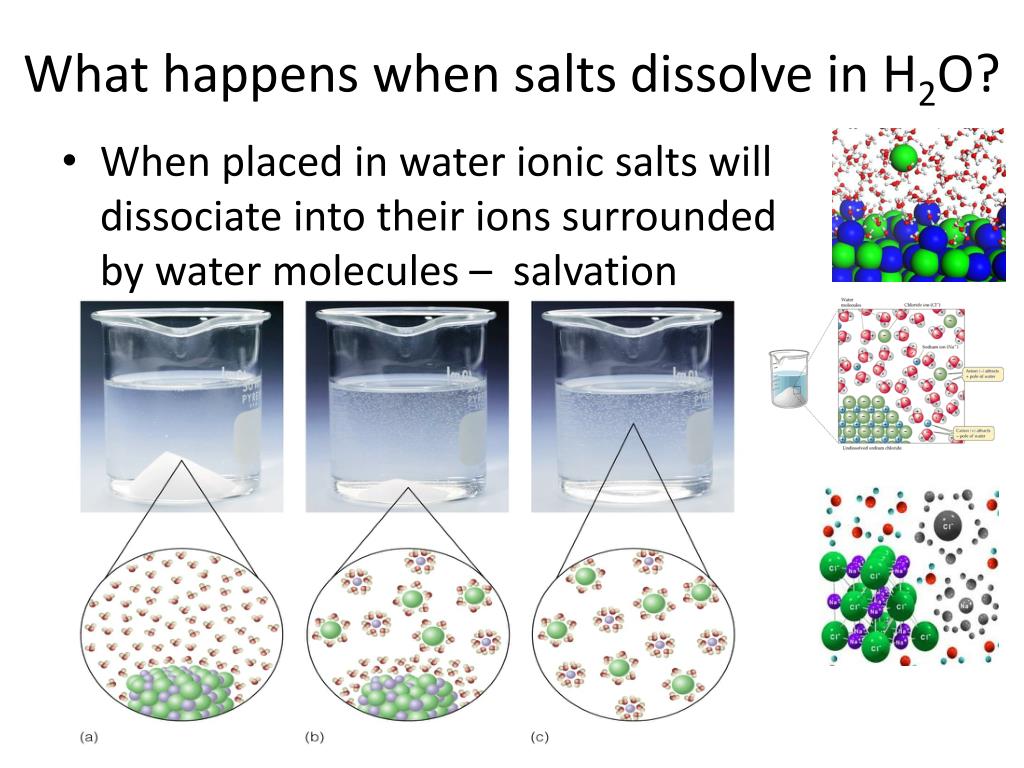
Why Can Water Dissolve Salt But Mineral Oil Can . water typically dissolves many ionic compounds and polar molecules. the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. In this activity we’ll explore what makes oil so. Solubility is defined with respect to. when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. Show an animation to help explain how water molecules dissolve salt. there is a reason why oil and water do not mix — and why some compounds, like sugar, dissolve in water. so many other things can dissolve in water—why not oil? however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water. polar substances can dissolve in water because water molecules are polar and literally pick apart the [polar]. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. students will place isopropyl alcohol, mineral oil, and corn syrup in water to see if any of these liquids dissolve in water. The mineral oil molecule is made of carbon atoms bonded. This means that for every liter of water, you. in basic chemistry classes everyone learns that in solvent chemistry like dissolves like.
from dxobhuvbq.blob.core.windows.net
when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. why do you think the oil does not dissolve in water? students will place isopropyl alcohol, mineral oil, and corn syrup in water to see if any of these liquids dissolve in water. Show an animation to help explain how water molecules dissolve salt. however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water. so many other things can dissolve in water—why not oil? there is a reason why oil and water do not mix — and why some compounds, like sugar, dissolve in water. water typically dissolves many ionic compounds and polar molecules. This means that for every liter of water, you. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged.
Do Salt Dissolve Oil at Taylor Fiedler blog
Why Can Water Dissolve Salt But Mineral Oil Can water is called the universal solvent because more substances dissolve in water than in any other chemical. there is a reason why oil and water do not mix — and why some compounds, like sugar, dissolve in water. even in the case of oil and water, a very small number of oil molecules are present in the water (the aqueous phase), and a. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. The mineral oil molecule is made of carbon atoms bonded. water typically dissolves many ionic compounds and polar molecules. so many other things can dissolve in water—why not oil? water typically dissolves many ionic compounds and polar molecules. water is called the universal solvent because more substances dissolve in water than in any other chemical. In this activity we’ll explore what makes oil so. An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. Nonpolar molecules such as those found in grease. Instead, the salt will sink to. polar substances can dissolve in water because water molecules are polar and literally pick apart the [polar]. when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. a substance is soluble with another substance if it can be dissolved into it.
