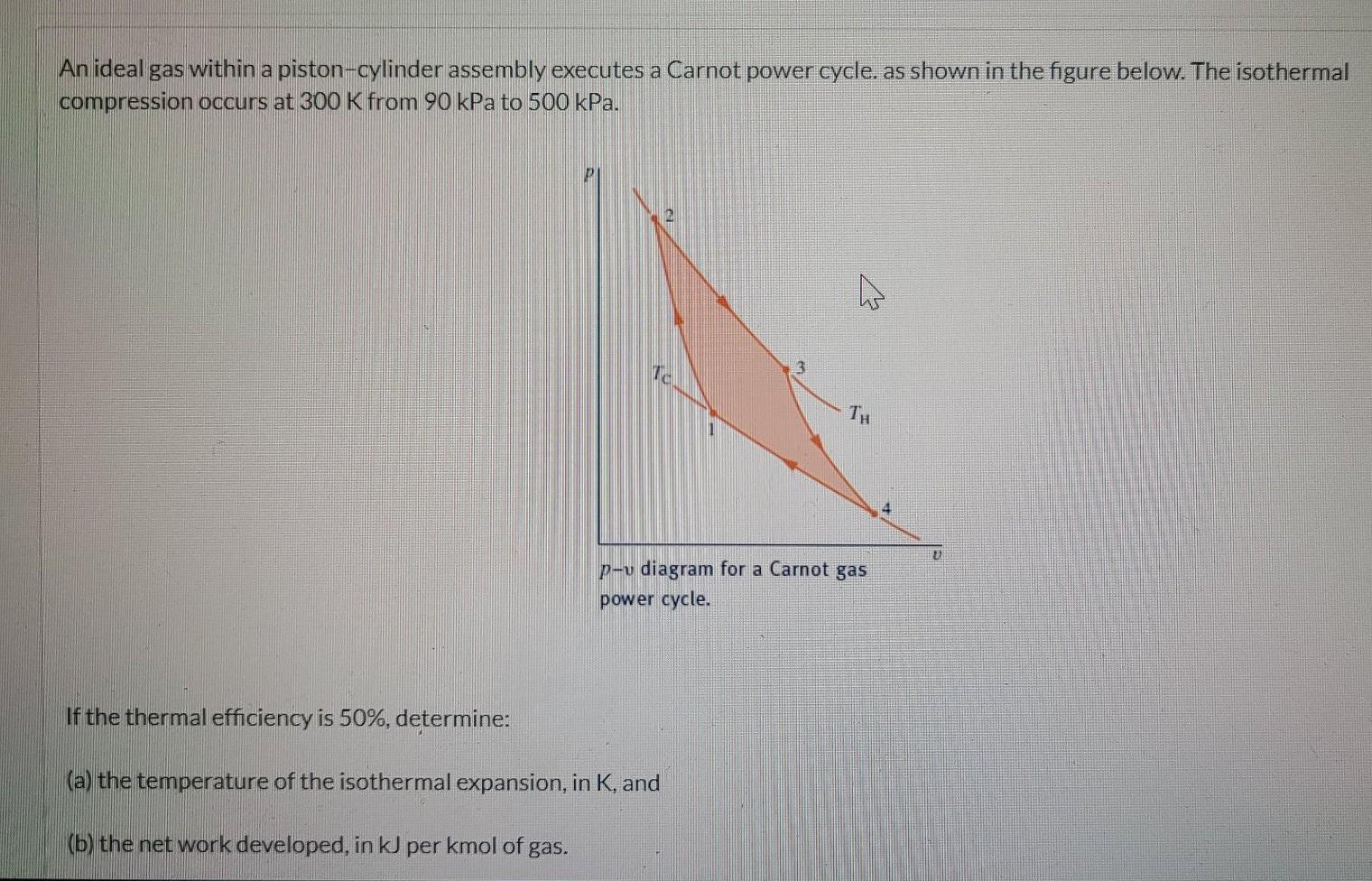
Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle . The thermal efficiency is divided into two parts: The rn is divided by 750 and it is 60 percent. For the cycle, th=400k and tc=300k. The energy rejected by heat. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, t h = 500 k and t c = 300. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The isothermal compression occurs at 325 k from 2.
from www.chegg.com
For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, t h = 500 k and t c = 300. The energy rejected by heat. For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The rn is divided by 750 and it is 60 percent. The isothermal compression occurs at 325 k from 2. The thermal efficiency is divided into two parts:
Solved An ideal gas within a pistoncylinder assembly
Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. The thermal efficiency is divided into two parts: Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The energy rejected by heat. The rn is divided by 750 and it is 60 percent. For the cycle, th=400k and tc=300k. The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, t h = 500 k and t c = 300.
