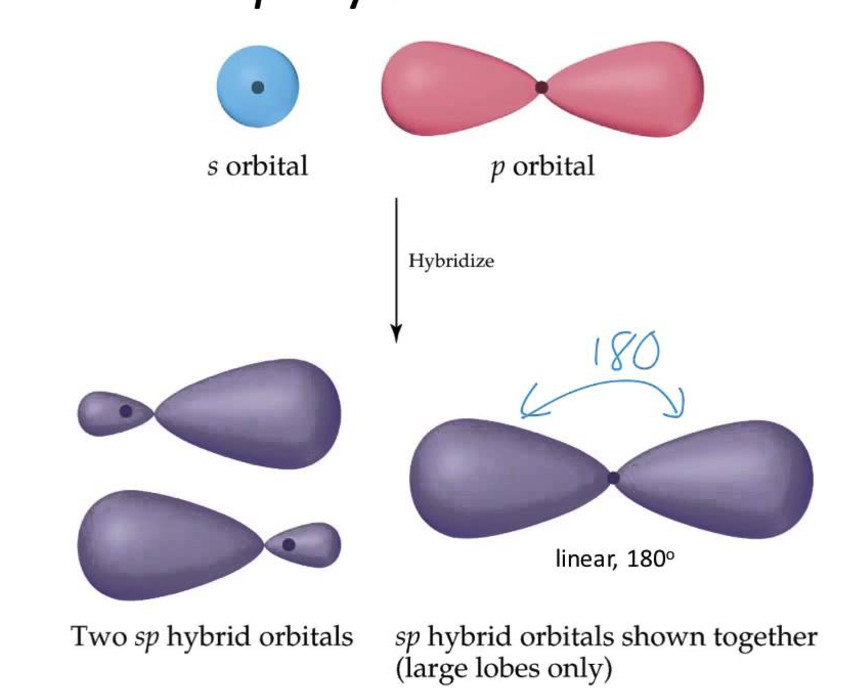
Hybridization With Examples . hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Consider an example of the simplest hydrocarbon. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. explanation of hybridization through examples. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing.
from classnotes.org.in
based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. Consider an example of the simplest hydrocarbon. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. explanation of hybridization through examples. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new.
Hybridisation Chemical Bonding and Molecular Structure, Chemistry
Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron explanation of hybridization through examples. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. Consider an example of the simplest hydrocarbon. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride.
From eduinput.com
Sp2hybridization, definition, explanation, examples and significance Hybridization With Examples other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Consider an example of the simplest hydrocarbon. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron based on the types of orbitals involved in. Hybridization With Examples.
From eduinput.com
sp3hybridization, definition, explanation, examples and significance Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron Consider an example of the simplest hydrocarbon. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly. Hybridization With Examples.
From www.geeksforgeeks.org
Hybridization Definition, Types, Rules, Examples Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. other than sp 3 hybridization, there are also other types of. Hybridization With Examples.
From sites.google.com
Hybridization of Orbitals Chemistry Topics Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. other than sp 3 hybridization, there are also other types of. Hybridization With Examples.
From www.youtube.com
5.2CHybridization Examples sp3d sp3d2 Lone Pairs YouTube Hybridization With Examples explanation of hybridization through examples. Consider an example of the simplest hydrocarbon. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy,. Hybridization With Examples.
From chemistrytalk.org
Hybridization and Hybrid Orbitals ChemTalk Hybridization With Examples other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. explanation of hybridization through examples. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2.. Hybridization With Examples.
From www.youtube.com
How to determine Hybridization s, sp, sp2, and sp3 Organic Hybridization With Examples hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. explanation. Hybridization With Examples.
From www.youtube.com
Hybridization sp3d sp3d2 sp3d3 Formation of PF5, SF6 and Hybridization With Examples explanation of hybridization through examples. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization is defined as the phenomenon of mixing up. Hybridization With Examples.
From www.vedantu.com
Hybridisation Important Concepts and Tips for JEE Hybridization With Examples Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3. Hybridization With Examples.
From chemwiki.ucdavis.edu
10.3 Hybridization of Atomic Orbitals Chemwiki Hybridization With Examples hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. Consider an example of the simplest hydrocarbon. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp,. Hybridization With Examples.
From eduinput.com
Sp2hybridization, definition, explanation, examples and significance Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. hybridization is defined as the phenomenon of mixing up (or merging). Hybridization With Examples.
From chemistnotes.com
Hybridization Definition, types and examples Chemistry Notes Hybridization With Examples Consider an example of the simplest hydrocarbon. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. explanation of hybridization through examples. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization can. Hybridization With Examples.
From www.ck12.org
Hybridization Example 4 ( Video ) Chemistry CK12 Foundation Hybridization With Examples Usually the hybridization on a certain atom can simply be determined by counting the total number of electron other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride.. Hybridization With Examples.
From www.collegesearch.in
Types of Hybridization Definitions, Examples, Key Features, Steps to Hybridization With Examples explanation of hybridization through examples. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. hybridization can be classified as sp 3, sp 2,. Hybridization With Examples.
