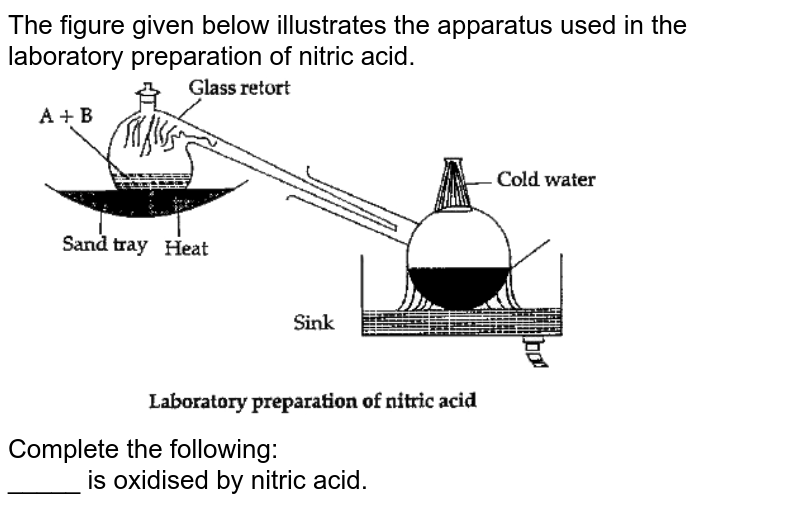
Laboratory Preparation Of Nitric Acid Pdf . (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. A more volatile acid can be displaced from its salt by a less volatile acid. The chemical nature and composition of nitric acid. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. This is the basic principle in the laboratory preparation of. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dilute 2 ml of standard iron.
from www.doubtnut.com
The chemical nature and composition of nitric acid. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. A more volatile acid can be displaced from its salt by a less volatile acid. Dilute 2 ml of standard iron. This is the basic principle in the laboratory preparation of. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact.
Explain the following In the laboratory preparation of nitric ac
Laboratory Preparation Of Nitric Acid Pdf Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. This is the basic principle in the laboratory preparation of. Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. A more volatile acid can be displaced from its salt by a less volatile acid. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. The chemical nature and composition of nitric acid.
From www.youtube.com
Preparation of Nitric Acid in Laboratory Reaction HNO3 in the Lab Laboratory Preparation Of Nitric Acid Pdf A more volatile acid can be displaced from its salt by a less volatile acid. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. The chemical nature and composition of nitric acid. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should. Laboratory Preparation Of Nitric Acid Pdf.
From ar.inspiredpencil.com
Manufacture Of Nitric Acid Laboratory Preparation Of Nitric Acid Pdf It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. This is the basic principle in the laboratory preparation of. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. Dilute. Laboratory Preparation Of Nitric Acid Pdf.
From www.doubtnut.com
Explain the following In the laboratory preparation of nitric ac Laboratory Preparation Of Nitric Acid Pdf 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. The chemical nature and composition of nitric acid. A more volatile acid. Laboratory Preparation Of Nitric Acid Pdf.
From www.scribd.com
Preparation of Nitric Acid PDF Laboratory Preparation Of Nitric Acid Pdf This is the basic principle in the laboratory preparation of. Dilute 2 ml of standard iron. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph. Laboratory Preparation Of Nitric Acid Pdf.
From brainly.in
Identify the process with which the image shown below is associated Laboratory Preparation Of Nitric Acid Pdf Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. Dilute 2 ml of standard iron. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Answer (i) dilute nitric acid is generally considered a typical acid except for it's. Laboratory Preparation Of Nitric Acid Pdf.
From testbook.com
Uses of Nitric Acid Introduction, Daily Life Use, Industrial Use Laboratory Preparation Of Nitric Acid Pdf Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. This is the basic principle in the laboratory. Laboratory Preparation Of Nitric Acid Pdf.
From solutionpharmacy.in
How is nitric acid prepared? Solution Parmacy Laboratory Preparation Of Nitric Acid Pdf This is the basic principle in the laboratory preparation of. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Laboratory preparation of nitric acid, although it has little or no commercial or. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
Nitric Acid & it's Preparation Method YouTube Laboratory Preparation Of Nitric Acid Pdf Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. A more volatile acid can be displaced from its salt by a less volatile acid. It is prepared in the laboratory by heating a mixture. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
How to prepare dilute solution of nitric acid HNO3 in laboratory YouTube Laboratory Preparation Of Nitric Acid Pdf It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. A more volatile acid can be displaced from its salt by a less volatile acid. This is the basic principle in the laboratory preparation of.. Laboratory Preparation Of Nitric Acid Pdf.
