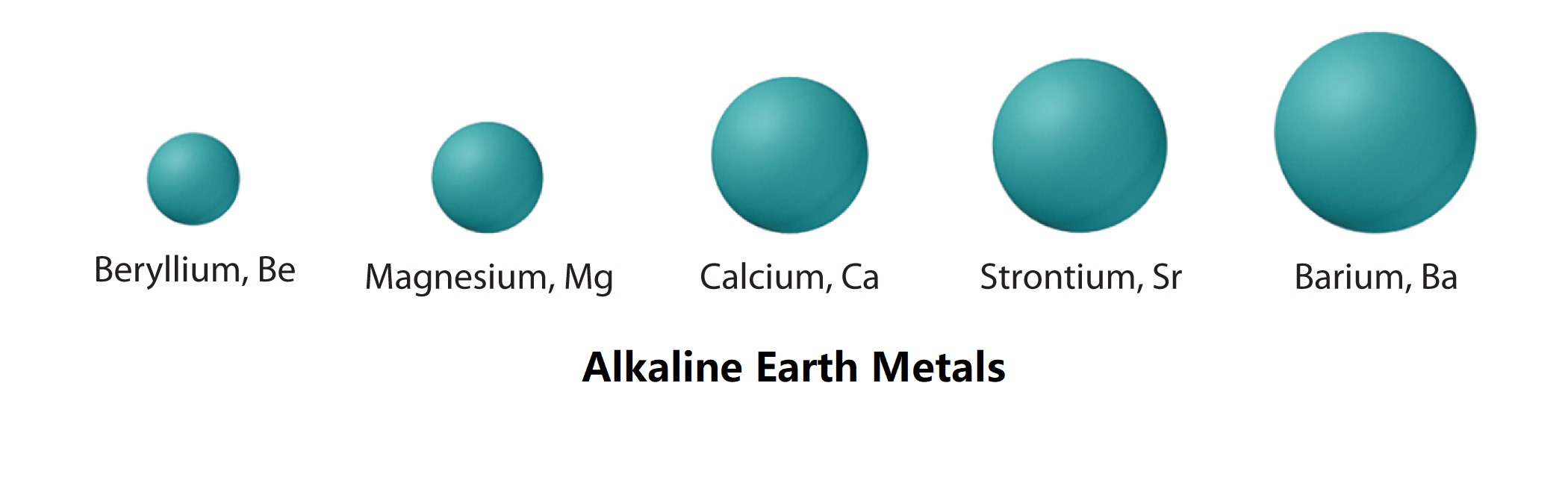
Properties And Facts Of Alkaline Earth Metals . The name “alkaline” comes from the fact that compounds. Learn where on the periodic table this element group is found. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals are six chemical elements in group 2 of the periodic table. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. They are beryllium (be), magnesium (mg),. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals have distinctive properties.
from facts.net
The name “alkaline” comes from the fact that compounds. The alkaline earth metals tend to. Learn where on the periodic table this element group is found. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals have distinctive properties. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. They are beryllium (be), magnesium (mg),. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the alkaline earth metals are six chemical elements in group 2 of the periodic table.
9 Mindblowing Facts About Alkaline Earth Metal
Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. alkaline earth metals have distinctive properties. The name “alkaline” comes from the fact that compounds. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Learn where on the periodic table this element group is found. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. They are beryllium (be), magnesium (mg),. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below).
From www.pw.live
What are the characteristics of the alkaline earth metals? Physics Wallah Properties And Facts Of Alkaline Earth Metals Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Learn where on the periodic table this element group is found. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals have distinctive properties. the alkaline earth metals or alkaline earths are a set of six elements. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation, free download ID Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. alkaline earth metals have distinctive properties. The alkaline earth metals tend to. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals or alkaline earths are a set of. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkali Metals and Alkaline Earth Metals PowerPoint Presentation Properties And Facts Of Alkaline Earth Metals The alkaline earth metals tend to. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The name “alkaline” comes from the fact that compounds. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Learn. Properties And Facts Of Alkaline Earth Metals.
From pediaa.com
Difference Between Alkali Metals and Alkaline Earth Metals Definition Properties And Facts Of Alkaline Earth Metals the alkaline earth metals are six chemical elements in group 2 of the periodic table. alkaline earth metals have distinctive properties. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The alkaline. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation ID2372229 Properties And Facts Of Alkaline Earth Metals Learn where on the periodic table this element group is found. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. the alkaline earth metals are six chemical elements in group 2 of the. Properties And Facts Of Alkaline Earth Metals.