From tableprofits.weebly.com
A molecule has sp3d hybridization with 3 lone pairs tableprofits Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Consider an example of the simplest hydrocarbon. Usually the hybridization on a certain atom can simply be determined. Hybridization With Examples.
From eduinput.com
Sphybridization, definition, explanation, examples and significance Hybridization With Examples Consider an example of the simplest hydrocarbon. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. explanation of hybridization through examples. based on the types of orbitals involved in mixing, the hybridization can be classified. Hybridization With Examples.
From www.organicchemistrytutor.com
Hybridization — Organic Chemistry Tutor Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. explanation of hybridization through examples. Consider an example of the simplest hydrocarbon. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron . Hybridization With Examples.
From eduinput.com
Sphybridization, definition, explanation, examples and significance Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Usually the hybridization on a certain. Hybridization With Examples.
From www.masterorganicchemistry.com
How To Determine Hybridization A Shortcut Master Organic Chemistry Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. explanation of hybridization through examples. Consider an example of the simplest hydrocarbon. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or. Hybridization With Examples.
From avantecnica.qualitypoolsboulder.com
Hybridization Definition, Types, Rules, Examples Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. explanation of hybridization through examples. Consider an example of the simplest hydrocarbon. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp. Hybridization With Examples.
From www.youtube.com
Hybridization of Atomic Orbitals Sigma & Pi Bonds Sp Sp2 Sp3 YouTube Hybridization With Examples Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an. Hybridization With Examples.
From general.chemistrysteps.com
Hybridization of Atomic Orbitals Chemistry Steps Hybridization With Examples Usually the hybridization on a certain atom can simply be determined by counting the total number of electron other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3. Hybridization With Examples.
From www.toppr.com
Explain sp^2 hybridization. Chemistry Questions Hybridization With Examples hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Consider an. Hybridization With Examples.
From classnotes.org.in
Hybridisation Chemical Bonding and Molecular Structure, Chemistry Hybridization With Examples explanation of hybridization through examples. Sp hybridization in ethyne the hybridization in ethyne is similar to the hybridization in magnesium hydride. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal. Hybridization With Examples.
From www.youtube.com
Lec1 Hybridization Theory of Organic Molecules YouTube Hybridization With Examples based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. hybridization can be classified as. Hybridization With Examples.
From www.collegesearch.in
Types of Hybridization Definitions, Examples, Key Features, Steps to Hybridization With Examples Usually the hybridization on a certain atom can simply be determined by counting the total number of electron based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. Consider an example of the simplest hydrocarbon. Sp hybridization in ethyne the hybridization in. Hybridization With Examples.
From collegedunia.com
Hybridization sp, sp2, sp3 & sp3d Atomic Orbitals, Properties & Examples Hybridization With Examples explanation of hybridization through examples. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. hybridization. Hybridization With Examples.
From www.animalia-life.club
Hybridization Orbitals Chart Hybridization With Examples hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. based on the types of orbitals involved in mixing, the hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2. other than sp 3 hybridization,. Hybridization With Examples.
From www.collegesearch.in
Types of Hybridization Definitions, Examples, Key Features, Steps to Hybridization With Examples hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Consider an example of the simplest hydrocarbon. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types. Hybridization With Examples.
From www.masterorganicchemistry.com
How To Determine Hybridization A Shortcut Master Organic Chemistry Hybridization With Examples hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. other than sp 3 hybridization, there are also other types of hybridization that include sp, sp 2, sp 3 d and sp 3 d 2. Usually the hybridization on a certain atom can. Hybridization With Examples.
From www.youtube.com
5.2BHybridization Examples sp sp2 sp3 YouTube Hybridization With Examples hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron hybridization is defined as the phenomenon of mixing up. Hybridization With Examples.
From www.collegesearch.in
Types of Hybridization Definitions, Examples, Key Features, Steps to Hybridization With Examples explanation of hybridization through examples. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Consider an example of the simplest hydrocarbon. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3. Hybridization With Examples.
From eduinput.com
Sphybridization, definition, explanation, examples and significance Hybridization With Examples explanation of hybridization through examples. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron based on the types of orbitals involved in mixing,. Hybridization With Examples.
From chem.libretexts.org
Hybridization Chemistry LibreTexts Hybridization With Examples hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types of orbitals involved in mixing. Consider an example of the simplest hydrocarbon. Usually the hybridization on a certain atom can simply be determined by counting the total number of electron explanation of. Hybridization With Examples.
From www.vedantu.com
Hybridization Types and Examples of Hybridization Hybridization With Examples Consider an example of the simplest hydrocarbon. hybridization is defined as the phenomenon of mixing up (or merging) of orbitals of an atom of nearly equal energy, giving rise to entirely new. hybridization can be classified as sp 3, sp 2, sp, sp 3 d, sp 3 d 2, or sp 3 d 3 based on the types. Hybridization With Examples.