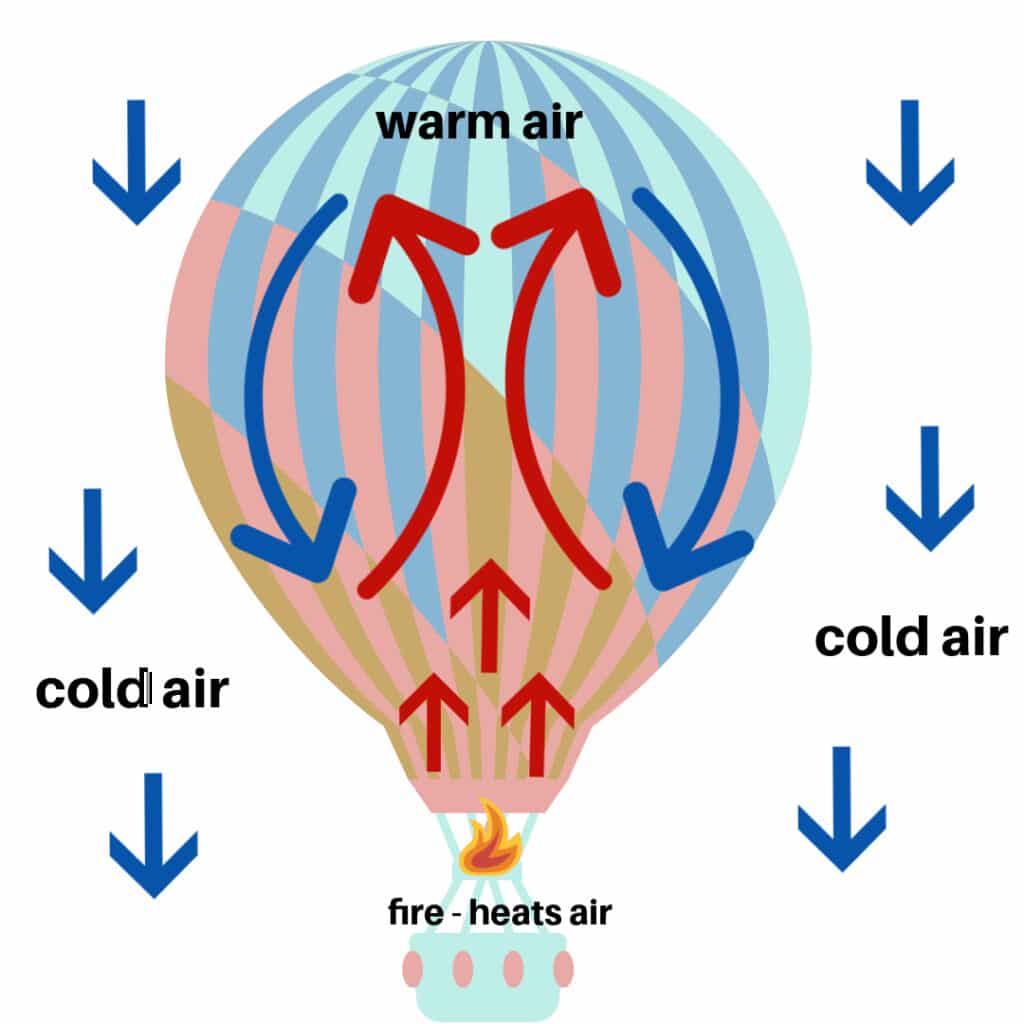
What Gas Laws Apply To Hot Air Balloons . State and explain the ideal gas law using boltzmann's constant; Hot air balloons fly based on charles’s law. The operator first ignites a propane burner to fill the balloon with air. The ideal gas law can be written in terms of the number of molecules of gas: The relationship between temperature and volume: The gas becomes more dense and the balloon sinks. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the kelvin scale (zero kelvin corresponds to. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. As the air continues to. The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. The law is expressed as v∝t, where v is volume and t is temperature. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. This decreases its density, so the balloon rises in the air.
from www.science-sparks.com
The ideal gas law can be written in terms of the number of molecules of gas: The gas becomes more dense and the balloon sinks. Heating the air in the balloon increases the balloon’s volume. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the kelvin scale (zero kelvin corresponds to. As the air continues to. This decreases its density, so the balloon rises in the air. The operator first ignites a propane burner to fill the balloon with air. The law is expressed as v∝t, where v is volume and t is temperature. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. Hot air balloons fly based on charles’s law.
