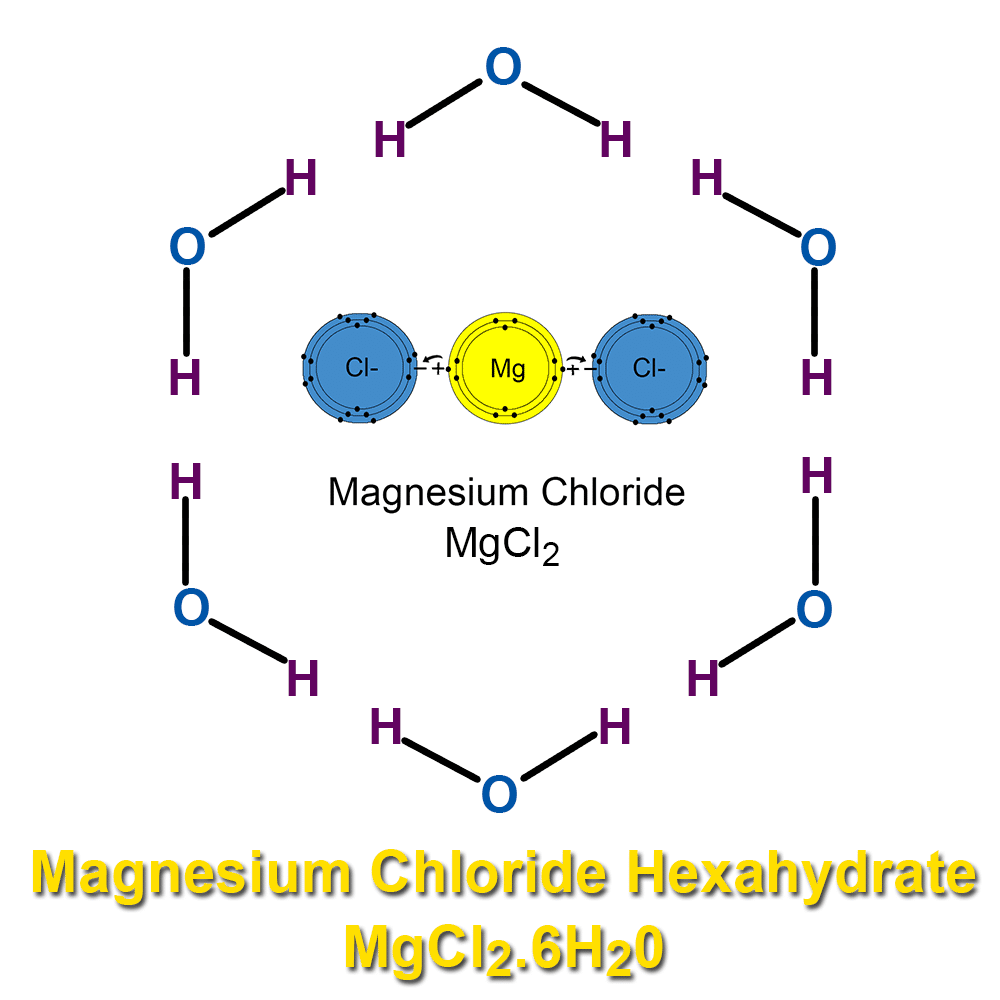
Magnesium Chloride Polar Or Nonpolar . Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. 3.8.5 illustrates the symmetric molecules that have polar. Determine if a molecule is polar or nonpolar. Electronegativity is a measure of the tendency of an atom to. And therefore, each chlorine atom accepts one electron from the magnesium atom. While each of the two chlorine atoms requires only one electron to be in the octet state. Explain how polar compounds differ from nonpolar compounds. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity.
from ar.inspiredpencil.com
For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. And therefore, each chlorine atom accepts one electron from the magnesium atom. 3.8.5 illustrates the symmetric molecules that have polar. Explain how polar compounds differ from nonpolar compounds. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Determine if a molecule is polar or nonpolar. While each of the two chlorine atoms requires only one electron to be in the octet state. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Electronegativity is a measure of the tendency of an atom to.
