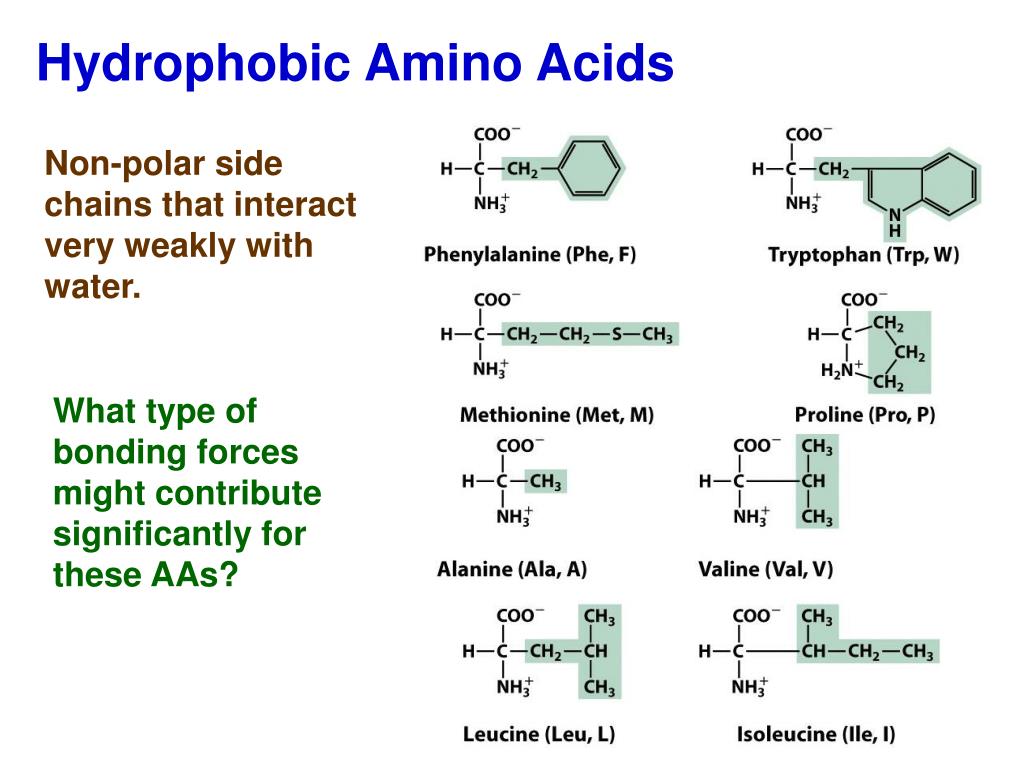
Branched Chain Amino Acids Are Insoluble In Water . We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. This is important, because it is. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and.
from www.slideserve.com
among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. This is important, because it is. We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4.
PPT Proteins Amino Acid Chains PowerPoint Presentation, free
Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. We review here the fundamentals of bcaa. This is important, because it is. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids.
From www.researchgate.net
8 Branchedchain amino acid catabolism pathways of B. linens (producing Branched Chain Amino Acids Are Insoluble In Water ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Chemical structures of branchedchain amino acids Download Scientific Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. This is important, because it is. We review here the fundamentals of bcaa. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction. Branched Chain Amino Acids Are Insoluble In Water.
From www.biohealth-int.com
Protein powder with aminoacids (BCAAs) Biohealth International Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Structures of branchedchain amino acids with hydrophobic aliphatic Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. We review here the fundamentals of bcaa. This is. Branched Chain Amino Acids Are Insoluble In Water.
From www.nursinghero.com
Amino Acids Structure Nutrition Study Guides Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. We review here the fundamentals of bcaa. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction between an aspartic acid carboxylate ion. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Branchedchain amino acids and branchedchain keto acids Download Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. This is important, because it is. ionic bonds. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Overview of the pathways of branchedchain amino acid (BCAA Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion. Branched Chain Amino Acids Are Insoluble In Water.
From dxoywizjo.blob.core.windows.net
BranchedChain Amino Acids List at Teresa Godinez blog Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. though. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Branched chain amino acids metabolism Download Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Pathways of branched chain fatty acid (BCFA) synthesis in B. subtilis Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Biosynthesis of branchedchain amino acids. Enzymes and corresponding Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. This is important, because it. Branched Chain Amino Acids Are Insoluble In Water.
From www.foodnetworksolution.com
Branchedchain amino acid Food Wiki Food Network Solution Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. though all of. Branched Chain Amino Acids Are Insoluble In Water.
From www.slideserve.com
PPT Pathways of Amino Acid Degredation PowerPoint Presentation, free Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. This is important, because it is. We review here the fundamentals of bcaa. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Illustration showing catabolism of branchedchain amino acids Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. We review here the fundamentals of bcaa. among them, the branched chain amino. Branched Chain Amino Acids Are Insoluble In Water.