How does a hot air balloon work?
What Gas Laws Apply To Hot Air Balloons According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. Heating the air in the balloon increases the balloon’s volume. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. As the air continues to. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the kelvin scale (zero kelvin corresponds to. The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. State and explain the ideal gas law using boltzmann's constant; This decreases its density, so the balloon rises in the air. Hot air balloons fly based on charles’s law. The gas becomes more dense and the balloon sinks. The ideal gas law can be written in terms of the number of molecules of gas: The law states that when pressure is constant, the volume of a gas varies directly with the temperature. The relationship between temperature and volume: The operator first ignites a propane burner to fill the balloon with air. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little.
From slideplayer.com
The Gas Laws. ppt download What Gas Laws Apply To Hot Air Balloons The law is expressed as v∝t, where v is volume and t is temperature. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. This decreases its density, so the balloon rises in the air. Hot air balloons fly based on charles’s law. As the air continues to. The ideal gas law. What Gas Laws Apply To Hot Air Balloons.
From slideplayer.com
The Gas Laws. ppt download What Gas Laws Apply To Hot Air Balloons As the air continues to. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. This decreases its density, so the balloon rises in the. What Gas Laws Apply To Hot Air Balloons.
From www.slideserve.com
PPT The Ideal Gas Law PowerPoint Presentation, free download ID4354594 What Gas Laws Apply To Hot Air Balloons State and explain the ideal gas law using boltzmann's constant; The ideal gas law can be written in terms of the number of molecules of gas: The gas becomes more dense and the balloon sinks. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. The law states that when pressure is. What Gas Laws Apply To Hot Air Balloons.
From www.thecoldwire.com
What Type Of Gas Is Used In Hot Air Balloons? (Everything To Know) What Gas Laws Apply To Hot Air Balloons The gas becomes more dense and the balloon sinks. The ideal gas law can be written in terms of the number of molecules of gas: The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. This decreases its density, so the balloon rises in the air. Heating the air in the balloon. What Gas Laws Apply To Hot Air Balloons.
From byjus.com
A hot air balloon loaded with sandbags has a total mass of M. It is What Gas Laws Apply To Hot Air Balloons Hot air balloons fly based on charles’s law. As the air continues to. Heating the air in the balloon increases the balloon’s volume. The law is expressed as v∝t, where v is volume and t is temperature. The gas becomes more dense and the balloon sinks. According to charles's law, heating a gas will increase its volume, and since gas. What Gas Laws Apply To Hot Air Balloons.
From www.slideserve.com
PPT Heat Transfer PowerPoint Presentation, free download ID1606939 What Gas Laws Apply To Hot Air Balloons Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. The ideal gas law can be written in terms of the number of molecules of gas: This decreases its density, so the balloon rises in the air. Hot air balloons fly based on charles’s law. The law is expressed. What Gas Laws Apply To Hot Air Balloons.
From flatearth.ws
Basic Science Page 4 FlatEarth.ws What Gas Laws Apply To Hot Air Balloons State and explain the ideal gas law using boltzmann's constant; The law states that when pressure is constant, the volume of a gas varies directly with the temperature. This decreases its density, so the balloon rises in the air. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. Use the ideal. What Gas Laws Apply To Hot Air Balloons.
From www.pinterest.com
You may have wondered, how do hot air balloons work? We answer this and What Gas Laws Apply To Hot Air Balloons As the air continues to. State and explain the ideal gas law using boltzmann's constant; According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. The relationship between temperature and volume: The ideal gas law. What Gas Laws Apply To Hot Air Balloons.
From hotairflight.com
What Are The Parts of a Hot Air Balloon? A Diagram and Guide Hot Air What Gas Laws Apply To Hot Air Balloons Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. Heating the air in the balloon increases the balloon’s volume. The ideal gas law relates. What Gas Laws Apply To Hot Air Balloons.
From courses.lumenlearning.com
Relating Pressure, Volume, Amount, and Temperature The Ideal Gas Law What Gas Laws Apply To Hot Air Balloons This decreases its density, so the balloon rises in the air. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. Heating the air in the balloon increases the balloon’s volume. The law states that. What Gas Laws Apply To Hot Air Balloons.
From www.scienceabc.com
How Do You Steer A Hot Air Balloon? » Science ABC What Gas Laws Apply To Hot Air Balloons \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. The ideal gas law relates the pressure and volume of a gas to the number. What Gas Laws Apply To Hot Air Balloons.