Magnesium Chloride Structure
Magnesium Chloride Polar Or Nonpolar And therefore, each chlorine atom accepts one electron from the magnesium atom. Determine if a molecule is polar or nonpolar. While each of the two chlorine atoms requires only one electron to be in the octet state. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Electronegativity is a measure of the tendency of an atom to. Explain how polar compounds differ from nonpolar compounds. And therefore, each chlorine atom accepts one electron from the magnesium atom. 3.8.5 illustrates the symmetric molecules that have polar. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity.
From askfilo.com
Fig. 4.15 Orbit structure of formation of magnesium chloride (MgCl2 ) mol.. Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Explain how polar compounds differ from nonpolar compounds. 3.8.5 illustrates the symmetric molecules that have polar. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Electronegativity is a measure. Magnesium Chloride Polar Or Nonpolar.
From avopix.com
Magnesium Chloride Properties and Chemical Royalty Free Stock Vector 1979677646 Magnesium Chloride Polar Or Nonpolar Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. 3.8.5 illustrates the symmetric molecules that have polar. And therefore, each chlorine atom accepts one electron from the magnesium atom. Explain how polar compounds differ from nonpolar compounds. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities,. Magnesium Chloride Polar Or Nonpolar.
From www.expii.com
Polar vs. Nonpolar Bonds — Overview & Examples Expii Magnesium Chloride Polar Or Nonpolar While each of the two chlorine atoms requires only one electron to be in the octet state. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. For example,. Magnesium Chloride Polar Or Nonpolar.
From whatsinsight.org
Is MgCl2 Ionic or Covalent? What's Insight Magnesium Chloride Polar Or Nonpolar Determine if a molecule is polar or nonpolar. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. While each of the two chlorine atoms requires only one electron to be in the octet state. And therefore, each chlorine atom accepts one electron from the magnesium atom. Electronegativity is a measure. Magnesium Chloride Polar Or Nonpolar.
From chem.libretexts.org
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts Magnesium Chloride Polar Or Nonpolar Explain how polar compounds differ from nonpolar compounds. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. While each of the two chlorine atoms requires only one electron to be in the octet state. Whether a bond is nonpolar or polar covalent is determined by a property of. Magnesium Chloride Polar Or Nonpolar.
From www.sciencephoto.com
Magnesium Chloride Stock Image C027/9847 Science Photo Library Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. And therefore, each chlorine atom accepts one electron from the magnesium atom. 3.8.5 illustrates the symmetric molecules that have polar. Explain how polar compounds differ from nonpolar compounds. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are. Magnesium Chloride Polar Or Nonpolar.
From tutorsuhu.com
What Is The Molecular Structure Of Magnesium Chloride Tutor Suhu Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. While each of the two chlorine atoms requires only one electron to be in the octet state. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each. Magnesium Chloride Polar Or Nonpolar.
From ar.inspiredpencil.com
Amino Acid Structure Chart Polar Nonpolar Magnesium Chloride Polar Or Nonpolar Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Explain how polar compounds differ from nonpolar compounds. 3.8.5 illustrates the symmetric molecules that have polar. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. And therefore,. Magnesium Chloride Polar Or Nonpolar.
From www.knowledgeboat.com
Chapter 2 Chemical Bonding Selina Solutions Concise Chemistry Class 10 ICSE KnowledgeBoat Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Electronegativity is a measure of the tendency of an atom to. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond.. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
Is MgCl2 (Magnesium chloride) Ionic or Covalent? YouTube Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Determine if a molecule is polar or nonpolar. Explain how polar compounds differ from nonpolar compounds. While each of the two chlorine atoms requires only one electron to be in the octet state. And therefore, each chlorine atom accepts one electron. Magnesium Chloride Polar Or Nonpolar.
From ar.inspiredpencil.com
Magnesium Chloride Structure Magnesium Chloride Polar Or Nonpolar And therefore, each chlorine atom accepts one electron from the magnesium atom. Electronegativity is a measure of the tendency of an atom to. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
Is HCl Polar or Nonpolar? YouTube Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar.. Magnesium Chloride Polar Or Nonpolar.