From www.theperfect105.com
Branched chain amino acids The Perfect 105 Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. This is important, because it is. We review here the fundamentals of bcaa. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of. Branched Chain Amino Acids Are Insoluble In Water.
From chemistnotes.com
Classification of Amino acids Chemistry Notes Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. We review here the fundamentals of bcaa. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a). Branched Chain Amino Acids Are Insoluble In Water.
From europepmc.org
BranchedChain Amino Acid Metabolism in Arabidopsis thaliana Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. We review here the fundamentals. Branched Chain Amino Acids Are Insoluble In Water.
From blog.nasm.org
What Do BranchedChain Amino Acids (BCAAs) Do? Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. We review here the fundamentals of bcaa. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion. Branched Chain Amino Acids Are Insoluble In Water.
From www.slideshare.net
Branch chain Amino Acid Metabolism Branched Chain Amino Acids Are Insoluble In Water ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. We review here the fundamentals of bcaa. This is important, because it is. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction between an. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
The branchedchain fatty acid biosynthesis pathway. (Upper) Leucine is Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For. Branched Chain Amino Acids Are Insoluble In Water.
From myhealthgoat.com
BCAA's 5 Tremendous Benefits of Branched Chain Amino Acids Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. This is important, because it is. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile),. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Pathway for branchedchain amino acid synthesis. AHAS, acetohydroxy Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. We review here the fundamentals of bcaa. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. This is. Branched Chain Amino Acids Are Insoluble In Water.
From education.med.nyu.edu
AMINO ACID METABOLISM BRANCHEDCHAIN AMINO ACID DEGRADATION Branched Chain Amino Acids Are Insoluble In Water ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For. Branched Chain Amino Acids Are Insoluble In Water.
From www.fullste.am
Branch Chain Amino Acids Fullsteam Branched Chain Amino Acids Are Insoluble In Water among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. though all of the amino acids are, in. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Biosynthesis of branchedchain amino acids in plants. Download Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Overview of the branched chain amino acid (BCAA) biosynthesis pathway Branched Chain Amino Acids Are Insoluble In Water ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For. Branched Chain Amino Acids Are Insoluble In Water.
From www.researchgate.net
Structures of branchedchain amino acids with hydrophobic aliphatic Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. We review here the fundamentals of bcaa. though all of the amino acids are, in fact, soluble. Branched Chain Amino Acids Are Insoluble In Water.
From biologysimple.com
Branched Chain Amino Acids Biology Simple Branched Chain Amino Acids Are Insoluble In Water though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. We review here the fundamentals. Branched Chain Amino Acids Are Insoluble In Water.
From blog.nasm.org
What Do BranchedChain Amino Acids (BCAAs) Do? Branched Chain Amino Acids Are Insoluble In Water though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. We review here the fundamentals of bcaa. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure. Branched Chain Amino Acids Are Insoluble In Water.
From europepmc.org
BranchedChain Amino Acid Metabolism in Arabidopsis thaliana Branched Chain Amino Acids Are Insoluble In Water though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. among them, the branched. Branched Chain Amino Acids Are Insoluble In Water.
From www.youtube.com
Branched Chain Amino Acid Metabolism BCAA Catabolism Pathway and Branched Chain Amino Acids Are Insoluble In Water For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. We review here the fundamentals. Branched Chain Amino Acids Are Insoluble In Water.
From themedicalbiochemistrypage.org
Amino Acid Biosynthesis and Catabolism The Medical Biochemistry Page Branched Chain Amino Acids Are Insoluble In Water This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure 22.4.5 22.4. We review here the fundamentals. Branched Chain Amino Acids Are Insoluble In Water.
From www.slideserve.com
PPT How to Consume BranchedChain Amino Acids PowerPoint Presentation Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. among them, the branched chain amino acids (bcaas) refer to leucine (leu), isoleucine (ile), and valine (val), and. For example, the mutual attraction between an aspartic acid carboxylate ion. Branched Chain Amino Acids Are Insoluble In Water.
From www.slideserve.com
PPT Proteins Amino Acid Chains PowerPoint Presentation, free Branched Chain Amino Acids Are Insoluble In Water though all of the amino acids are, in fact, soluble in water, the interactions of their side chains with water differ significantly. We review here the fundamentals of bcaa. This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. among them, the branched chain amino. Branched Chain Amino Acids Are Insoluble In Water.
From healthjade.com
BCAA amino acids, BCAA function, benefits and BCAA supplement uses Branched Chain Amino Acids Are Insoluble In Water We review here the fundamentals of bcaa. This is important, because it is. ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of figure. Branched Chain Amino Acids Are Insoluble In Water.