From byjus.com
Alkaline Earth Metals Occurrence and Extraction,Physical Properties Properties And Facts Of Alkaline Earth Metals alkaline earth metals have distinctive properties. They are beryllium (be), magnesium (mg),. The alkaline earth metals tend to. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. the alkaline earth metals are six chemical elements in group 2 of the periodic table. The name “alkaline”. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation ID2372300 Properties And Facts Of Alkaline Earth Metals Learn where on the periodic table this element group is found. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The name “alkaline” comes from the fact that compounds. They are beryllium (be), magnesium (mg),. the alkaline earth metals are six chemical elements in group 2. Properties And Facts Of Alkaline Earth Metals.
From www.britannica.com
Alkalineearth metal chemical element Britannica Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. The alkaline earth metals tend to. The name “alkaline” comes from the fact that compounds. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Learn where on the periodic table this element group is found. alkaline earth metals have distinctive properties. the alkaline earth metals. Properties And Facts Of Alkaline Earth Metals.
From www.youtube.com
Occurrence of ALKALI & ALKALINE Earth Metals SBlock Elements F Properties And Facts Of Alkaline Earth Metals The name “alkaline” comes from the fact that compounds. the alkaline earth metals are six chemical elements in group 2 of the periodic table. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. the alkaline earth. Properties And Facts Of Alkaline Earth Metals.
From www.sliderbase.com
Element Classes Presentation Chemistry Properties And Facts Of Alkaline Earth Metals alkaline earth metals have distinctive properties. Learn where on the periodic table this element group is found. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the alkaline earth. Properties And Facts Of Alkaline Earth Metals.
From thechemistrynotes.com
Comparison of properties of Alkali and Alkaline Earth Metals Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals have distinctive properties. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Learn where on the periodic table this element group is found. the alkaline earth metals or alkaline. Properties And Facts Of Alkaline Earth Metals.
From www.thoughtco.com
What Are the Properties of the Alkaline Earth Metals? Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. alkaline earth metals have distinctive properties. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The name “alkaline” comes from the fact that compounds. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state.. Properties And Facts Of Alkaline Earth Metals.
From facts.net
9 Mindblowing Facts About Alkaline Earth Metal Properties And Facts Of Alkaline Earth Metals The name “alkaline” comes from the fact that compounds. They are beryllium (be), magnesium (mg),. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. Understand properties, electronic configuration,. Properties And Facts Of Alkaline Earth Metals.
From www.britannica.com
Alkalineearth metal Properties, List, & Reactivity Britannica Properties And Facts Of Alkaline Earth Metals alkaline earth metals have distinctive properties. Learn where on the periodic table this element group is found. the alkaline earth metals are six chemical elements in group 2 of the periodic table. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals. Properties And Facts Of Alkaline Earth Metals.
From www.tes.com
Alkali and Alkaline Earth Metals Venn Diagram Teaching Resources Properties And Facts Of Alkaline Earth Metals The alkaline earth metals tend to. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the alkaline earth metals are six chemical elements in group 2 of the periodic table.. Properties And Facts Of Alkaline Earth Metals.
From www.redbubble.com
"Element Infographics The Alkaline Earth Metals" Posters by Compound Properties And Facts Of Alkaline Earth Metals Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals are six chemical elements in group 2 of the periodic table. Learn where on the periodic table this element group is found. The alkaline earth metals tend to. alkaline earth metals have distinctive properties. They are beryllium (be), magnesium (mg),. Understand. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Chapter 20 alkali metals, alkalineearth metals PowerPoint Properties And Facts Of Alkaline Earth Metals Learn where on the periodic table this element group is found. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The alkaline earth metals tend to. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth. Properties And Facts Of Alkaline Earth Metals.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Properties And Facts Of Alkaline Earth Metals Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). They are beryllium (be), magnesium (mg),. the alkaline earth metals or alkaline earths are a. Properties And Facts Of Alkaline Earth Metals.
From www.expii.com
Alkaline Earth Metals — Overview & Properties Expii Properties And Facts Of Alkaline Earth Metals Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals are six chemical elements in group 2 of the periodic table. alkaline earth metals have distinctive properties. They are beryllium (be), magnesium (mg),. the alkaline earth metals or alkaline earths are a set of six elements found in the second. Properties And Facts Of Alkaline Earth Metals.
From www.youtube.com
Properties of the Alkaline Earth Metals YouTube Properties And Facts Of Alkaline Earth Metals alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Group 2 elements are referred to as “alkaline earth” metals (tan column below). Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline earth metals tend to. the alkaline earth metals are six chemical elements in. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT ELEMENTS CHEMICAL & PHYSICAL PROPERTIES PowerPoint Presentation Properties And Facts Of Alkaline Earth Metals the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals are six chemical elements in group 2 of the periodic table. The name “alkaline” comes from the fact that compounds.. Properties And Facts Of Alkaline Earth Metals.