From www.visionlearning.com
Membranes II Biology Visionlearning Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. While each of the two chlorine atoms requires only one electron to be in the octet state. And therefore, each chlorine atom accepts one electron from the magnesium atom. Electronegativity is a measure of the tendency of an atom to. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities,. Magnesium Chloride Polar Or Nonpolar.
From www.science-revision.co.uk
Introduction to ionic bonding Magnesium Chloride Polar Or Nonpolar Explain how polar compounds differ from nonpolar compounds. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. 3.8.5 illustrates the symmetric molecules that have polar. And therefore, each chlorine atom accepts one electron from the magnesium atom. While each of the two chlorine atoms requires only one electron. Magnesium Chloride Polar Or Nonpolar.
From molekula.com
Purchase Magnesium chloride hexahydrate [7791186] online • Catalog • Molekula Group Magnesium Chloride Polar Or Nonpolar Explain how polar compounds differ from nonpolar compounds. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Determine if a molecule is polar or nonpolar. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Magnesium chloride. Magnesium Chloride Polar Or Nonpolar.
From www.slideserve.com
PPT 6 Electronegativity PowerPoint Presentation, free download ID5108349 Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. While each of the two chlorine atoms requires only one electron to be in the octet state. Determine if a. Magnesium Chloride Polar Or Nonpolar.
From www.numerade.com
SOLVED Classify each of the solutes . polar or nonpolar (yes/no} Polar Nonpolar Sucrose, Czh Magnesium Chloride Polar Or Nonpolar Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Explain how polar compounds differ from nonpolar compounds. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and. Magnesium Chloride Polar Or Nonpolar.
From www.dreamstime.com
3D Image of Magnesium Chloride Skeletal Formula Stock Illustration Illustration of control Magnesium Chloride Polar Or Nonpolar For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. While each of the two chlorine atoms requires only one electron to be in the octet state. Explain how polar compounds differ from nonpolar compounds. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
Is HCl Polar or Nonpolar ? YouTube Magnesium Chloride Polar Or Nonpolar Explain how polar compounds differ from nonpolar compounds. While each of the two chlorine atoms requires only one electron to be in the octet state. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Whether a bond is nonpolar or polar covalent is determined by a property of the. Magnesium Chloride Polar Or Nonpolar.
From www.chemicalslearning.com
Magnesium Chloride Formula, Properties and Uses Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. Electronegativity is a measure of the tendency of an atom to. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. While each of the two chlorine atoms requires only one electron to be in the octet state. Magnesium chloride is. Magnesium Chloride Polar Or Nonpolar.
From www.energycochem.com
Buy Magnesium Chloride on ENERGYCO LTD. Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Explain how polar compounds differ from nonpolar compounds. And therefore, each chlorine atom. Magnesium Chloride Polar Or Nonpolar.
From www.chegg.com
A student wanted to illustrate the structure of Magnesium Chloride Polar Or Nonpolar While each of the two chlorine atoms requires only one electron to be in the octet state. Electronegativity is a measure of the tendency of an atom to. 3.8.5 illustrates the symmetric molecules that have polar. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Explain how polar. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
Polar vs Nonpolar molecules How to tell? [GCE A Level Chemistry] YouTube Magnesium Chloride Polar Or Nonpolar 3.8.5 illustrates the symmetric molecules that have polar. Explain how polar compounds differ from nonpolar compounds. And therefore, each chlorine atom accepts one electron from the magnesium atom. Determine if a molecule is polar or nonpolar. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Electronegativity is a. Magnesium Chloride Polar Or Nonpolar.
From www.numerade.com
SOLVED Med commeni PreLaboratory Assignment Name Section Chemical Bonding and the Properties Magnesium Chloride Polar Or Nonpolar Determine if a molecule is polar or nonpolar. While each of the two chlorine atoms requires only one electron to be in the octet state. And therefore, each chlorine atom accepts one electron from the magnesium atom. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Explain how. Magnesium Chloride Polar Or Nonpolar.
From courses.lumenlearning.com
Covalent Bonds Biology for Majors I Magnesium Chloride Polar Or Nonpolar Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. While each of the two chlorine atoms requires only one electron to be in the octet state. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. 3.8.5 illustrates. Magnesium Chloride Polar Or Nonpolar.