From outdoortroop.com
How Do Hot Air Balloons Work? (A helpful guide with pictures) Outdoor What Gas Laws Apply To Hot Air Balloons This decreases its density, so the balloon rises in the air. State and explain the ideal gas law using boltzmann's constant; The relationship between temperature and volume: Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. As the air continues to. The law is expressed as v∝t, where. What Gas Laws Apply To Hot Air Balloons.
From www.youtube.com
PS1 7 Gas Laws CHARLES'S LAW YouTube What Gas Laws Apply To Hot Air Balloons The ideal gas law can be written in terms of the number of molecules of gas: Hot air balloons fly based on charles’s law. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules. What Gas Laws Apply To Hot Air Balloons.
From stock.adobe.com
Charles Law Infographic Diagram Example helium balloon when volume What Gas Laws Apply To Hot Air Balloons The operator first ignites a propane burner to fill the balloon with air. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. State and explain the ideal gas law using boltzmann's constant; The relationship. What Gas Laws Apply To Hot Air Balloons.
From outdoortroop.com
How Do Hot Air Balloons Work? (A helpful guide with pictures) Outdoor What Gas Laws Apply To Hot Air Balloons The gas becomes more dense and the balloon sinks. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. The law is expressed as v∝t, where v is volume and t is temperature. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied. What Gas Laws Apply To Hot Air Balloons.
From www.slideserve.com
PPT Chapter 12 PowerPoint Presentation, free download ID5745686 What Gas Laws Apply To Hot Air Balloons The relationship between temperature and volume: As the air continues to. The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the number of molecules in a given. State and explain the. What Gas Laws Apply To Hot Air Balloons.
From www.tec-science.com
How does a hot air balloon work Buoyancy in gases tecscience What Gas Laws Apply To Hot Air Balloons The relationship between temperature and volume: \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. Hot air balloons fly based on charles’s law. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The ideal gas law relates. What Gas Laws Apply To Hot Air Balloons.
From www.youtube.com
Charles Law & Hot Air Balloon Gaseous Law Volume Temperature What Gas Laws Apply To Hot Air Balloons According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The relationship between temperature and volume: The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. The physical principle known as charles' law states that the volume of a. What Gas Laws Apply To Hot Air Balloons.
From www.science-sparks.com
How does a hot air balloon work? What Gas Laws Apply To Hot Air Balloons Heating the air in the balloon increases the balloon’s volume. The law is expressed as v∝t, where v is volume and t is temperature. The relationship between temperature and volume: \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. As the air continues to. According. What Gas Laws Apply To Hot Air Balloons.
From sciencenotes.org
Combined Gas Law Definition, Formula, Examples What Gas Laws Apply To Hot Air Balloons As the air continues to. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. Heating the air in the balloon increases the balloon’s volume. This decreases its. What Gas Laws Apply To Hot Air Balloons.
From www.tdk.com
Sensors Technology used at air sports like in hotair balloon What Gas Laws Apply To Hot Air Balloons The law states that when pressure is constant, the volume of a gas varies directly with the temperature. As the air continues to. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the. What Gas Laws Apply To Hot Air Balloons.
From www.slideserve.com
PPT GASES 13.2 PowerPoint Presentation, free download ID6530844 What Gas Laws Apply To Hot Air Balloons Hot air balloons fly based on charles’s law. The relationship between temperature and volume: The ideal gas law can be written in terms of the number of molecules of gas: State and explain the ideal gas law using boltzmann's constant; The operator first ignites a propane burner to fill the balloon with air. The ideal gas law relates the pressure. What Gas Laws Apply To Hot Air Balloons.
From rainbowryders.com
How A Hot Air Balloon Works Rainbow Ryders What Gas Laws Apply To Hot Air Balloons Hot air balloons fly based on charles’s law. The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. State and explain the ideal gas law using boltzmann's constant; The operator first ignites a propane burner to fill the balloon with air. Heating the air in the balloon increases the balloon’s volume. \[pv. What Gas Laws Apply To Hot Air Balloons.
From issuu.com
Hot Air Balloons by Scouting Ireland Issuu What Gas Laws Apply To Hot Air Balloons The law is expressed as v∝t, where v is volume and t is temperature. The ideal gas law can be written in terms of the number of molecules of gas: The law states that when pressure is constant, the volume of a gas varies directly with the temperature. The ideal gas law relates the pressure and volume of a gas. What Gas Laws Apply To Hot Air Balloons.