From iqclasses.in
Class 10 ICSE Chemistry Mostlikely QuestionBank Chapter Nitric Acid Laboratory Preparation Of Nitric Acid Pdf 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. Laboratory preparation of nitric acid, although it has little or no commercial. Laboratory Preparation Of Nitric Acid Pdf.
From www.shutterstock.com
Nitric Acid Bottle Chemical Laboratory Industry Stock Photo 2161868079 Laboratory Preparation Of Nitric Acid Pdf 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid. Laboratory Preparation Of Nitric Acid Pdf.
From www.alamy.com
Fully labelled diagram of the laboratory preparation of hydrogen Stock Laboratory Preparation Of Nitric Acid Pdf A more volatile acid can be displaced from its salt by a less volatile acid. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. This is the basic principle in the laboratory preparation of. The chemical nature and composition of nitric acid. It is prepared in the laboratory by. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
Laboratory Preparation of Nitric Acid Part 1 ICSE Class 10 Laboratory Preparation Of Nitric Acid Pdf It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. This is the basic principle. Laboratory Preparation Of Nitric Acid Pdf.
From studylib.net
Laboratory Preparation Of Nitric Acid Pdf Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. This is the basic principle in the laboratory preparation of. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is. Laboratory Preparation Of Nitric Acid Pdf.
From icsechemistry16.blogspot.com
Nitric acid lab preparation precautions Laboratory Preparation Of Nitric Acid Pdf Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. A more volatile acid can be displaced from its salt by a less volatile acid. This is the basic principle. Laboratory Preparation Of Nitric Acid Pdf.
From www.dbioscharts.com
CH 900 PREPARATION OF NITRIC ACID Dbios Charts Laboratory Preparation Of Nitric Acid Pdf This is the basic principle in the laboratory preparation of. Dilute 2 ml of standard iron. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. A more volatile acid can be displaced. Laboratory Preparation Of Nitric Acid Pdf.
From www.slideserve.com
PPT Nitric Acid (HNO3) PowerPoint Presentation ID2478951 Laboratory Preparation Of Nitric Acid Pdf This is the basic principle in the laboratory preparation of. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive. Laboratory Preparation Of Nitric Acid Pdf.
From www.scribd.com
Comprehensive Analysis of the Laboratory Preparation, Chemical Laboratory Preparation Of Nitric Acid Pdf Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. The chemical nature and composition of nitric acid. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals.. Laboratory Preparation Of Nitric Acid Pdf.
From studylib.net
Chemistry Nitric Acid (Solved) Laboratory Preparation Of Nitric Acid Pdf Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Dilute 2 ml of standard iron. The chemical nature and composition of nitric acid. Laboratory preparation of nitric acid, although it has little. Laboratory Preparation Of Nitric Acid Pdf.
From www.indiamart.com
Liquid Nitric Acid 60, Grade Standard Reagent Grade, for Laboratory Laboratory Preparation Of Nitric Acid Pdf (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. The chemical nature and composition of nitric acid. Dilute 2 ml of standard iron. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very. Laboratory Preparation Of Nitric Acid Pdf.
From www.studypool.com
SOLUTION Nitric acid explained laboratory preparation and properties Laboratory Preparation Of Nitric Acid Pdf 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Dissolve a specified quantity of the substance in 20 ml of water. Laboratory Preparation Of Nitric Acid Pdf.
From www.vecteezy.com
Preparation of Nitric Acidin in laboratory 23587363 Vector Art at Vecteezy Laboratory Preparation Of Nitric Acid Pdf Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid. Laboratory Preparation Of Nitric Acid Pdf.
