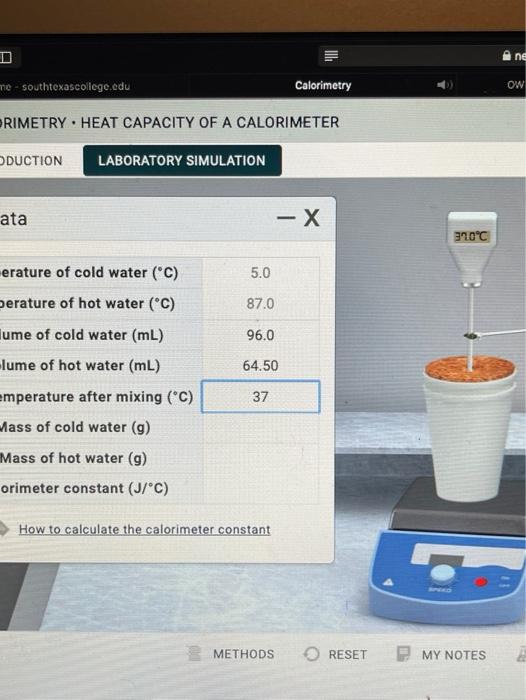
Heat Capacity Of A Calorimeter Lab Chegg . Heat always flows from high temperature to low temperature. The amount of heat absorbed or released (q) by the object. Heat capacity of a calorimeter. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Here’s the best way to solve it. Your solution’s ready to go! 100% (2 ratings) here’s how to approach this question. Use the equation to calculate the heat lost by the. Your solution’s ready to go! Sign up to see more!
from www.chegg.com
The amount of heat absorbed or released (q) by the object. Here’s the best way to solve it. 100% (2 ratings) here’s how to approach this question. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Your solution’s ready to go! Heat always flows from high temperature to low temperature. Sign up to see more! Your solution’s ready to go! Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c.
Solved connect Calorimetry TRY. HEAT
Heat Capacity Of A Calorimeter Lab Chegg Heat always flows from high temperature to low temperature. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). 100% (2 ratings) here’s how to approach this question. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Heat always flows from high temperature to low temperature. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Your solution’s ready to go! Here’s the best way to solve it. Use the equation to calculate the heat lost by the. The amount of heat absorbed or released (q) by the object. Heat capacity of a calorimeter. Your solution’s ready to go! Sign up to see more!
From www.slideserve.com
PPT Unit 13 Thermochemistry PowerPoint Presentation ID6591088 Heat Capacity Of A Calorimeter Lab Chegg Heat capacity of a calorimeter. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. 100% (2 ratings) here’s how to approach this question. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved CALORIMETRY HEAT CAPACITY OF A CALORIMETER Heat Capacity Of A Calorimeter Lab Chegg 100% (2 ratings) here’s how to approach this question. Sign up to see more! Your solution’s ready to go! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat always flows from high temperature to low temperature.. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved In the laboratory a "coffee cup" calorimeter, or Heat Capacity Of A Calorimeter Lab Chegg Use the equation to calculate the heat lost by the. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Your solution’s ready to go! Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Here’s the. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved To determine the heat capacity of a calorimeter, 50.0 Heat Capacity Of A Calorimeter Lab Chegg Use the equation to calculate the heat lost by the. Heat capacity of a calorimeter. 100% (2 ratings) here’s how to approach this question. Sign up to see more! Your solution’s ready to go! Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). The heat capacity (c). Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved HEAT CAPACITY OF A CALORIMETER Lab Data How to Heat Capacity Of A Calorimeter Lab Chegg Heat capacity of a calorimeter. The amount of heat absorbed or released (q) by the object. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Use the equation to calculate the heat lost by the. Your solution’s ready to go! Calculate the mass of cold water by multiplying. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved The indicator diagram for an ideal heat engine shown Heat Capacity Of A Calorimeter Lab Chegg The amount of heat absorbed or released (q) by the object. Sign up to see more! Heat capacity of a calorimeter. Use the equation to calculate the heat lost by the. Heat always flows from high temperature to low temperature. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved CALORIMETRY. HEAT CAPACITY OF A CALORIMETER Heat Capacity Of A Calorimeter Lab Chegg Your solution’s ready to go! Heat always flows from high temperature to low temperature. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat capacity is defined as the amount of heat needed to increase the temperature. Heat Capacity Of A Calorimeter Lab Chegg.