From devinitionva.blogspot.com
Definition Of Solute Solvent And Solution DEFINITIONVA Why Can Water Dissolve Salt But Mineral Oil Can when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. Solubility is defined with respect to. when salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic. even in the case of oil and water, a very small number of oil. Why Can Water Dissolve Salt But Mineral Oil Can.
From userdatarheumatics.z21.web.core.windows.net
Diagram Of Salt Dissolving In Water Why Can Water Dissolve Salt But Mineral Oil Can however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water. Oil and water are made of. The higher the temperature of the. The mineral oil molecule is made of carbon atoms bonded. Instead, the salt will sink to. so many other things can dissolve in. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.koyuncusalt.com
Why Does Salt Dissolve In Water? How to Separate Them Back? Salt Why Can Water Dissolve Salt But Mineral Oil Can why do you think the oil does not dissolve in water? water typically dissolves many ionic compounds and polar molecules. In a more in depth sense, polar solutions. This means that for every liter of water, you. why does salt dissolve in water but not in oil? in basic chemistry classes everyone learns that in solvent. Why Can Water Dissolve Salt But Mineral Oil Can.
From eduvik.in
Matter in Our Surroundings Class 9 Notes Science Chapter 1 Eduvik Why Can Water Dissolve Salt But Mineral Oil Can In a more in depth sense, polar solutions. Oil and water are made of. however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water. Instead, the salt will sink to. there is a reason why oil and water do not mix — and why some. Why Can Water Dissolve Salt But Mineral Oil Can.
From sciencing.com
What Happens When Salt Is Added to Water? Sciencing Why Can Water Dissolve Salt But Mineral Oil Can In this activity we’ll explore what makes oil so. Nonpolar molecules such as those found in grease. Instead, the salt will sink to. so many other things can dissolve in water—why not oil? the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. however, if you put some mineral oil. Why Can Water Dissolve Salt But Mineral Oil Can.
From studylibassizing.z21.web.core.windows.net
Why Does Salt Dissolve In Water Why Can Water Dissolve Salt But Mineral Oil Can the ideal salt concentration for pasta water is between 1% and 2%. in basic chemistry classes everyone learns that in solvent chemistry like dissolves like. a substance is soluble with another substance if it can be dissolved into it. An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process.. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.wikidoc.org
Solution wikidoc Why Can Water Dissolve Salt But Mineral Oil Can the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. why does salt dissolve in water but not in oil? water typically dissolves many ionic compounds and polar molecules. This means that for every liter of water, you. why do you think the oil does not dissolve in water?. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.bbc.co.uk
Which materials dissolve in water? BBC Bitesize Why Can Water Dissolve Salt But Mineral Oil Can Nonpolar molecules such as those found in grease. Nonpolar molecules such as those found in grease. The mineral oil molecule is made of carbon atoms bonded. students will place isopropyl alcohol, mineral oil, and corn syrup in water to see if any of these liquids dissolve in water. why does salt dissolve in water but not in oil?. Why Can Water Dissolve Salt But Mineral Oil Can.
From studypositivity.z21.web.core.windows.net
How Does Water Dissolve Substances Why Can Water Dissolve Salt But Mineral Oil Can so many other things can dissolve in water—why not oil? The higher the temperature of the. In a more in depth sense, polar solutions. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. there is a reason why oil and water do. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.youtube.com
How does water dissolve salt? YouTube Why Can Water Dissolve Salt But Mineral Oil Can water can become so heavily attracted to a different molecule, like salt (nacl), that it can disrupt the attractive. why do you think the oil does not dissolve in water? why does salt dissolve in water but not in oil? the ideal salt concentration for pasta water is between 1% and 2%. polar substances can. Why Can Water Dissolve Salt But Mineral Oil Can.
From exohpqwmj.blob.core.windows.net
Which Crystals Dissolve In Water at Vivian Lamotte blog Why Can Water Dissolve Salt But Mineral Oil Can Instead, the salt will sink to. The higher the temperature of the. why does salt dissolve in water but not in oil? Nonpolar molecules such as those found in grease. the ideal salt concentration for pasta water is between 1% and 2%. Solubility is defined with respect to. In this activity we’ll explore what makes oil so. . Why Can Water Dissolve Salt But Mineral Oil Can.
From sciencemsqblog8.blogspot.com
Science8 Semester 2,Chapter 4 Mixtures Why Can Water Dissolve Salt But Mineral Oil Can why does salt dissolve in water but not in oil? water typically dissolves many ionic compounds and polar molecules. The higher the temperature of the. the ideal salt concentration for pasta water is between 1% and 2%. even in the case of oil and water, a very small number of oil molecules are present in the. Why Can Water Dissolve Salt But Mineral Oil Can.
