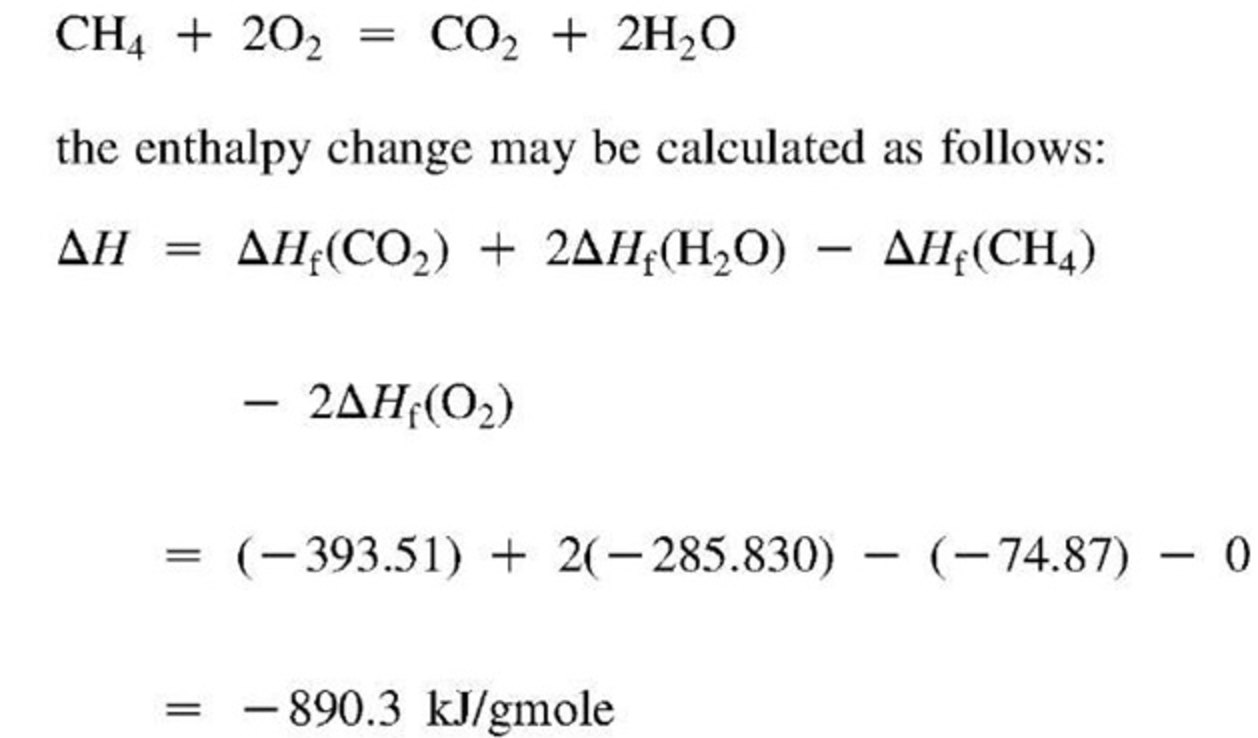Standard Heat Of Formation In Chemistry . standard enthalpy of formation. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. If a solution is being. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent.
from materiallistgaskell.z21.web.core.windows.net
standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. standard enthalpy of formation. If a solution is being.
Heat Of Formation Equations
Standard Heat Of Formation In Chemistry the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. standard enthalpy of formation. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. If a solution is being. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created.
From kunduz.com
[ANSWERED] Using standard heats of formation, calcu... Standard Heat Of Formation In Chemistry If a solution is being. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Chemistry 17.4 PowerPoint Presentation, free download ID2772524 Standard Heat Of Formation In Chemistry A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. If a solution is being. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard enthalpy of formation, also known as. Standard Heat Of Formation In Chemistry.
From worksheetmediasnouts.z13.web.core.windows.net
How To Determine The Heat Of Formation Standard Heat Of Formation In Chemistry One atmosphere and 25 °c (or 298 k). standard enthalpy of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance. Standard Heat Of Formation In Chemistry.
From learningschoolandy.z21.web.core.windows.net
Heat Of Formation List Standard Heat Of Formation In Chemistry A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. standard enthalpy of formation. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. . Standard Heat Of Formation In Chemistry.
From dxorvtvqp.blob.core.windows.net
Standard Enthalpy Of Formation Gibbsite at Samuel Speed blog Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change. Standard Heat Of Formation In Chemistry.
From printablelistquinta.z21.web.core.windows.net
Explain Heat Of Formation Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Standard Heats of Reaction PowerPoint Presentation, free download Standard Heat Of Formation In Chemistry One atmosphere and 25 °c (or 298 k). standard enthalpy of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from. Standard Heat Of Formation In Chemistry.
From lessonluft.z19.web.core.windows.net
Heat Of Formation Chart Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. If a solution is being. standard enthalpy of formation. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. Standard Heat Of Formation In Chemistry.
From slideplayer.com
Chemistry ppt download Standard Heat Of Formation In Chemistry standard enthalpy of formation. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. . Standard Heat Of Formation In Chemistry.