From ar.inspiredpencil.com
Coffee Cup Calorimeter Diagram Heat Capacity Of A Calorimeter Lab Chegg 100% (2 ratings) here’s how to approach this question. Use the equation to calculate the heat lost by the. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Here’s the best way to solve it. Sign up. Heat Capacity Of A Calorimeter Lab Chegg.
From janiyahabbgates.blogspot.com
Calorimetry Specific Heat Capacity of Metals Lab Report JaniyahabbGates Heat Capacity Of A Calorimeter Lab Chegg Your solution’s ready to go! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Sign up. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved Calculate Heat capacity of the calorimeter (J/ ºC) , Heat Capacity Of A Calorimeter Lab Chegg Sign up to see more! Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Your solution’s ready to go! Here’s the best way to solve it.. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved Combustion (bomb) calorimeter. In an experiment, a Heat Capacity Of A Calorimeter Lab Chegg Here’s the best way to solve it. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Sign up to see more! Your solution’s ready to go! The amount of heat absorbed or released (q) by the object. Use the equation to calculate the heat lost by the.. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved Experiment 25 Report Sheet Calorimetry Lab Sec Name Heat Capacity Of A Calorimeter Lab Chegg Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat always flows from high temperature to. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved CALORIMETRY • HEAT CAPACITY OF A CALORIMETER Heat Capacity Of A Calorimeter Lab Chegg 100% (2 ratings) here’s how to approach this question. The amount of heat absorbed or released (q) by the object. Your solution’s ready to go! Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Heat always flows from high temperature to low temperature. Heat capacity of a calorimeter.. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved Calculations I. Heat Capacity of the Calorimeter The Heat Capacity Of A Calorimeter Lab Chegg Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Sign up to see more! The amount of heat absorbed or released (q) by the object. Use the equation to calculate the heat lost by the. Here’s the best way to solve it. Heat capacity is defined as. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved CALORIMETRY • HEAT CAPACITY OF A CALORIMETER A 30.0 g Heat Capacity Of A Calorimeter Lab Chegg Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Your solution’s ready to go! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Here’s the. Heat Capacity Of A Calorimeter Lab Chegg.
From rachaelocean.blogspot.com
32+ calorimeter constant calculator RachaelOcean Heat Capacity Of A Calorimeter Lab Chegg Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Use the equation to calculate the heat lost by the. 100% (2 ratings) here’s how to approach this question. Here’s the best way to solve it. Your solution’s ready to go! Heat capacity of a calorimeter. The heat capacity. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved In the laboratory a "coffee cup" calorimeter, or Heat Capacity Of A Calorimeter Lab Chegg Use the equation to calculate the heat lost by the. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Here’s the best way to solve it. Sign up to see more! Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved When a solid dissolves in water, heat may be evolved Heat Capacity Of A Calorimeter Lab Chegg The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. The amount of heat absorbed or released (q) by the object. Heat capacity of a calorimeter. Here’s the best way to solve it. Your solution’s ready to go!. Heat Capacity Of A Calorimeter Lab Chegg.
From exodnulby.blob.core.windows.net
Lab Calorimetry And Specific Heat Summary at Barbara Bailey blog Heat Capacity Of A Calorimeter Lab Chegg Here’s the best way to solve it. Sign up to see more! Heat always flows from high temperature to low temperature. Use the equation to calculate the heat lost by the. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Your solution’s ready to go! Heat capacity. Heat Capacity Of A Calorimeter Lab Chegg.
From janiyahabbgates.blogspot.com
Calorimetry Specific Heat Capacity of Metals Lab Report JaniyahabbGates Heat Capacity Of A Calorimeter Lab Chegg Heat always flows from high temperature to low temperature. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Your solution’s ready to go! Heat capacity of a calorimeter. Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved CHEMISTRY DETERMINING HEAT CAPACITY OF A CALORIMETER Heat Capacity Of A Calorimeter Lab Chegg Sign up to see more! Your solution’s ready to go! The amount of heat absorbed or released (q) by the object. Use the equation to calculate the heat lost by the. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Heat always flows from high temperature to low. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved PART A HEAT CAPACITY OF THE CALORIMETER 1. Heat Capacity Of A Calorimeter Lab Chegg Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). Heat always flows from high temperature to low temperature. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Sign up to see more! 100% (2 ratings). Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved = EXP 11 CALORIMETRY. HEAT CAPACITY OF A Heat Capacity Of A Calorimeter Lab Chegg Your solution’s ready to go! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. 100% (2 ratings) here’s how to approach this question. The amount of heat absorbed or released (q) by the object. Use the equation. Heat Capacity Of A Calorimeter Lab Chegg.