From slidedocnow.blogspot.com
Properties Of Alkaline Earth Metals slidedocnow Properties And Facts Of Alkaline Earth Metals The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). Learn where on the periodic table this element group is found. the alkaline earth metals are six chemical elements in group 2 of the periodic table. alkaline earth metals are good reducing agents that tend to form the +2. Properties And Facts Of Alkaline Earth Metals.
From studylib.net
1819Alkaline Earth Metals Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Learn where on the periodic table this element group is found. The name “alkaline” comes from the fact that compounds. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column. Properties And Facts Of Alkaline Earth Metals.
From factfile.org
10 Facts about Alkaline Earth Metals Fact File Properties And Facts Of Alkaline Earth Metals the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The name “alkaline” comes from the fact that compounds. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. Group 2 elements are referred to as “alkaline earth” metals (tan column below). . Properties And Facts Of Alkaline Earth Metals.
From chemistnotes.com
Alkali and alkaline earth metals Archives Chemistry Notes Properties And Facts Of Alkaline Earth Metals the alkaline earth metals are six chemical elements in group 2 of the periodic table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). Learn where on the periodic table this element group is found. The name “alkaline” comes from the fact that compounds. The alkaline earth metals tend to. Understand properties, electronic configuration, analogous. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Section 7.7 Group Trends for the Active Metals PowerPoint Properties And Facts Of Alkaline Earth Metals The alkaline earth metals tend to. They are beryllium (be), magnesium (mg),. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. the alkaline earth metals are six chemical elements in group 2 of the periodic table. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of. Properties And Facts Of Alkaline Earth Metals.
From thechemistrynotes.com
Trends of the Properties of Group 2 (Alkaline Earth) Metals Properties And Facts Of Alkaline Earth Metals Group 2 elements are referred to as “alkaline earth” metals (tan column below). The alkaline earth metals tend to. The name “alkaline” comes from the fact that compounds. Learn where on the periodic table this element group is found. alkaline earth metals have distinctive properties. the alkaline earth metals are six chemical elements in group 2 of the. Properties And Facts Of Alkaline Earth Metals.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. alkaline earth metals have distinctive properties. the alkaline earth metals are six chemical elements in group 2 of the periodic table. The alkaline earth metals tend to. The name “alkaline” comes from the fact that compounds.. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkali Metals and Alkaline Earth Metals PowerPoint Presentation Properties And Facts Of Alkaline Earth Metals the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The name “alkaline” comes from the fact that compounds. They are beryllium (be), magnesium (mg),. alkaline earth metals have distinctive properties. Learn where on the periodic table this element group is found. Group 2 elements are referred. Properties And Facts Of Alkaline Earth Metals.
From www.youtube.com
Alkali Metals Alkaline Earth Metals Understanding the Properties and Properties And Facts Of Alkaline Earth Metals Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals have distinctive properties. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The name “alkaline” comes from the fact that compounds. the alkaline earth metals or alkaline earths are a set of six elements found in. Properties And Facts Of Alkaline Earth Metals.
From knordslearning.com
Alkaline Earth Metals Periodic Table (With Images) Properties And Facts Of Alkaline Earth Metals the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. The name “alkaline” comes from the fact that compounds. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. They are beryllium (be), magnesium (mg),. Learn. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation, free download ID Properties And Facts Of Alkaline Earth Metals alkaline earth metals have distinctive properties. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Learn where on the periodic table this element group is found. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the alkaline earth metals are six chemical elements in group 2 of. Properties And Facts Of Alkaline Earth Metals.
From www.haikudeck.com
Alkaline Earth Metals by novi.czyscon Properties And Facts Of Alkaline Earth Metals The name “alkaline” comes from the fact that compounds. Understand properties, electronic configuration, analogous behaviour, reactivity, ionization energy of group 2 alkaline earth metals. The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the alkaline earth metals or alkaline earths are a set of six elements. Properties And Facts Of Alkaline Earth Metals.
From utedzz.blogspot.com
Periodic Table Showing Alkali Metals Alkaline Earth Metals Periodic Properties And Facts Of Alkaline Earth Metals The alkaline earth metals tend to. alkaline earth metals have distinctive properties. Learn where on the periodic table this element group is found. the alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (be), magnesium (mg),. the alkaline earth metals or alkaline earths are a set of six elements. Properties And Facts Of Alkaline Earth Metals.
From www.slideserve.com
PPT Elements and their Properties PowerPoint Presentation, free Properties And Facts Of Alkaline Earth Metals They are beryllium (be), magnesium (mg),. the alkaline earth metals or alkaline earths are a set of six elements found in the second group (column) of the. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth. Properties And Facts Of Alkaline Earth Metals.