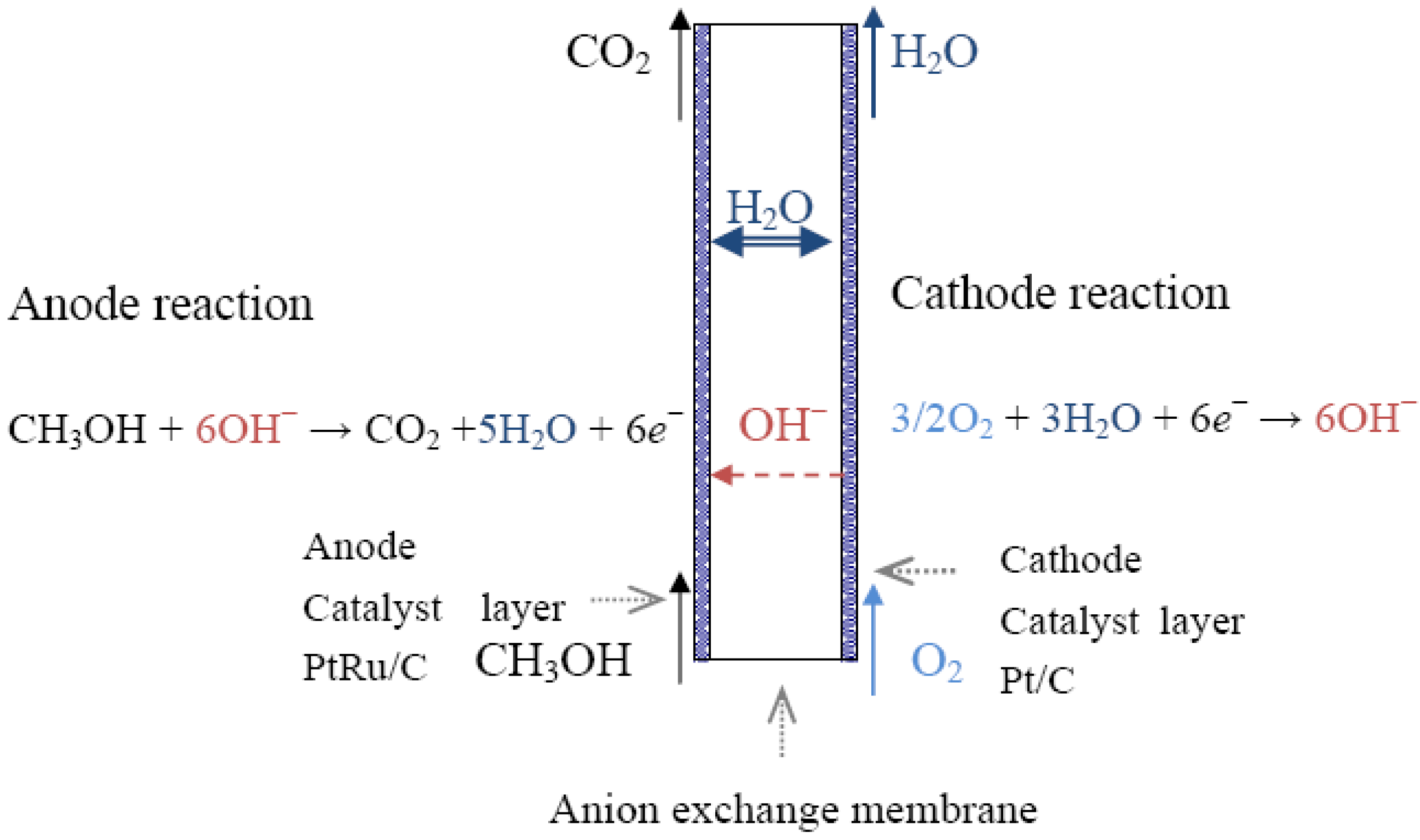
Reactions Of Fuel Cell . By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A fuel cell resembles a battery in many respects, but it. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. 2 h2 + o2 2 h2o. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel, such as hydrogen, is fed to the anode, and.
from www.mdpi.com
2 h2 + o2 2 h2o. A fuel, such as hydrogen, is fed to the anode, and. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell resembles a battery in many respects, but it.
Energies Free FullText Principles and Materials Aspects of Direct
Reactions Of Fuel Cell The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. 2 h2 + o2 2 h2o. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A fuel cell resembles a battery in many respects, but it. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A fuel, such as hydrogen, is fed to the anode, and. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell.
From www.researchgate.net
Schematic representation of a molten carbonate fuel cell (MCFC Reactions Of Fuel Cell The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A fuel cell resembles a battery. Reactions Of Fuel Cell.
