Heat Capacity Ratio Of N2 . Specific heat of nitrogen is 1.04 j/g k. Heat of dissociation of the nitrogen molecule (n2): The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Nitrogen, n2, is at standard conditions a colorless odorless gas. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The gas makes up the major portion of the atmosphere, but will not.
from www.numerade.com
Heat of dissociation of the nitrogen molecule (n2): 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Specific heat of nitrogen is 1.04 j/g k. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Nitrogen, n2, is at standard conditions a colorless odorless gas. The gas makes up the major portion of the atmosphere, but will not.
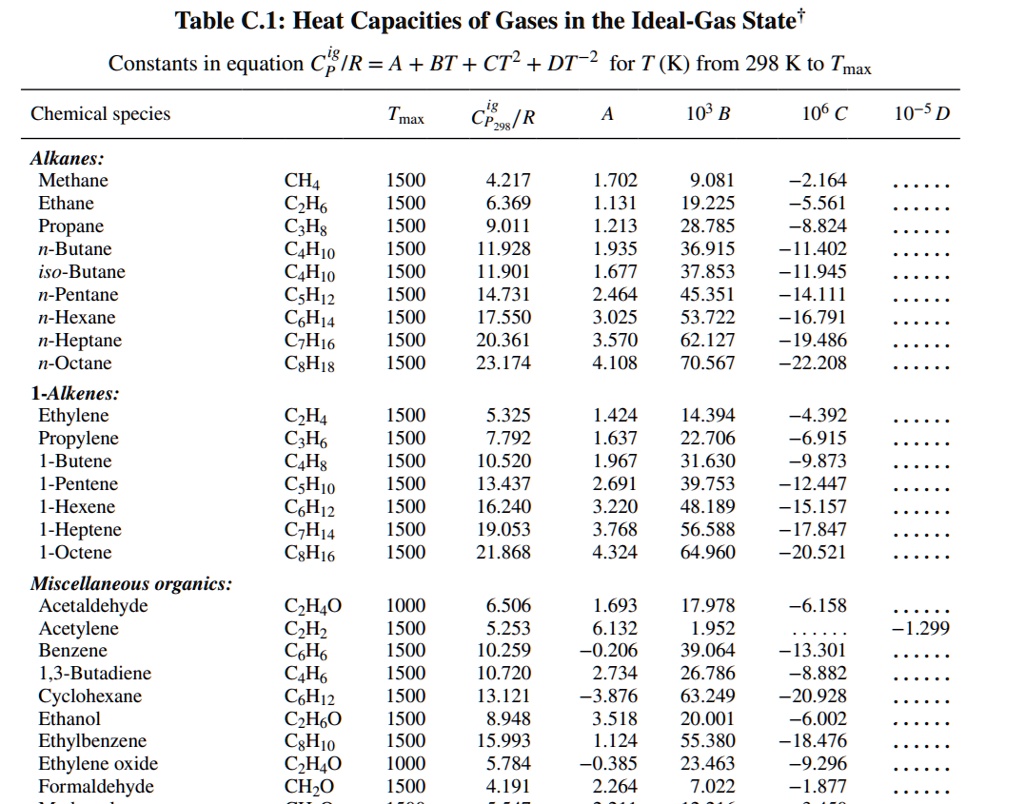
Table C.I Heat Capacities of Gases in the IdealGas State Constants in
Heat Capacity Ratio Of N2 S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Nitrogen, n2, is at standard conditions a colorless odorless gas. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Heat of dissociation of the nitrogen molecule (n2): Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. The gas makes up the major portion of the atmosphere, but will not. Specific heat of nitrogen is 1.04 j/g k. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas.
From www.slideserve.com
PPT Chapter 17 The first law of thermodynamics PowerPoint Heat Capacity Ratio Of N2 The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat of nitrogen is 1.04 j/g k. Nitrogen, n2, is at standard conditions a colorless odorless gas. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Specific heat, or. Heat Capacity Ratio Of N2.
From www.slideserve.com
PPT Determining the Specific Heat Capacity of Air PowerPoint Heat Capacity Ratio Of N2 The gas makes up the major portion of the atmosphere, but will not. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. 55 rows the table of specific heat capacities gives the. Heat Capacity Ratio Of N2.
From www.youtube.com
3. 11P13.2 CV 2 Specific Heat Capacities of Gases and Mean Free Path Heat Capacity Ratio Of N2 Nitrogen, n2, is at standard conditions a colorless odorless gas. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Heat of dissociation of the nitrogen molecule (n2): S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2). Heat Capacity Ratio Of N2.
From www.youtube.com
Ratio of Specific Heat Capacities YouTube Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. C p = heat capacity (j/mol*k) h°. Heat Capacity Ratio Of N2.