From www.chegg.com
Solved Name 2) (25 pts) Air within a pistoncylinder Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The thermal efficiency is divided into two parts: Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The rn is divided by 750 and it is 60 percent. The energy rejected by heat. The isothermal compression occurs at 325 k from 2. For the cycle,. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVED Two kilograms of air within a pistoncylinder assembly executes Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, t h = 500 k and t c = 300. The thermal efficiency is divided into two parts: The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The energy rejected by heat. The. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.coursehero.com
[Solved] A gas within a pistoncylinder assembly executes a Carnot Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. The rn is divided by 750 and it is 60 percent. The energy rejected by heat. For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The isothermal compression occurs at 325 k from. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From brainly.com
0.1 kg of air as an ideal gas executes a Carnot power cycle in a piston Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. The thermal efficiency is divided into two parts: For the cycle, th=400k and tc=300k. The rn is divided by 750 and it is 60 percent. The isothermal compression occurs at 325 k from 2. For the cycle, t h = 500 k and t c = 300. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Two kg of air within a pistoncylinder assembly Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: The energy rejected by heat. For the cycle, t. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.coursehero.com
[Solved] Air in a pistoncylinder assembly undergoes a Carnot power Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. For the cycle, th=400k and tc=300k. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The thermal efficiency is divided into two parts: Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The isothermal compression occurs at 325 k from 2. The rn is divided by 750 and it. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
2. Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The thermal efficiency is divided into two parts: For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From schematicpartjillets.z14.web.core.windows.net
Heat Engine Cycle Diagram Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The rn is divided by 750 and it is 60 percent. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The thermal efficiency is divided into two parts: For the cycle, th=400k and tc=300k. For the cycle, t h = 500 k and t c = 300. The energy rejected by heat. Imagine a carnot heat pump operates between an outside temperature. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.eigenplus.com
Carnot Cycle Thermodynamics of Carnot Engine Theorem, Explanation Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The thermal efficiency is divided into two parts: For the cycle, t h = 500 k and t c = 300. For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The rn is divided by 750 and it is 60 percent. The isothermal compression occurs at. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
3. 1 kg of air within a pistoncylinder assembly Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The rn is divided by 750 and it is 60 percent. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The energy rejected by heat. The isothermal compression occurs at 325 k from 2. For the cycle, th=400k and tc=300k. The thermal efficiency is divided into two parts: For the cycle, t. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Exercise 2/3 (40 pts) Air within a pistoncylinder Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The thermal efficiency is divided into two parts: The rn is divided by 750 and it is 60 percent. For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. The energy rejected by heat. Imagine. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air in a piston/cylinder goes through a Carnot cycle Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. For the cycle, th=400k and tc=300k. For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The thermal efficiency is divided into two parts: The rn is divided. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air in a piston/cylinder goes through a Carnot cycle Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. The thermal efficiency is divided into two parts: For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, th=400k and tc=300k. For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of.. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.coursehero.com
[Solved] Air in a pistoncylinder assembly undergoes a Carnot power Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, th=400k and tc=300k. The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: The energy rejected by heat. For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Two kg of air within a pistoncylinder assembly Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. The rn is divided by 750 and it is 60 percent. For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The thermal efficiency is. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Two kilograms of air within a pistoncylinder Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. The rn is divided by 750 and it is 60 percent. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The thermal efficiency is divided into two parts: The energy rejected by heat. For the cycle,. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVEDAir within a pistoncylinder assembly executes a Carnot heat Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. The thermal efficiency is divided into two parts: For the cycle, th=400k and tc=300k. For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From manualdiagramausterlitz.z19.web.core.windows.net
