Back Titration Analysis Of A Commercial Antacid . The reactions above are reversible, which. Perform a titration with naoh to figure out the amount of excess hcl. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. Back titration is different from the direct. A technique called back titration is used to determine how much stomach acid is neutralized. Calculate how much acid reacted with the antacid.
from www.studocu.com
Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid is neutralized. Calculate how much acid reacted with the antacid. Compare the active ingredients in two different. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Back titration is different from the direct. The reactions above are reversible, which. Then, from this, we can calculate how much acid reacted with the.
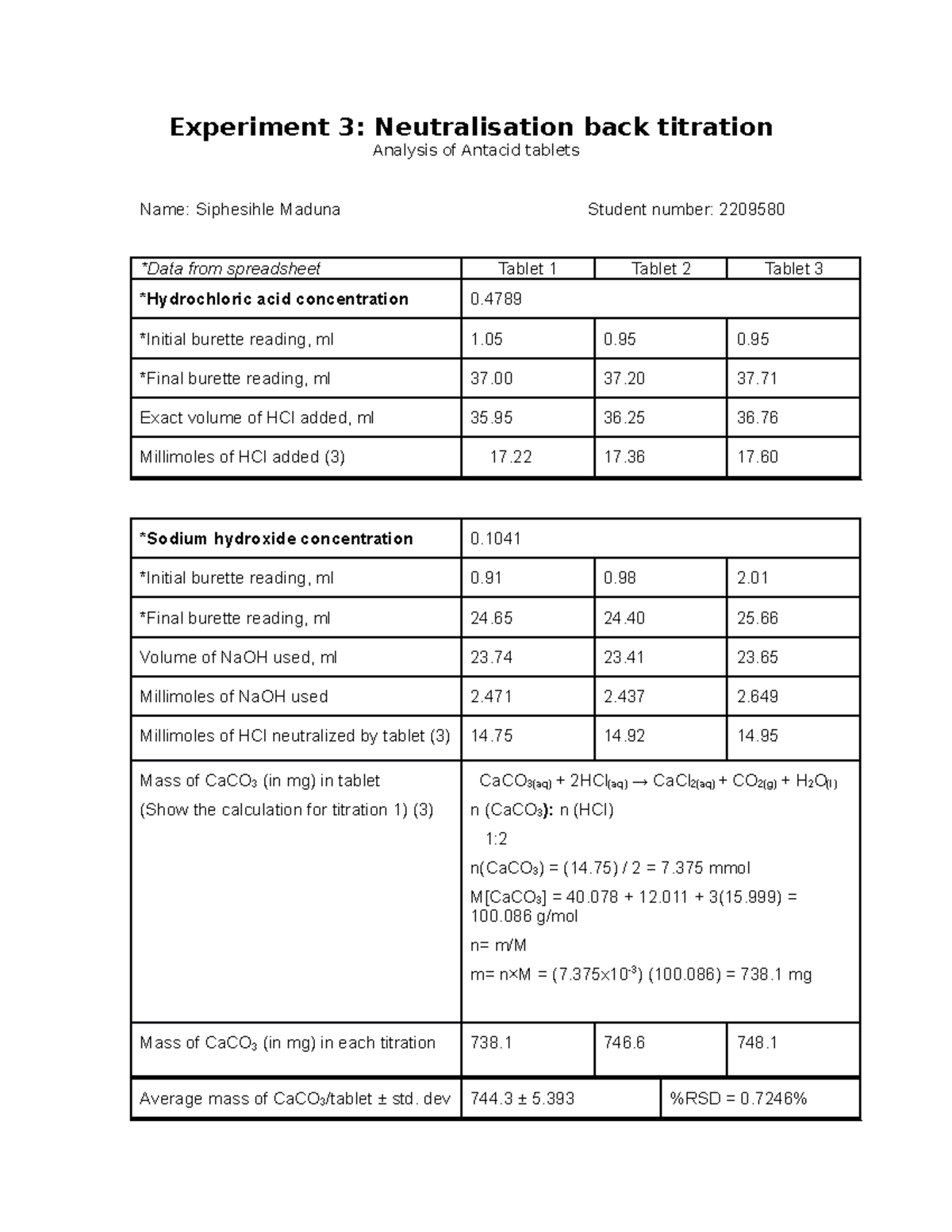
Neutralisation Back Titration Analysis of Antacid Tablets CHEM2001
Back Titration Analysis Of A Commercial Antacid The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. The reactions above are reversible, which. Calculate how much acid reacted with the antacid. A technique called back titration is used to determine how much stomach acid is neutralized. Back titration is different from the direct. Perform a titration with naoh to figure out the amount of excess hcl. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration.
From studylib.net
Acid Neutralizing Capacity of an Antacid Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. A technique called back titration is used to determine how much stomach acid is neutralized. Calculate how much acid reacted with the antacid. The reactions above are reversible, which. Back titration is different from the direct. Perform a titration with naoh to figure out the amount of excess. Back Titration Analysis Of A Commercial Antacid.