From www.numerade.com
SOLVED Compute the specific heat capacity at constant volume of Heat Capacity Ratio Of N2 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The gas makes up the major portion of the atmosphere, but will not. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas.. Heat Capacity Ratio Of N2.
From www.youtube.com
Equipartition Theorem Internal Energy (U) and Heat Capacity (Cv) L6 Heat Capacity Ratio Of N2 Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Nitrogen, n2, is at standard conditions a colorless odorless gas. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Heat. Heat Capacity Ratio Of N2.
From www.slideserve.com
PPT The First Law of Thermodynamics PowerPoint Presentation, free Heat Capacity Ratio Of N2 The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia ,. Heat Capacity Ratio Of N2.
From www.slideserve.com
PPT Heat Capacity PowerPoint Presentation, free download ID4997894 Heat Capacity Ratio Of N2 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Specific heat of nitrogen is 1.04 j/g k. Nitrogen, n2, is at standard conditions a colorless odorless gas. Specific heat, or specific. Heat Capacity Ratio Of N2.
From www.chegg.com
Solved EXPERIMENT 2 HEAT CAPACITY RATIO OF GASES The Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Nitrogen, n2, is at standard conditions a colorless odorless gas. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol). Heat Capacity Ratio Of N2.
From www.scribd.com
Heat capacity ratio PDF Gases Continuum Mechanics Heat Capacity Ratio Of N2 Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Heat of dissociation of the nitrogen molecule (n2): The gas makes up the major portion of the atmosphere, but will not. C p. Heat Capacity Ratio Of N2.
From studylib.net
HEAT CAPACITY RATIO FOR GASES (γ) Heat Capacity Ratio Of N2 Heat of dissociation of the nitrogen molecule (n2): C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. The specific heat. Heat Capacity Ratio Of N2.
From ar.inspiredpencil.com
Heat Capacity Chart Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Heat of dissociation of the nitrogen molecule (n2): 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The gas makes up the major portion of the. Heat Capacity Ratio Of N2.
From www.numerade.com
Table C.I Heat Capacities of Gases in the IdealGas State Constants in Heat Capacity Ratio Of N2 Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Heat of dissociation of the nitrogen molecule (n2): C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Specific heat of nitrogen is 1.04 j/g k. 55 rows the table of specific heat capacities gives the volumetric heat capacity as. Heat Capacity Ratio Of N2.
From www.toppr.com
The heat of reaction for N2 + 3H2→ 2NH3 at 27^oC is 91.94 kJ . What Heat Capacity Ratio Of N2 C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Heat of dissociation of the nitrogen molecule (n2): Specific heat of nitrogen is 1.04 j/g k. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3. Heat Capacity Ratio Of N2.
From www.slideserve.com
PPT Chapter 9 Energy, Enthalpy and Thermochemistry PowerPoint Heat Capacity Ratio Of N2 Specific heat of nitrogen is 1.04 j/g k. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. S° = a*ln (t) + b*t + c*t 2 /2. Heat Capacity Ratio Of N2.
From dokumen.tips
(PDF) Heat Capacity Ratio DOKUMEN.TIPS Heat Capacity Ratio Of N2 Nitrogen, n2, is at standard conditions a colorless odorless gas. Specific heat of nitrogen is 1.04 j/g k. The gas makes up the major portion of the atmosphere, but will not. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Combines with oxygen and hydrogen on sparking, forming nitric oxide. Heat Capacity Ratio Of N2.
From www.youtube.com
Heat Capacity and Specific Heat Chemistry Tutorial YouTube Heat Capacity Ratio Of N2 Specific heat of nitrogen is 1.04 j/g k. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Nitrogen, n2, is at standard conditions a colorless odorless gas. S° = a*ln (t) +. Heat Capacity Ratio Of N2.
From www.careerpower.in
What is Heat Capacity Definition, Equations, Examples and Types Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Specific heat of nitrogen is 1.04 j/g k. Heat of dissociation of the nitrogen molecule (n2): S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Nitrogen, n2, is at standard. Heat Capacity Ratio Of N2.
From www.youtube.com
CHEMISTRY 101 Specific heat capacity and calculating heat YouTube Heat Capacity Ratio Of N2 S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k).. Heat Capacity Ratio Of N2.
From www.researchgate.net
Temperature dependence of heat capacity ratio γ(t). Download Heat Capacity Ratio Of N2 The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific. Heat Capacity Ratio Of N2.
From www.researchgate.net
(a) Pressure dependence of the heat capacity ratio γ = c p /c v for Heat Capacity Ratio Of N2 S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. C p = heat capacity (j/mol*k) h°. Heat Capacity Ratio Of N2.
