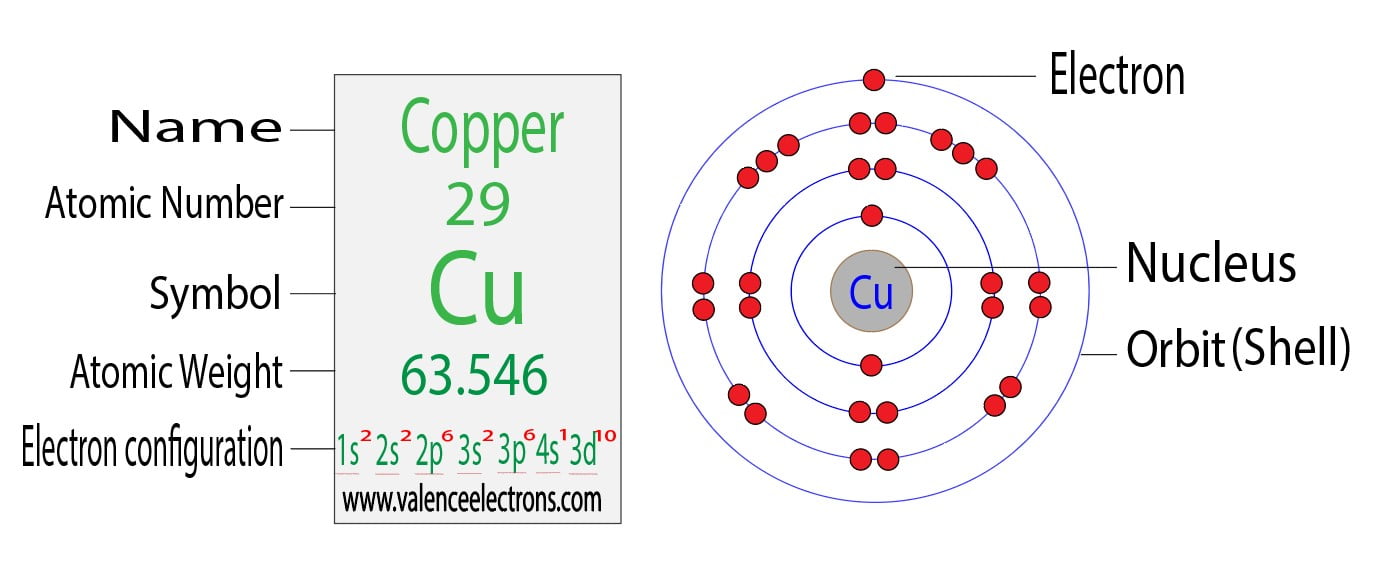
Copper Electron Valence Configuration . The full electron configuration for copper is [ar] 3d10 4s1. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. Full electron configuration for copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. it draws the dots around the symbol of copper to show up the valence electrons. let us use this smart electron configuration calculator to determine the electron configuration of copper: how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. What is the electron configuration of copper. The numbers of dots remain equal to the numbers of valence electrons of atoms. electron configuration of copper. there are two valence electrons in the copper.
from valenceelectrons.com
The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. let us use this smart electron configuration calculator to determine the electron configuration of copper: Lewis dot diagram can help you in the proper analysis of copper valence electrons. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. there are two valence electrons in the copper. What is the electron configuration of copper. how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. The full electron configuration for copper is [ar] 3d10 4s1. it draws the dots around the symbol of copper to show up the valence electrons. Full electron configuration for copper.
Electron Configuration for Copper (Cu, Cu+, Cu2+)
Copper Electron Valence Configuration What is the electron configuration of copper. What is the electron configuration of copper. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. The numbers of dots remain equal to the numbers of valence electrons of atoms. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. let us use this smart electron configuration calculator to determine the electron configuration of copper: electron configuration of copper. Full electron configuration for copper. The full electron configuration for copper is [ar] 3d10 4s1. how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. there are two valence electrons in the copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. it draws the dots around the symbol of copper to show up the valence electrons.