From askfilo.com
10.2 LABORATORY PREPARATION OF NITRIC ACID RETORT METHOD1 potassium nit.. Laboratory Preparation Of Nitric Acid Pdf The chemical nature and composition of nitric acid. Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. This is the basic principle in the laboratory preparation. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
10ALab preparation of Nitric Acid YouTube Laboratory Preparation Of Nitric Acid Pdf Dilute 2 ml of standard iron. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. A more volatile acid can be displaced from its salt by a less volatile acid. This is the basic principle. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
Laboratory Preparation of Nitric AcidNITRIC ACID Class10 ICSE Laboratory Preparation Of Nitric Acid Pdf Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. 1 preparation of chromic acid (cleaning solution) h 2. Laboratory Preparation Of Nitric Acid Pdf.
From www.youtube.com
Preparation of Nitric Acid in Laboratory, Chemistry Lecture Sabaq.pk Laboratory Preparation Of Nitric Acid Pdf 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. (iii) in the laboratory preparation of nitric acid, an all glass apparatus. Laboratory Preparation Of Nitric Acid Pdf.
From www.alamy.com
Laboratory preparation of nitric (V) acid fully labeled diagram Stock Laboratory Preparation Of Nitric Acid Pdf This is the basic principle in the laboratory preparation of. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. Dilute 2 ml of standard iron. (iii) in the laboratory preparation of nitric. Laboratory Preparation Of Nitric Acid Pdf.
From www.doubtnut.com
In the laboratory preparation of nitric acid Name the reactants A Laboratory Preparation Of Nitric Acid Pdf Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Dilute 2 ml of standard iron. The chemical nature and composition. Laboratory Preparation Of Nitric Acid Pdf.
From askfilo.com
B. PREPARATION Of Nitrle Acid By distilling a mixture of Potassium or.. Laboratory Preparation Of Nitric Acid Pdf Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. The chemical nature and composition of nitric acid. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. This is the basic principle in the laboratory preparation of. Answer (i) dilute nitric acid is generally. Laboratory Preparation Of Nitric Acid Pdf.
From www.doubtnut.com
Explain the following In the laboratory preparation of nitric ac Laboratory Preparation Of Nitric Acid Pdf Laboratory preparation of nitric acid, although it has little or no commercial or industrial value. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Dilute 2 ml of standard iron. The chemical nature and composition of nitric acid. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals.. Laboratory Preparation Of Nitric Acid Pdf.
From www.alamy.com
Labelled diagram for laboratory preparation of hydrogen from zinc and Laboratory Preparation Of Nitric Acid Pdf Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. 1 preparation of chromic acid (cleaning solution) h 2 cro 4 it is a very corrosive solution and care should be taken to avoid its coming in contact. A more volatile acid can be displaced from its salt by a. Laboratory Preparation Of Nitric Acid Pdf.
From owlcation.com
3 Ways to Prepare Nitric Acid Owlcation Laboratory Preparation Of Nitric Acid Pdf A more volatile acid can be displaced from its salt by a less volatile acid. It is prepared in the laboratory by heating a mixture of alkali nitrate and concentrated sulphuric acid in a glass. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. (iii) in the laboratory preparation. Laboratory Preparation Of Nitric Acid Pdf.
From www.vcpmaps.com
Preparation of Nitric Acid Chart at Lowest Price in Delhi Laboratory Preparation Of Nitric Acid Pdf A more volatile acid can be displaced from its salt by a less volatile acid. This is the basic principle in the laboratory preparation of. Dissolve a specified quantity of the substance in 20 ml of water prescribed in the monograph in a nessler cylinder. Dilute 2 ml of standard iron. It is prepared in the laboratory by heating a. Laboratory Preparation Of Nitric Acid Pdf.
From owlcation.com
3 Ways to Prepare Nitric Acid Owlcation Laboratory Preparation Of Nitric Acid Pdf (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. Answer (i) dilute nitric acid is generally considered a typical acid except for it's reaction with metals. This is the basic principle in the laboratory preparation of. Dilute 2 ml of standard iron. The chemical nature and composition of nitric acid. Laboratory preparation of nitric acid,. Laboratory Preparation Of Nitric Acid Pdf.
From byjus.com
Preparation of Acetanilide Chemistry Practicals Class 12 Laboratory Preparation Of Nitric Acid Pdf A more volatile acid can be displaced from its salt by a less volatile acid. (iii) in the laboratory preparation of nitric acid, an all glass apparatus is used. This is the basic principle in the laboratory preparation of. Dilute 2 ml of standard iron. The chemical nature and composition of nitric acid. It is prepared in the laboratory by. Laboratory Preparation Of Nitric Acid Pdf.