From www.shalom-education.com
Titrations Shalom Education Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the. A technique called back titration is used to determine how much stomach acid is neutralized. The reactions above are reversible, which. The number of moles of acid that. Back Titration Analysis Of A Commercial Antacid.
From studylib.net
Back Titration Analysis of a Commercial Antacid Back Titration Analysis Of A Commercial Antacid Calculate how much acid reacted with the antacid. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the. Back titration is different from the direct. Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid. Back Titration Analysis Of A Commercial Antacid.
From www.slideserve.com
PPT Antacid Analysis Lab PowerPoint Presentation, free download ID Back Titration Analysis Of A Commercial Antacid The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Compare the active ingredients in two different. Calculate how much acid reacted with the antacid. Then, from this, we can calculate how much acid reacted with the. A technique called back titration is used to determine. Back Titration Analysis Of A Commercial Antacid.
From www.chegg.com
Solved Titration of a Commercial Antacid Objective To Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. Calculate how much acid reacted with the antacid. A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Back. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
Antacid Titration Experimental Set Up YouTube Back Titration Analysis Of A Commercial Antacid Compare the active ingredients in two different. Calculate how much acid reacted with the antacid. The reactions above are reversible, which. A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration.. Back Titration Analysis Of A Commercial Antacid.
From www.chemedx.org
How do antacids work? Titration of an Antacid Chemical Education Xchange Back Titration Analysis Of A Commercial Antacid Compare the active ingredients in two different. Back titration is different from the direct. Then, from this, we can calculate how much acid reacted with the. Perform a titration with naoh to figure out the amount of excess hcl. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined. Back Titration Analysis Of A Commercial Antacid.
From studylib.net
Experiment 7 Titration of an Antacid Back Titration Analysis Of A Commercial Antacid Compare the active ingredients in two different. Back titration is different from the direct. Then, from this, we can calculate how much acid reacted with the. A technique called back titration is used to determine how much stomach acid is neutralized. Perform a titration with naoh to figure out the amount of excess hcl. The reactions above are reversible, which.. Back Titration Analysis Of A Commercial Antacid.
From www.chegg.com
Solved Antacid Analysis and TitrationLab Report Assistant Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid is neutralized. Compare the active ingredients in two different. Back titration is different from the direct. Calculate how much acid reacted with the antacid. Then, from this, we can calculate how much acid reacted. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
Neutralisation Back Titration Analysis of Antacid Tablets CHEM2001 Back Titration Analysis Of A Commercial Antacid Back titration is different from the direct. The reactions above are reversible, which. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the. Perform a titration with naoh to figure out the amount of excess hcl. Calculate how much acid reacted with the antacid. A technique called back titration is used. Back Titration Analysis Of A Commercial Antacid.
From www.coursehero.com
[Solved] Analytical chemistry ACIDBASED TITRATION II (BACK TITRATION Back Titration Analysis Of A Commercial Antacid A technique called back titration is used to determine how much stomach acid is neutralized. Back titration is different from the direct. The reactions above are reversible, which. Perform a titration with naoh to figure out the amount of excess hcl. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the.. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
Back Titration Example YouTube Back Titration Analysis Of A Commercial Antacid The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Back titration is different from the direct. The reactions above are reversible, which. Then, from this, we can calculate how much acid reacted with the. Perform a titration with naoh to figure out the amount of. Back Titration Analysis Of A Commercial Antacid.
From www.studypool.com
SOLUTION Drug antacid effectiveness analysis to determine the Back Titration Analysis Of A Commercial Antacid Calculate how much acid reacted with the antacid. The reactions above are reversible, which. Perform a titration with naoh to figure out the amount of excess hcl. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. A technique called back titration is used to determine how much stomach acid is. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
TUMS LAB Calculations Review YouTube Back Titration Analysis Of A Commercial Antacid The reactions above are reversible, which. Back titration is different from the direct. A technique called back titration is used to determine how much stomach acid is neutralized. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the. The number of moles of acid that can be neutralized by a single. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
Experiment Back Titration (antacid) YouTube Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. The reactions above are reversible, which. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that. Back Titration Analysis Of A Commercial Antacid.
From dokumen.tips
(DOCX) AcidBase Titrations Analysis of Antacid Tablets DOKUMEN.TIPS Back Titration Analysis Of A Commercial Antacid Compare the active ingredients in two different. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Back titration is different from the direct. The reactions above are reversible, which. Then, from this, we can calculate how much acid reacted with the. Calculate how much acid. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
7. antacid effectiveness by back titration YouTube Back Titration Analysis Of A Commercial Antacid Calculate how much acid reacted with the antacid. A technique called back titration is used to determine how much stomach acid is neutralized. Perform a titration with naoh to figure out the amount of excess hcl. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. The number of moles of. Back Titration Analysis Of A Commercial Antacid.
From studylib.net
Lab Acid Base Titration of an Antacid Back Titration Analysis Of A Commercial Antacid A technique called back titration is used to determine how much stomach acid is neutralized. Back titration is different from the direct. Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. Calculate how much acid reacted with the antacid. Perform a titration with naoh to figure out the amount of. Back Titration Analysis Of A Commercial Antacid.