From www.chegg.com
Solved Compute the average specific heat at constant Heat Capacity Ratio Of N2 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3. Heat Capacity Ratio Of N2.
From physics.stackexchange.com
pressure Why isn't Argon's heat capacity ratio constant with Heat Capacity Ratio Of N2 Nitrogen, n2, is at standard conditions a colorless odorless gas. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Heat of dissociation of the nitrogen molecule (n2): S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) +. Heat Capacity Ratio Of N2.
From www.toppr.com
The Molar heat capacities of nitrogen at constant pressure and constant Heat Capacity Ratio Of N2 S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat of nitrogen is 1.04 j/g k. C p = heat capacity (j/mol*k) h° = standard. Heat Capacity Ratio Of N2.
From www.youtube.com
Ratio Of Heat Capacities Thermodynamic Relations Engineering Heat Capacity Ratio Of N2 The gas makes up the major portion of the atmosphere, but will not. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Nitrogen, n2, is at standard conditions a colorless odorless gas. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio. Heat Capacity Ratio Of N2.
From www.scribd.com
Heat Capacity Ratios For Selected Gases Download Free PDF Physical Heat Capacity Ratio Of N2 The gas makes up the major portion of the atmosphere, but will not. Heat of dissociation of the nitrogen molecule (n2): Nitrogen, n2, is at standard conditions a colorless odorless gas. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat, or specific heat capacity,. Heat Capacity Ratio Of N2.
From haipernews.com
How To Calculate Heat Capacity Ratio Haiper Heat Capacity Ratio Of N2 Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. The gas makes up the major portion of the atmosphere, but will not. 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Specific heat, or specific heat capacity, is a property. Heat Capacity Ratio Of N2.
From www.slideserve.com
PPT First Law of Thermodynamics PowerPoint Presentation, free Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. S° = a*ln (t) + b*t + c*t 2 /2 + d*t 3 /3 − e/ (2*t 2) + g. Specific heat of nitrogen is 1.04 j/g k. Nitrogen, n2, is at standard conditions a colorless odorless gas. The gas makes. Heat Capacity Ratio Of N2.
From haipernews.com
How To Calculate Specific Heat Capacity Bbc Bitesize Haiper Heat Capacity Ratio Of N2 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. Heat of dissociation of the nitrogen molecule (n2): Nitrogen, n2, is at standard conditions a colorless odorless gas. Specific heat of nitrogen is 1.04 j/g k. C p = heat capacity (j/mol*k) h° = standard enthalpy. Heat Capacity Ratio Of N2.
From calculator.academy
Heat Capacity Ratio Calculator Calculator Academy Heat Capacity Ratio Of N2 Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat of nitrogen is 1.04 j/g k. Combines with oxygen and hydrogen on sparking, forming nitric oxide. Heat Capacity Ratio Of N2.
From chempedia.info
Heat capacities, ratio Big Chemical Encyclopedia Heat Capacity Ratio Of N2 55 rows the table of specific heat capacities gives the volumetric heat capacity as well as the specific heat capacity of some substances and. The gas makes up the major portion of the atmosphere, but will not. Specific heat of nitrogen is 1.04 j/g k. Nitrogen, n2, is at standard conditions a colorless odorless gas. Heat of dissociation of the. Heat Capacity Ratio Of N2.
From www.chegg.com
Solved Experiment Ratio Heat Capacities Table.1 Data (The Heat Capacity Ratio Of N2 Nitrogen, n2, is at standard conditions a colorless odorless gas. The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). 55 rows the table of specific heat capacities gives the volumetric. Heat Capacity Ratio Of N2.
From www.chegg.com
Solved Table 2 Heat capacities of gases in the idealgas Heat Capacity Ratio Of N2 Nitrogen, n2, is at standard conditions a colorless odorless gas. C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Specific heat of nitrogen is 1.04 j/g k. 55 rows the table of specific heat capacities gives the volumetric heat. Heat Capacity Ratio Of N2.
From www.researchgate.net
Effect of heat capacity ratios on dimensionless a static and b total Heat Capacity Ratio Of N2 C p = heat capacity (j/mol*k) h° = standard enthalpy (kj/mol) s° = standard entropy (j/mol*k). The gas makes up the major portion of the atmosphere, but will not. Specific heat of nitrogen is 1.04 j/g k. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Heat of dissociation of. Heat Capacity Ratio Of N2.
From www.chegg.com
Solved Heat capacity of N2 at a constant pressure of 1 atm Heat Capacity Ratio Of N2 The specific heat (= specific heat capacity) at constant pressure and constant volume processes, and the ratio of specific heats and individual gas. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Combines with oxygen and hydrogen on sparking, forming nitric oxide and ammonia , respectively. Specific heat of nitrogen. Heat Capacity Ratio Of N2.