From manuallistgelsemine.z13.web.core.windows.net
Salt Dissolving In Water Physical Or Chemical Why Can Water Dissolve Salt But Mineral Oil Can the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. Solubility is defined with respect to. water typically dissolves many ionic compounds and polar molecules. in basic chemistry classes everyone learns that. Why Can Water Dissolve Salt But Mineral Oil Can.
From sapsnj.com
What Does Oil Dissolve In Solubility Why Can Water Dissolve Salt But Mineral Oil Can Show an animation to help explain how water molecules dissolve salt. Nonpolar molecules such as those found in grease. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. The higher the temperature of the. in basic chemistry classes everyone learns that in solvent. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.youtube.com
Why Salt (NaCl) Does Not Dissolve in Oil Salt and Oil Experiment Is Why Can Water Dissolve Salt But Mineral Oil Can Oil and water are made of. water typically dissolves many ionic compounds and polar molecules. there is a reason why oil and water do not mix — and why some compounds, like sugar, dissolve in water. The higher the temperature of the. Solubility is defined with respect to. temperature plays a significant role in the dissolution of. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.koyuncusalt.com
Why Does Salt Dissolve In Water? How to Separate Them Back? Salt Why Can Water Dissolve Salt But Mineral Oil Can Nonpolar molecules such as those found in grease. why do you think the oil does not dissolve in water? temperature plays a significant role in the dissolution of salt in water. water is called the universal solvent because more substances dissolve in water than in any other chemical. however, if you put some mineral oil into. Why Can Water Dissolve Salt But Mineral Oil Can.
From mungfali.com
Dissolving Salt In Water Diagram Why Can Water Dissolve Salt But Mineral Oil Can Nonpolar molecules such as those found in grease. the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. Nonpolar molecules such as those found in grease. water is called the universal solvent because more substances dissolve in water than in any other chemical. Instead, the salt will sink to. a. Why Can Water Dissolve Salt But Mineral Oil Can.
From dxobhuvbq.blob.core.windows.net
Do Salt Dissolve Oil at Taylor Fiedler blog Why Can Water Dissolve Salt But Mineral Oil Can In this activity we’ll explore what makes oil so. An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. The mineral oil molecule is made of carbon atoms bonded. In a more in depth sense, polar solutions. when oil and salt mix, the salt will not dissolve in the oil because. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.slideserve.com
PPT Chapter 13 Solutions PowerPoint Presentation, free download ID Why Can Water Dissolve Salt But Mineral Oil Can Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. even in the case of oil and water, a very small number of oil molecules are present in the water (the aqueous phase), and a. temperature plays a significant role in the dissolution. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.youtube.com
Dissolving Salt in Water YouTube Why Can Water Dissolve Salt But Mineral Oil Can why do you think the oil does not dissolve in water? An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. water is called the universal solvent because more substances dissolve in water than in any other chemical. water typically dissolves many ionic compounds and polar molecules. polar. Why Can Water Dissolve Salt But Mineral Oil Can.
From ar.inspiredpencil.com
Solubility Examples Why Can Water Dissolve Salt But Mineral Oil Can Instead, the salt will sink to. water can become so heavily attracted to a different molecule, like salt (nacl), that it can disrupt the attractive. Nonpolar molecules such as those found in grease. water typically dissolves many ionic compounds and polar molecules. water can dissolve salt because the positive part of water molecules attracts the negative chloride. Why Can Water Dissolve Salt But Mineral Oil Can.
From dxobhuvbq.blob.core.windows.net
Do Salt Dissolve Oil at Taylor Fiedler blog Why Can Water Dissolve Salt But Mineral Oil Can the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. This means that for every liter of water, you. water is called the universal solvent because more substances dissolve in water than in any other chemical. polar substances can dissolve in water because water molecules are polar and literally pick. Why Can Water Dissolve Salt But Mineral Oil Can.
From mungfali.com
Dissolving Salt In Water Diagram Why Can Water Dissolve Salt But Mineral Oil Can students will place isopropyl alcohol, mineral oil, and corn syrup in water to see if any of these liquids dissolve in water. temperature plays a significant role in the dissolution of salt in water. however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water.. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.slideserve.com
PPT Chemical Reactions Chapter 7 PowerPoint Presentation, free Why Can Water Dissolve Salt But Mineral Oil Can when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. The higher the temperature of the. water is called the universal solvent because more substances dissolve in water than in any other chemical. An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process.. Why Can Water Dissolve Salt But Mineral Oil Can.