From www.alamy.com
Copper (Cu). Diagram of the valence orbitals of an atom of copper64 Copper Electron Valence Configuration there are two valence electrons in the copper. it draws the dots around the symbol of copper to show up the valence electrons. Lewis dot diagram can help you in the proper analysis of copper valence electrons. Full electron configuration for copper. how to write the electron configuration for copper (cu, cu+, and cu2+) in order to. Copper Electron Valence Configuration.
From valenceelectrons.com
How to Find the Valence Electrons for Copper (Cu)? Copper Electron Valence Configuration there are two valence electrons in the copper. it draws the dots around the symbol of copper to show up the valence electrons. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. Full electron configuration for copper. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of. Copper Electron Valence Configuration.
From valenceelectrons.com
Electron Configuration for Copper (Cu, Cu+, Cu2+) Copper Electron Valence Configuration Full electron configuration for copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. it draws the dots around the symbol of copper to show up the valence electrons. let us use this smart electron configuration calculator to determine the electron configuration of copper: there are two valence electrons in the copper. What is. Copper Electron Valence Configuration.
From www.alamy.com
Copper (Cu). Diagram of the nuclear composition, electron configuration Copper Electron Valence Configuration The full electron configuration for copper is [ar] 3d10 4s1. it draws the dots around the symbol of copper to show up the valence electrons. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. Lewis dot diagram can help you in the proper analysis of copper valence electrons. The numbers of dots remain equal to. Copper Electron Valence Configuration.
From www.coursehero.com
[Solved] 15. Fill in the electron configuration diagram for the copper Copper Electron Valence Configuration the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. What is the electron configuration of copper. it draws the dots around the symbol of copper to show up the valence electrons. Full electron configuration for copper. how to write the electron configuration. Copper Electron Valence Configuration.
From www.mooramo.com
Ions of Transition Elements Mooramo Copper Electron Valence Configuration What is the electron configuration of copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. there are two valence electrons in the copper. in this article, we will explore. Copper Electron Valence Configuration.
From general.chemistrysteps.com
Orbital Diagrams Chemistry Steps Copper Electron Valence Configuration in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. there are two valence electrons in the copper. Full electron configuration for copper. The numbers of dots remain equal to the numbers of valence electrons of atoms.. Copper Electron Valence Configuration.
From www.youtube.com
How to Find the Valence Electrons for Copper (Cu) YouTube Copper Electron Valence Configuration electron configuration of copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. there are two valence electrons in the copper. What is the electron configuration of copper. The full electron configuration for copper is [ar] 3d10 4s1. the electron distribution in copper spans across the k, l, m, and n shells,. Copper Electron Valence Configuration.
From www.alamy.com
Symbol and electron diagram for Copper Stock Vector Image & Art Alamy Copper Electron Valence Configuration What is the electron configuration of copper. let us use this smart electron configuration calculator to determine the electron configuration of copper: electron configuration of copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. The numbers of dots remain equal to the numbers of valence electrons of atoms. in this article,. Copper Electron Valence Configuration.
From www.vectorstock.com
Symbol and electron diagram for copper Royalty Free Vector Copper Electron Valence Configuration it draws the dots around the symbol of copper to show up the valence electrons. The numbers of dots remain equal to the numbers of valence electrons of atoms. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons. Copper Electron Valence Configuration.
From www.youtube.com
Electron Configuration for Cu, Cu+, and Cu2+ (Copper and Copper Ions Copper Electron Valence Configuration The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. What is the electron configuration of copper. Full electron configuration for copper. the electron distribution in copper spans across the k, l, m, and. Copper Electron Valence Configuration.
From organicful44.blogspot.com
copper orbital diagram Organicful Copper Electron Valence Configuration it draws the dots around the symbol of copper to show up the valence electrons. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. Full electron configuration for copper. there are two valence electrons in the copper. let us use this. Copper Electron Valence Configuration.