From www.shutterstock.com
Magnesium Chloride Ionic Mineral Compound Necessary Stock Photo 2136112701 Shutterstock Magnesium Chloride Polar Or Nonpolar For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. 3.8.5 illustrates the symmetric molecules that have polar. Electronegativity is a measure of the tendency of an atom to.. Magnesium Chloride Polar Or Nonpolar.
From www.slideserve.com
PPT Nomenclature Chemical Formulas Reactions PowerPoint Presentation ID639117 Magnesium Chloride Polar Or Nonpolar Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. Explain how polar compounds differ from nonpolar compounds. And therefore, each chlorine atom accepts one electron from the magnesium atom. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity.. Magnesium Chloride Polar Or Nonpolar.
From www.toppr.com
Draw electron dot representation for the formation of magnesium chloride. Magnesium Chloride Polar Or Nonpolar Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. 3.8.5 illustrates the symmetric molecules that have polar. While each of the two chlorine atoms requires only one electron to be in the octet state. Explain how polar compounds differ from nonpolar compounds. Determine if a molecule is polar. Magnesium Chloride Polar Or Nonpolar.
From inci.guide
Magnesium Chloride Ingredient INCIGuide Magnesium Chloride Polar Or Nonpolar Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one to each of the chlorine atoms. Determine if a molecule is polar or nonpolar. Electronegativity is a measure of the tendency of an atom to. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. And therefore,. Magnesium Chloride Polar Or Nonpolar.
From stock.adobe.com
Amino Acids Types Table. Showing The Chemical Structure of Nonpolar Amino Acids. Vector Magnesium Chloride Polar Or Nonpolar Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. While each of the two chlorine atoms requires only one electron to be in the octet state. Electronegativity is a measure of the tendency of an atom to. And therefore, each chlorine atom accepts one electron from the magnesium atom. Determine. Magnesium Chloride Polar Or Nonpolar.
From sydneyyouthenglish.blogspot.com
Ch3cl Polar or Nonpolar Magnesium Chloride Polar Or Nonpolar Determine if a molecule is polar or nonpolar. Calculate the electronegativity difference (δen) and average (en) of the two electronegativities, and use the table below to determine the bond. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. Electronegativity is a measure of the tendency of an atom. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
NaCl Polar or Nonpolar (Sodium Chloride) YouTube Magnesium Chloride Polar Or Nonpolar Explain how polar compounds differ from nonpolar compounds. Electronegativity is a measure of the tendency of an atom to. 3.8.5 illustrates the symmetric molecules that have polar. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Magnesium chloride is an ionic compound because magnesium having two valence electrons donates one. Magnesium Chloride Polar Or Nonpolar.
From www.youtube.com
Is CH2Cl2 Polar or Nonpolar? (Dichloromethane) YouTube Magnesium Chloride Polar Or Nonpolar Determine if a molecule is polar or nonpolar. Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. And therefore, each chlorine atom accepts one electron from the magnesium atom. While each of the two chlorine atoms requires only one electron to be in the octet state. 3.8.5 illustrates the symmetric. Magnesium Chloride Polar Or Nonpolar.
From www.alibaba.com
Customized Is Mgcl2 Polar Or Nonpolar Of Bottom Price Buy Is Mgcl2 Polar Or Magnesium Chloride Polar Or Nonpolar Determine if a molecule is polar or nonpolar. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. 3.8.5 illustrates the symmetric molecules that have polar. Electronegativity is a measure of the tendency of an atom to. While each of the two chlorine atoms requires only one electron to. Magnesium Chloride Polar Or Nonpolar.
From www.toppr.com
Give orbital diagram of the followingmagnesium chloride, Magnesium Chloride Polar Or Nonpolar While each of the two chlorine atoms requires only one electron to be in the octet state. For example, symmetric molecules, like co 2, bf 3, and ccl 4, are nonpolar, although each bond in them is polar. 3.8.5 illustrates the symmetric molecules that have polar. Determine if a molecule is polar or nonpolar. And therefore, each chlorine atom accepts. Magnesium Chloride Polar Or Nonpolar.