From www.slideserve.com
PPT Fuel cells PowerPoint Presentation, free download ID1590103 Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. The. Reactions Of Fuel Cell.
From www.electroniclinic.com
Hydrogen Fuel Cell, Application of Fuel Cells, construction, and Working Reactions Of Fuel Cell By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel, such as hydrogen, is fed to the anode, and. A fuel cell resembles a battery in many respects, but it. Also hydrocarbons such as natural gas. Reactions Of Fuel Cell.
From www.slideserve.com
PPT Fuel Cell Catalysts Based on Metal Nanoparticles PowerPoint Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell resembles a battery in many respects, but it. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A fuel, such as hydrogen, is fed to the anode, and.. Reactions Of Fuel Cell.
From large.stanford.edu
Hydrogen Fuel Cells Reactions Of Fuel Cell By contrast to a conventional cell, where only limited quantities of oxidizing agent and. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A fuel cell can be defined as an electrochemical cell that generates. Reactions Of Fuel Cell.
From www.researchgate.net
6. Hydrogenoxygen fuel cell scheme. Download Scientific Diagram Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. 2 h2 +. Reactions Of Fuel Cell.
From metabolicprocesses-ethanol.weebly.com
Ethanol Fuel Ethanol Reactions Of Fuel Cell 2 h2 + o2 2 h2o. A fuel, such as hydrogen, is fed to the anode, and. A fuel cell resembles a battery in many respects, but it. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. If we burn hydrogen. Reactions Of Fuel Cell.
From www.researchgate.net
Scheme of a solid oxide fuel cell. Download Scientific Diagram Reactions Of Fuel Cell Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A fuel cell resembles a battery. Reactions Of Fuel Cell.
From www.researchgate.net
(a) Ethanolbased fuel cell reactions in different electrolytes Reactions Of Fuel Cell Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. By contrast to a conventional cell, where only. Reactions Of Fuel Cell.
From www.researchgate.net
Schematic representation of energy related applications, i.e., oxygen Reactions Of Fuel Cell A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. 2 h2 + o2 2 h2o. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by. Reactions Of Fuel Cell.
From www.mdpi.com
Energies Free FullText Principles and Materials Aspects of Direct Reactions Of Fuel Cell A fuel cell resembles a battery in many respects, but it. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. 2 h2 +. Reactions Of Fuel Cell.
From www.researchgate.net
Schematic representation of Direct Methanol Fuel Cell (DMFC). This fuel Reactions Of Fuel Cell A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A fuel, such as hydrogen, is fed to the anode, and. 2 h2 + o2 2 h2o. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A fuel cell resembles a battery in many respects,. Reactions Of Fuel Cell.
From www.thesciencehive.co.uk
Fuel Cells (AQA) — the science sauce Reactions Of Fuel Cell A fuel, such as hydrogen, is fed to the anode, and. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. 2 h2 + o2 2 h2o. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. By contrast to a conventional cell, where only limited quantities. Reactions Of Fuel Cell.
From www.youtube.com
Proton Exchange Membrane Fuel Cell, Introduction, Principle, Advantages Reactions Of Fuel Cell 2 h2 + o2 2 h2o. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. A fuel, such as hydrogen, is fed to the anode, and. A type of. Reactions Of Fuel Cell.
From www.researchgate.net
Chemical reaction in proton exchange membrane fuel cell (PEMFC Reactions Of Fuel Cell Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. 2 h2 + o2 2 h2o. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. This review discusses the history, fundamentals,. Reactions Of Fuel Cell.
From www.researchgate.net
Electrochemical reactions in a fuel cell with (A) H 2 O 2 and (B) CH 4 Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. This review discusses the history, fundamentals, and applications. Reactions Of Fuel Cell.
From klavsfwka.blob.core.windows.net
What Is The Purpose Of Creating A Reaction In A Fuel Cell at Brian Lacy Reactions Of Fuel Cell Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. This review discusses the. Reactions Of Fuel Cell.
From www.researchgate.net
Oxidation reactions in fuel cells (cathodic and anodic half reactions Reactions Of Fuel Cell A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A fuel cell resembles. Reactions Of Fuel Cell.
From www.britannica.com
Fuel cell Proton Exchange, Alkaline, Polymer Britannica Reactions Of Fuel Cell 2 h2 + o2 2 h2o. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including. Reactions Of Fuel Cell.
From www.vedantu.com
Draw a neat labelled diagram of {H_2} {O_2} fuel cell. Write the Reactions Of Fuel Cell 2 h2 + o2 2 h2o. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. If we burn hydrogen in an internal combustion engine, like the ones. Reactions Of Fuel Cell.