From imgtwitter3.blogspot.com
Electron Configuration Of Copper In Ground State Farm house customer Copper Electron Valence Configuration The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. electron configuration of copper. The full electron configuration for copper is [ar] 3d10 4s1. Lewis dot diagram can help you in the. Copper Electron Valence Configuration.
From www.youtube.com
Copper Electron Configuration Organic Chemistry Examples YouTube Copper Electron Valence Configuration in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. Full electron configuration for copper. it draws the dots around the symbol of copper to show up the valence electrons. What is the electron configuration of copper. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu.. Copper Electron Valence Configuration.
From periodictable.me
How To Find A Electron Configuration For Copper Dynamic Periodic Copper Electron Valence Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. What is the electron configuration of copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. The numbers of dots remain equal to the numbers of valence electrons of atoms. how to write the electron configuration for copper (cu, cu+, and. Copper Electron Valence Configuration.
From www.toppr.com
The electronic configuration of copper (29Cu) is. Copper Electron Valence Configuration let us use this smart electron configuration calculator to determine the electron configuration of copper: electron configuration of copper. What is the electron configuration of copper. The numbers of dots remain equal to the numbers of valence electrons of atoms. Full electron configuration for copper. The full electron configuration for copper is [ar] 3d10 4s1. how to. Copper Electron Valence Configuration.
From www.sciencephoto.com
Copper, atomic structure Stock Image C013/1552 Science Photo Library Copper Electron Valence Configuration in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. Full electron configuration for copper. What is the electron configuration of copper. electron configuration of copper. let us use this smart electron configuration calculator to determine the electron configuration of copper: there are two valence electrons in the. Copper Electron Valence Configuration.
From www.dreamstime.com
Electron of the Element Copper Stock Vector Illustration of Copper Electron Valence Configuration there are two valence electrons in the copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. it draws the dots around the symbol of copper to show up the valence electrons. electron configuration of copper. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of. Copper Electron Valence Configuration.
From www.sciencephoto.com
Copper, atomic structure Stock Image C018/3710 Science Photo Library Copper Electron Valence Configuration The full electron configuration for copper is [ar] 3d10 4s1. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. there are two valence electrons in the copper. electron configuration of copper. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and. Copper Electron Valence Configuration.
From www.alamy.com
Copper (Cu). Diagram of the nuclear composition, electron configuration Copper Electron Valence Configuration how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. Lewis dot diagram can help you in the proper analysis of copper valence electrons. The numbers of dots remain equal to the numbers of valence. Copper Electron Valence Configuration.
From www.alamy.com
3d render of atom structure of copper isolated over white background Copper Electron Valence Configuration let us use this smart electron configuration calculator to determine the electron configuration of copper: The numbers of dots remain equal to the numbers of valence electrons of atoms. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. The electron configuration of copper. Copper Electron Valence Configuration.
From aliceandallthatjazz.blogspot.com
Electron Configuration Of Copper 1+ worksheet Copper Electron Valence Configuration The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. it draws the dots around the symbol of copper to show up the valence electrons. electron configuration of copper. let us use this smart electron configuration calculator to determine the electron configuration of copper: the electron distribution in copper spans across the k, l,. Copper Electron Valence Configuration.
From valenceelectrons.com
How Many Valence Electrons Does Copper (Cu) Have? Copper Electron Valence Configuration electron configuration of copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. The numbers of dots remain equal to the numbers of valence electrons of atoms. let us use this smart electron configuration calculator to determine the electron configuration of copper: there are two valence electrons in the copper. in this article,. Copper Electron Valence Configuration.
From www.youtube.com
ELECTRONIC CONFIGURATION OF COPPER ATOM and STABILITY YouTube Copper Electron Valence Configuration it draws the dots around the symbol of copper to show up the valence electrons. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. What is the electron configuration of copper. let us use this smart electron configuration calculator to determine the. Copper Electron Valence Configuration.