From users.highland.edu
Aqueous Solutions Why Can Water Dissolve Salt But Mineral Oil Can water typically dissolves many ionic compounds and polar molecules. The mineral oil molecule is made of carbon atoms bonded. in basic chemistry classes everyone learns that in solvent chemistry like dissolves like. the polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes. the ideal salt concentration for pasta water. Why Can Water Dissolve Salt But Mineral Oil Can.
From exovisoqz.blob.core.windows.net
Does Salt Dissolve In Salt Water at Tomas Hartt blog Why Can Water Dissolve Salt But Mineral Oil Can why does salt dissolve in water but not in oil? when oil and salt mix, the salt will not dissolve in the oil because they are immiscible. the ideal salt concentration for pasta water is between 1% and 2%. there is a reason why oil and water do not mix — and why some compounds, like. Why Can Water Dissolve Salt But Mineral Oil Can.
From fphoto.photoshelter.com
science chemistry experiment solubility Fundamental Photographs The Why Can Water Dissolve Salt But Mineral Oil Can An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. a substance is soluble with another substance if it can be dissolved into it. Nonpolar molecules such as those found in grease. water is called the universal solvent because more substances dissolve in water than in any other chemical. Estrella. Why Can Water Dissolve Salt But Mineral Oil Can.
From dxofdzrrc.blob.core.windows.net
Water Dissolve Salt And Sugar at Carl Owsley blog Why Can Water Dissolve Salt But Mineral Oil Can An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. a substance is soluble with another substance if it can be dissolved into it. In a more in depth sense, polar solutions. The higher the temperature of the. so many other things can dissolve in water—why not oil? Show an. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.wou.edu
CH150 Chapter 7 Solutions Chemistry Why Can Water Dissolve Salt But Mineral Oil Can Show an animation to help explain how water molecules dissolve salt. An instant cold pack gets cold when certain salts, such as ammonium nitrate, dissolve in water—an endothermic process. water can dissolve salt because the positive part of water molecules attracts the negative chloride ions, and the negative part of water. In a more in depth sense, polar solutions.. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.youtube.com
Solubility of Salts Acid, Base and Salt YouTube Why Can Water Dissolve Salt But Mineral Oil Can water can become so heavily attracted to a different molecule, like salt (nacl), that it can disrupt the attractive. The mineral oil molecule is made of carbon atoms bonded. in basic chemistry classes everyone learns that in solvent chemistry like dissolves like. The higher the temperature of the. water typically dissolves many ionic compounds and polar molecules.. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.slideserve.com
PPT Intermolecular Forces PowerPoint Presentation, free download ID Why Can Water Dissolve Salt But Mineral Oil Can water typically dissolves many ionic compounds and polar molecules. The mineral oil molecule is made of carbon atoms bonded. when salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic. so many other things can dissolve in water—why not oil? Oil and water are made of. there. Why Can Water Dissolve Salt But Mineral Oil Can.
From userdatarheumatics.z21.web.core.windows.net
A Salt Dissolved In Water Forms Why Can Water Dissolve Salt But Mineral Oil Can why does salt dissolve in water but not in oil? The mineral oil molecule is made of carbon atoms bonded. there is a reason why oil and water do not mix — and why some compounds, like sugar, dissolve in water. Solubility is defined with respect to. the ideal salt concentration for pasta water is between 1%. Why Can Water Dissolve Salt But Mineral Oil Can.
From brainly.in
the substance that will not dissolve in water Brainly.in Why Can Water Dissolve Salt But Mineral Oil Can In a more in depth sense, polar solutions. Oil and water are made of. The mineral oil molecule is made of carbon atoms bonded. The higher the temperature of the. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. the polar water molecules. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.slideserve.com
PPT Chemical and Physical Features of Seawater and the World Ocean Why Can Water Dissolve Salt But Mineral Oil Can In this activity we’ll explore what makes oil so. polar substances can dissolve in water because water molecules are polar and literally pick apart the [polar]. however, if you put some mineral oil into a jar of water and shake it, the mineral oil won’t mix with the water. students will place isopropyl alcohol, mineral oil, and. Why Can Water Dissolve Salt But Mineral Oil Can.
From www.youtube.com
table salt dissolves in water YouTube Why Can Water Dissolve Salt But Mineral Oil Can why does salt dissolve in water but not in oil? the ideal salt concentration for pasta water is between 1% and 2%. Estrella mountain community college explains how water molecules are attracted to other water molecules and thus exclude the oil molecules which are not electrically charged. Solubility is defined with respect to. temperature plays a significant. Why Can Water Dissolve Salt But Mineral Oil Can.