From opentextbc.ca
Applications of Redox Reactions Voltaic Cells Introductory Chemistry Reactions Of Fuel Cell A fuel cell resembles a battery in many respects, but it. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. This review discusses the history, fundamentals, and applications of different fuel cell technologies,. Reactions Of Fuel Cell.
From www.vectorstock.com
Molten carbonate fuel cells process Royalty Free Vector Reactions Of Fuel Cell A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. 2 h2 + o2 2 h2o. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. A fuel cell can be defined as an electrochemical cell that generates electrical energy from. Reactions Of Fuel Cell.
From www.youtube.com
Direct Methanol Fuel Cell, Introduction, Principle, Advantages Reactions Of Fuel Cell By contrast to a conventional cell, where only limited quantities of oxidizing agent and. 2 h2 + o2 2 h2o. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A fuel cell consists of two. Reactions Of Fuel Cell.
From www.nagwa.com
Question Video Identifying Which Equation Shows the Reaction at a Reactions Of Fuel Cell If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around. Reactions Of Fuel Cell.
From www.researchgate.net
(a) Ethanolbased fuel cell reactions in different electrolytes Reactions Of Fuel Cell A fuel cell resembles a battery in many respects, but it. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel, such as hydrogen, is fed to the anode, and. A fuel cell can be defined. Reactions Of Fuel Cell.
From www.researchgate.net
Layer structure and electrochemical reactions of PEM fuel cell Reactions Of Fuel Cell By contrast to a conventional cell, where only limited quantities of oxidizing agent and. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. The most simple and favored reactants are hydrogen as fuel. Reactions Of Fuel Cell.
From revisechemistry.uk
Organic chemistry (chemistry only) OCR Gateway C6 revisechemistry.uk Reactions Of Fuel Cell This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A fuel, such as hydrogen, is fed to the anode, and. If we burn hydrogen in an internal combustion engine, like. Reactions Of Fuel Cell.
From 640orfree.com
How Does A Hydrogen Fuel Cell Work? A Comprehensive Guide Linquip (2022) Reactions Of Fuel Cell Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. Fuel cell, any of a class of devices that convert the chemical energy of a fuel directly into electricity by electrochemical reactions. 2 h2 + o2 2 h2o. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell.. Reactions Of Fuel Cell.
From www.researchgate.net
15 Fuel cell types and reactions. Download Scientific Diagram Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. Also hydrocarbons such as natural gas and alcohols. Reactions Of Fuel Cell.
From www.vedantu.com
What are ‘fuel cells’? Write cathode and anode reaction in a fuel cell. Reactions Of Fuel Cell A fuel cell resembles a battery in many respects, but it. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A type of galvanic cell which promises to become increasingly important in the future is. Reactions Of Fuel Cell.
From www.britannica.com
Fuel cell Definition, Types, Applications, & Facts Britannica Reactions Of Fuel Cell A fuel, such as hydrogen, is fed to the anode, and. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A fuel cell can be defined as an electrochemical cell that generates electrical energy from fuel via an electrochemical reaction. A type of galvanic cell which promises to become increasingly important in the future is. Reactions Of Fuel Cell.
From www.betase.nl
Electrochemistry of the fuel cell Betase BV Reactions Of Fuel Cell The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. A fuel cell resembles a battery in many respects, but it. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. Also hydrocarbons such as natural gas and alcohols like methanol are used as fuels. If we burn. Reactions Of Fuel Cell.
From www.youtube.com
OCR C6 Fuel Cells (Higher) YouTube Reactions Of Fuel Cell The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. By contrast to a conventional cell, where only limited quantities of oxidizing agent and. A fuel cell resembles a battery in many respects, but it. 2 h2 + o2 2 h2o. This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton. Reactions Of Fuel Cell.
From www.chalmers.se
Fuel Cell Systems Reactions Of Fuel Cell A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. If we burn hydrogen in an internal combustion engine, like the ones in normal cars, the reaction is rapid and. A fuel cell resembles a battery in many respects, but it. The most simple and favored reactants are hydrogen as fuel and. Reactions Of Fuel Cell.
From www.researchgate.net
1 Fuel Cell Reaction through and external circuit, thus creating Reactions Of Fuel Cell This review discusses the history, fundamentals, and applications of different fuel cell technologies, including proton exchange membrane fuel cells. The most simple and favored reactants are hydrogen as fuel and oxygen as oxidant. 2 h2 + o2 2 h2o. A type of galvanic cell which promises to become increasingly important in the future is the fuel cell. A fuel, such. Reactions Of Fuel Cell.