From www.youtube.com
CH5 Q5 Calculating the Heat Capacity of a Calorimeter YouTube Heat Capacity Of A Calorimeter Lab Chegg Sign up to see more! 100% (2 ratings) here’s how to approach this question. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Your solution’s ready to go! Heat always flows from high temperature to low temperature.. Heat Capacity Of A Calorimeter Lab Chegg.
From www.scienceabc.com
Molar Heat Capacity Definition, Formula, Equation, Calculation Heat Capacity Of A Calorimeter Lab Chegg Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). 100% (2 ratings) here’s how to approach this question. Sign up to see more! Heat capacity of a calorimeter. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved A bomb calorimeter, or constant volume calorimeter, Heat Capacity Of A Calorimeter Lab Chegg Your solution’s ready to go! Your solution’s ready to go! Here’s the best way to solve it. Use the equation to calculate the heat lost by the. Heat capacity of a calorimeter. Sign up to see more! Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). 100%. Heat Capacity Of A Calorimeter Lab Chegg.
From www.pinterest.com
ConstantVolume Calorimetry for more precise work than the coffeecup Heat Capacity Of A Calorimeter Lab Chegg Sign up to see more! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat capacity of a calorimeter. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved I need help calculating calorimeter constant!! mass Heat Capacity Of A Calorimeter Lab Chegg The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. The amount of heat absorbed or released (q) by the object. Heat always flows from high temperature to low temperature. 100% (2 ratings) here’s how to approach this. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved In the laboratory a "coffee cup" calorimeter, or Heat Capacity Of A Calorimeter Lab Chegg Your solution’s ready to go! Heat capacity of a calorimeter. Here’s the best way to solve it. 100% (2 ratings) here’s how to approach this question. Your solution’s ready to go! Calculate the mass of cold water by multiplying its volume ( 100.0 ml) by the density of water ( 1 g/ml ). The heat capacity (c) of a body. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved What is the heat capacity of a calorimeter in JC if Heat Capacity Of A Calorimeter Lab Chegg Heat capacity of a calorimeter. Sign up to see more! Here’s the best way to solve it. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat always flows from high temperature to low temperature. Your solution’s. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved In The Laboratory A "coffee Cup" Calorimeter, Or C... Heat Capacity Of A Calorimeter Lab Chegg 100% (2 ratings) here’s how to approach this question. The amount of heat absorbed or released (q) by the object. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Your solution’s ready to go! Heat capacity of a calorimeter. The heat capacity (c) of a body of matter. Heat Capacity Of A Calorimeter Lab Chegg.
From www.youtube.com
CHEMISTRY 101 Calculating Heat Capacity of a Bomb Calorimeter YouTube Heat Capacity Of A Calorimeter Lab Chegg Use the equation to calculate the heat lost by the. The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree celsius (or equivalently,. Heat capacity of a calorimeter. Heat capacity is defined as the amount of heat needed to increase the. Heat Capacity Of A Calorimeter Lab Chegg.
From ar.inspiredpencil.com
Specific Heat Lab Calorimeter Heat Capacity Of A Calorimeter Lab Chegg Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Heat capacity of a calorimeter. Your solution’s ready to go! The heat capacity (c) of a body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (δt) of 1 degree. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved In the laboratory a "coffee cup" calorimeter, or Heat Capacity Of A Calorimeter Lab Chegg 100% (2 ratings) here’s how to approach this question. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Your solution’s ready to go! The amount of heat absorbed or released (q) by the object. Use the equation to calculate the heat lost by the. Calculate the mass of. Heat Capacity Of A Calorimeter Lab Chegg.
From www.chegg.com
Solved connect Calorimetry TRY. HEAT Heat Capacity Of A Calorimeter Lab Chegg Here’s the best way to solve it. Heat capacity is defined as the amount of heat needed to increase the temperature of the entire calorimeter by 1 c. Heat capacity of a calorimeter. The amount of heat absorbed or released (q) by the object. Sign up to see more! Your solution’s ready to go! Calculate the mass of cold water. Heat Capacity Of A Calorimeter Lab Chegg.