From www.alamy.com
Copper (Cu). Diagram of the nuclear composition and electron Copper Electron Valence Configuration Lewis dot diagram can help you in the proper analysis of copper valence electrons. What is the electron configuration of copper. Full electron configuration for copper. electron configuration of copper. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. The full electron configuration for copper is [ar] 3d10 4s1. the electron distribution in copper. Copper Electron Valence Configuration.
From wiringguidefrosts.z19.web.core.windows.net
Copper Electron Configuration Diagram Copper Electron Valence Configuration Full electron configuration for copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. let us use this smart electron configuration calculator to determine the electron configuration of copper: What is the electron configuration of copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. the electron distribution in copper. Copper Electron Valence Configuration.
From www.youtube.com
How to Write the Atomic Orbital Diagram for Copper (Cu) YouTube Copper Electron Valence Configuration there are two valence electrons in the copper. it draws the dots around the symbol of copper to show up the valence electrons. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. The full electron configuration for copper is [ar] 3d10 4s1. the electron distribution in copper spans across the k, l, m,. Copper Electron Valence Configuration.
From valenceelectrons.com
Electron Configuration for Copper (Cu, Cu+, Cu2+) Copper Electron Valence Configuration The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. Full electron configuration for copper. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. The numbers of dots remain equal to the numbers of valence electrons of atoms. 1s2 2s2 2p6 3s2. Copper Electron Valence Configuration.
From elchoroukhost.net
Copper Periodic Table Valence Electrons Elcho Table Copper Electron Valence Configuration Lewis dot diagram can help you in the proper analysis of copper valence electrons. how to write the electron configuration for copper (cu, cu+, and cu2+) in order to write the copper electron configuration we first. What is the electron configuration of copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. electron configuration of. Copper Electron Valence Configuration.
From www.earthdate.org
Copper’s Superpower EarthDate Copper Electron Valence Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. it draws the dots around the symbol of copper to show up the valence electrons. electron configuration of copper. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. Full. Copper Electron Valence Configuration.
From electraschematics.com
Understanding the Electron Configuration Diagram for Copper Copper Electron Valence Configuration The full electron configuration for copper is [ar] 3d10 4s1. Full electron configuration for copper. Lewis dot diagram can help you in the proper analysis of copper valence electrons. there are two valence electrons in the copper. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. The numbers of. Copper Electron Valence Configuration.
From www.electrical4u.com
Understanding Valence Electrons and Electrical Conductivity Electrical4U Copper Electron Valence Configuration let us use this smart electron configuration calculator to determine the electron configuration of copper: electron configuration of copper. The numbers of dots remain equal to the numbers of valence electrons of atoms. there are two valence electrons in the copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. it draws the. Copper Electron Valence Configuration.
From valenceelectrons.com
How to Find the Valence Electrons for Copper (Cu)? Copper Electron Valence Configuration let us use this smart electron configuration calculator to determine the electron configuration of copper: Full electron configuration for copper. in this article, we will explore the electron orbital arrangement, shell configuration, and valence electron distribution of copper. The full electron configuration for copper is [ar] 3d10 4s1. electron configuration of copper. The electron configuration of copper. Copper Electron Valence Configuration.
From valenceelectrons.com
Electron Configuration for Copper (Cu, Cu+, Cu2+) Copper Electron Valence Configuration let us use this smart electron configuration calculator to determine the electron configuration of copper: Full electron configuration for copper. What is the electron configuration of copper. there are two valence electrons in the copper. The electron configuration of copper can be represented as [ar] 3d¹⁰ 4s¹. electron configuration of copper. the electron distribution in copper. Copper Electron Valence Configuration.
From izayahmeowcrosby.blogspot.com
Electronic Configuration of Copper Copper Electron Valence Configuration electron configuration of copper. the electron distribution in copper spans across the k, l, m, and n shells, with valence electrons located in the 4s and 3d orbitals. 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is the electron configuration of cu. The numbers of dots remain equal to the numbers of valence electrons of atoms. Lewis dot diagram. Copper Electron Valence Configuration.