From seattleballooning.com
How Hot Air Balloons Work? (It's Amazing) Seattle Ballooning What Gas Laws Apply To Hot Air Balloons According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The law is expressed as v∝t, where v is volume and t is temperature. Hot air balloons fly based on charles’s law. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its. What Gas Laws Apply To Hot Air Balloons.
From sciencenotes.org
Ideal Gas Law Formula and Examples What Gas Laws Apply To Hot Air Balloons The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. This decreases its density, so the balloon rises in the air. \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. As the air continues to. According to. What Gas Laws Apply To Hot Air Balloons.
From www.atmo.arizona.edu
Lecture 6 Ideal gas law, rising and sinking air What Gas Laws Apply To Hot Air Balloons The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. The law states that when pressure is constant, the volume of a gas varies directly with the temperature. State and explain the ideal gas law using boltzmann's constant; The physical principle known as charles' law states. What Gas Laws Apply To Hot Air Balloons.
From www.slideshare.net
3 behavior of gases What Gas Laws Apply To Hot Air Balloons \[pv = nkt,\] where \(p\) is pressure, \(v\) is volume, \(t\) is temperature, \(n\) is number of molecules, and \(k\) is the boltzmann constant. According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. Heating the air in the balloon increases the balloon’s volume. Hot air balloons fly based on charles’s law. The. What Gas Laws Apply To Hot Air Balloons.
From www.thecoldwire.com
What Type Of Gas Is Used In Hot Air Balloons? (Everything To Know) What Gas Laws Apply To Hot Air Balloons The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. Hot air balloons fly based on charles’s law. State and explain the ideal gas law using boltzmann's constant; As the air continues to. The relationship between temperature and volume: This decreases its density, so the balloon rises in the air. Heating the. What Gas Laws Apply To Hot Air Balloons.
From www.slideshare.net
Ppa6 Lecture Ch 10 What Gas Laws Apply To Hot Air Balloons The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. Heating the air in the balloon increases the balloon’s volume. The relationship between temperature and volume: This decreases its density, so the balloon rises in the air. Use the ideal gas law to calculate pressure change, temperature change, volume change, or the. What Gas Laws Apply To Hot Air Balloons.
From www.dreamstime.com
Molecules in a Hot Air Balloon Stock Illustration Illustration of What Gas Laws Apply To Hot Air Balloons The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. The operator first ignites a propane burner to fill the balloon with air. As the air continues to. State and explain the ideal gas law using boltzmann's constant; Hot air balloons fly based on charles’s law. The physical principle known as charles'. What Gas Laws Apply To Hot Air Balloons.
From quizlet.com
Hot Air Balloon Terms Physics Diagram Quizlet What Gas Laws Apply To Hot Air Balloons According to charles's law, heating a gas will increase its volume, and since gas molecules exert little. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the kelvin scale (zero kelvin corresponds to. State and explain the ideal gas law using boltzmann's constant; Heating. What Gas Laws Apply To Hot Air Balloons.
From www.youtube.com
Ideal Gas Equation Explained with Balloons! PV = nRT YouTube What Gas Laws Apply To Hot Air Balloons The operation of a hot air balloon depends on charles' law, the ideal gas law, and archimedes' principle. The ideal gas law can be written in terms of the number of molecules of gas: The gas becomes more dense and the balloon sinks. The physical principle known as charles' law states that the volume of a gas equals a constant. What Gas Laws Apply To Hot Air Balloons.
From hotairflyer.com
How Does The Molecular Theory Related To Hot Air Balloons? What Gas Laws Apply To Hot Air Balloons The gas becomes more dense and the balloon sinks. The physical principle known as charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the kelvin scale (zero kelvin corresponds to. Hot air balloons fly based on charles’s law. The law states that when pressure is constant, the volume of. What Gas Laws Apply To Hot Air Balloons.
From www.storyboardthat.com
hot cold air balloon charles law Storyboard by 05918d98 What Gas Laws Apply To Hot Air Balloons The gas becomes more dense and the balloon sinks. The ideal gas law relates the pressure and volume of a gas to the number of gas molecules and the temperature of the gas. Hot air balloons fly based on charles’s law. State and explain the ideal gas law using boltzmann's constant; This decreases its density, so the balloon rises in. What Gas Laws Apply To Hot Air Balloons.