From rayb78.github.io
Heat Of Formation Chart Standard Heat Of Formation In Chemistry standard enthalpy of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard heat of formation. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT THERMOCHEMISTRY PowerPoint Presentation, free download ID4499046 Standard Heat Of Formation In Chemistry If a solution is being. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. A standard enthalpy of formation δh. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Chemistry 122 PowerPoint Presentation, free download ID3181424 Standard Heat Of Formation In Chemistry A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. the standard enthalpy of formation. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Thermochemistry Chemical Energy PowerPoint Presentation, free Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation, also known as the heat of formation, is. Standard Heat Of Formation In Chemistry.
From www.scribd.com
Standard Heats of Formation WS SOLUTIONS PDF Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. One atmosphere and 25 °c (or 298 k). A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. standard enthalpy of formation (or heat of formation),. Standard Heat Of Formation In Chemistry.
From www.coursehero.com
[Solved] 1. All of the following compounds have a standard heat of Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. One atmosphere and 25 °c (or 298 k). standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Thermochemistry PowerPoint Presentation, free download ID314871 Standard Heat Of Formation In Chemistry One atmosphere and 25 °c (or 298 k). A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. If a solution is being. the standard enthalpy. Standard Heat Of Formation In Chemistry.
From www.meritnation.com
Standard heat of formation of HgO(s) at 298 K and at constant pressure Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. If a solution. Standard Heat Of Formation In Chemistry.
From askfilo.com
CHEMISTRY Example Calculate the standard heat of formation of carbon di.. Standard Heat Of Formation In Chemistry the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. A standard enthalpy of. Standard Heat Of Formation In Chemistry.
From www.chem.fsu.edu
CHM1045 Enthalpy Lecture Standard Heat Of Formation In Chemistry standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation, also known as the heat. Standard Heat Of Formation In Chemistry.
From printabledeancraigxqn4.z22.web.core.windows.net
Heat Of Formation Worksheets Answers Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation. If a solution is being. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. . Standard Heat Of Formation In Chemistry.
From www.showme.com
11.4Calculating Heat ChangesStandard Heats of Formation (II) Science Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. the standard enthalpy of formation,. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Chemistry 17.4 PowerPoint Presentation, free download ID2772524 Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard heat of formation. Standard Heat Of Formation In Chemistry.
From studylib.net
heats of formation worksheet key Standard Heat Of Formation In Chemistry If a solution is being. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. standard enthalpy of formation. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. the standard enthalpy of formation, also known. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Chemistry 17.4 PowerPoint Presentation, free download ID2772524 Standard Heat Of Formation In Chemistry One atmosphere and 25 °c (or 298 k). standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. If a solution is being. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of. Standard Heat Of Formation In Chemistry.
From stahonorschemistry.weebly.com
III Calculating Enthalpies STA Form IV Honors Chemistry Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. the standard. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Thermochemistry Chemical Energy PowerPoint Presentation, free Standard Heat Of Formation In Chemistry If a solution is being. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Thermochemistry Chemical Energy PowerPoint Presentation, free Standard Heat Of Formation In Chemistry A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1. Standard Heat Of Formation In Chemistry.
From www.showme.com
Standard heat of formation Science, Chemistry, thermochemistry ShowMe Standard Heat Of Formation In Chemistry standard enthalpy of formation. One atmosphere and 25 °c (or 298 k). the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. If. Standard Heat Of Formation In Chemistry.
From www.youtube.com
Chemistry 122 Heats of Reaction Standard Heats of Formation (Part 2 Standard Heat Of Formation In Chemistry A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1. Standard Heat Of Formation In Chemistry.
From exosbjzsg.blob.core.windows.net
Standard Enthalpy Of Formation Al2O3 at Lois Woodburn blog Standard Heat Of Formation In Chemistry standard enthalpy of formation. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and. Standard Heat Of Formation In Chemistry.
From slideplayer.com
Chemistry ppt download Standard Heat Of Formation In Chemistry standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. One atmosphere and 25 °c (or 298 k). the standard heat of formation. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Chapter 11 PowerPoint Presentation, free download ID1084959 Standard Heat Of Formation In Chemistry standard enthalpy of formation (or heat of formation), δhof , is the enthalpy change when 1 mol of the substance is formed from its constituent. standard enthalpy of formation. the standard heat of formation (δhof) (δ h f o) is the enthalpy change associated with the formation of one mole. the standard enthalpy of formation, also. Standard Heat Of Formation In Chemistry.
From slideplayer.com
How are they related? Chemistry and Energy. ppt download Standard Heat Of Formation In Chemistry If a solution is being. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. One atmosphere and 25 °c (or 298 k). standard enthalpy of. Standard Heat Of Formation In Chemistry.
From www.slideserve.com
PPT Thermochemistry PowerPoint Presentation, free download ID2694049 Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. the standard. Standard Heat Of Formation In Chemistry.
From materiallistgaskell.z21.web.core.windows.net
Heat Of Formation Equations Standard Heat Of Formation In Chemistry the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. If a solution. Standard Heat Of Formation In Chemistry.