Carnot Cycle Schematic Diagram Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The thermal efficiency is divided into two parts: The rn is divided by 750 and it is 60 percent. For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVED Air within a pistoncylinder assembly executes a Carnot heat Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. The isothermal compression occurs at 325 k from 2. For the cycle, t h = 500 k and t c = 300. The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVED Using Energy Concepts and the Ideal Gas Model Air contained in Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The rn is divided by 750 and it is 60 percent. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle,. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The thermal efficiency is divided into two parts: The energy rejected by heat. For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, th=400k and tc=300k. The isothermal compression occurs at 325 k from 2. For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVEDAn ideal gas Carnot cycle with air in a piston cylinder has a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, t h = 500 k and t c = 300. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The energy rejected by heat. The thermal efficiency is divided into two parts: The isothermal compression occurs at 325 k from 2. For the cycle, th=400k and tc=300k. For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved 5.83 Two kilograms of air within a pistoncylinder Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. The thermal efficiency is divided into two parts: For the cycle, t h = 500 k and t c = 300. For the cycle, th=400k and tc=300k. The energy rejected by heat. The rn is divided by 750 and it is 60 percent. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved An ideal gas within a pistoncylinder assembly Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The thermal efficiency is divided into two parts: For the cycle, t h = 500 k and t c = 300. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, th=400k and tc=300k. The rn is divided by 750 and it is. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly execute an Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the cycle, th=400k and tc=300k. The energy rejected by heat. The thermal efficiency is divided into two parts: Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The rn is divided by 750 and it. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Problem 5.053 SI Air within a pistoncylinder Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The rn is divided by 750 and it is 60 percent. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The isothermal compression occurs at 325 k from 2. For the cycle, t h = 500 k and t c = 300. The thermal efficiency is divided into two parts: For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The energy rejected by heat. The rn is divided by 750 and it is 60 percent. For the cycle, t h = 500 k and t c = 300. The thermal efficiency is divided into two parts:. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.bartleby.com
Answered Air within a pistoncylinder assembly… bartleby Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The energy rejected by heat. The thermal efficiency is divided into two parts: The rn is divided by 750 and it is 60 percent. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The isothermal compression occurs at 325 k from 2. For the cycle,. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.coursehero.com
[Solved] Air within a pistoncylinder assembly executes a Carnot heat Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. For the cycle, t h = 500 k and t c = 300. The isothermal compression occurs at 325 k from 2. The rn is divided by 750 and it is 60 percent. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The energy rejected by heat. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: For the cycle, th=400k and tc=300k. The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air is contained in a pistoncylinder assembly, which Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The energy rejected by heat. The isothermal compression occurs at 325 k from 2. The rn is divided by 750 and it is 60 percent. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. The thermal efficiency is divided. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.numerade.com
SOLVED Two kilograms of air within a pistoncylinder assembly execute Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, th=400k and tc=300k. The isothermal compression occurs at 325 k from 2. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: The energy. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The rn is divided by 750 and it is 60 percent. The thermal efficiency is divided into two parts: The isothermal compression occurs at 325 k from 2. For the cycle, t h = 500 k and t c = 300. For the cycle, th=400k and tc=300k. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved An ideal gas within a pistoncylinder assembly Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle The isothermal compression occurs at 325 k from 2. Imagine a carnot heat pump operates between an outside temperature of [latex]0\phantom{\rule{0.2em}{0ex}}\text{°c}[/latex] and an inside temperature of. For the cycle, th=400k and tc=300k. The thermal efficiency is divided into two parts: The rn is divided by 750 and it is 60 percent. For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. For the. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.
From www.chegg.com
Solved Air within a pistoncylinder assembly executes a Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle For the cycle, th=400k and tc=300k. The energy rejected by heat. The thermal efficiency is divided into two parts: For the cycle, $t_{\mathrm{h}}=$ $600 \mathrm{~k}$ and. The isothermal compression occurs at 325 k from 2. For the cycle, t h = 500 k and t c = 300. The rn is divided by 750 and it is 60 percent. Imagine. Air Within A Piston-Cylinder Assembly Executes A Carnot Heat Pump Cycle.