From www.studypool.com
SOLUTION Drug antacid effectiveness analysis to determine the Back Titration Analysis Of A Commercial Antacid Back titration is different from the direct. Then, from this, we can calculate how much acid reacted with the. A technique called back titration is used to determine how much stomach acid is neutralized. Compare the active ingredients in two different. The reactions above are reversible, which. The number of moles of acid that can be neutralized by a single. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
BackTitration F24 Lab for back titration BackTitration of a Back Titration Analysis Of A Commercial Antacid The reactions above are reversible, which. Compare the active ingredients in two different. A technique called back titration is used to determine how much stomach acid is neutralized. Back titration is different from the direct. Then, from this, we can calculate how much acid reacted with the. Calculate how much acid reacted with the antacid. Perform a titration with naoh. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
Complexometric Titration Pharmaceutical Analysis Studocu Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. The reactions above are reversible, which. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Compare the active ingredients in two different. Calculate how much acid reacted with the antacid. A technique. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
A backtitration lab report Antacid Analysis A BackTitration I Back Titration Analysis Of A Commercial Antacid The reactions above are reversible, which. A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Perform a titration with naoh to figure out the amount of excess hcl. Calculate how. Back Titration Analysis Of A Commercial Antacid.
From www.studypool.com
SOLUTION Drug antacid effectiveness analysis to determine the Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid is neutralized. Then, from this, we can calculate how much acid reacted with the. Calculate how much acid reacted with the antacid. The number of moles of acid that can be neutralized by a. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
BackTitration F22 Copy BackTitration of a Commercial Antacid Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. Compare the active ingredients in two different. Back titration is different from the direct. The reactions above are reversible, which. Perform a titration with naoh to figure out the amount of excess hcl. Calculate how much acid reacted with the antacid. A technique called back titration is used. Back Titration Analysis Of A Commercial Antacid.
From www.behsscience.com
Untitled Document Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. Back titration is different from the direct. Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid is neutralized. The reactions above are reversible, which. The number of moles of acid that. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
Analysis of Antacid through back Titration Experiment Analysis of Back Titration Analysis Of A Commercial Antacid The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Calculate how much acid reacted with the antacid. A technique called back titration is used to determine how much stomach acid is neutralized. Back titration is different from the direct. Compare the active ingredients in two. Back Titration Analysis Of A Commercial Antacid.
From www.chegg.com
Solved Antacid Lab Part 1 A Determination of the Amount of Back Titration Analysis Of A Commercial Antacid Calculate how much acid reacted with the antacid. Perform a titration with naoh to figure out the amount of excess hcl. The reactions above are reversible, which. Then, from this, we can calculate how much acid reacted with the. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined. Back Titration Analysis Of A Commercial Antacid.
From mavink.com
Titration Analysis Back Titration Analysis Of A Commercial Antacid The reactions above are reversible, which. Perform a titration with naoh to figure out the amount of excess hcl. Then, from this, we can calculate how much acid reacted with the. Back titration is different from the direct. A technique called back titration is used to determine how much stomach acid is neutralized. Calculate how much acid reacted with the. Back Titration Analysis Of A Commercial Antacid.
From www.chegg.com
Exercise 1 Back Titration of Antacid Neutralization Back Titration Analysis Of A Commercial Antacid Calculate how much acid reacted with the antacid. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. The reactions above are reversible, which. Compare the active ingredients in two different. Back titration is different from the direct. Perform a titration with naoh to figure out. Back Titration Analysis Of A Commercial Antacid.
From www.studypool.com
SOLUTION Drug antacid effectiveness analysis to determine the Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Perform a titration with naoh to figure out the amount of excess hcl. Calculate how much acid reacted with the antacid. Compare the active. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
Experiment 11 Titration Commercial Antacid Experiment 11 Titration Back Titration Analysis Of A Commercial Antacid A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. Calculate how much acid reacted with the antacid. Compare the active ingredients in two different. Then, from this, we can calculate. Back Titration Analysis Of A Commercial Antacid.
From studylib.net
Experiment 11 Titration of a Commercial Antacid Back Titration Analysis Of A Commercial Antacid Back titration is different from the direct. Compare the active ingredients in two different. Perform a titration with naoh to figure out the amount of excess hcl. A technique called back titration is used to determine how much stomach acid is neutralized. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid. Back Titration Analysis Of A Commercial Antacid.
From www.slideserve.com
PPT CHAPTER 5 Volumetric Analysis PowerPoint Presentation, free Back Titration Analysis Of A Commercial Antacid Compare the active ingredients in two different. The reactions above are reversible, which. The number of moles of acid that can be neutralized by a single tablet of a commercial antacid will be determined by back titration. A technique called back titration is used to determine how much stomach acid is neutralized. Then, from this, we can calculate how much. Back Titration Analysis Of A Commercial Antacid.
From www.youtube.com
Determination of acid neutralising power of a commercial antacid tablet Back Titration Analysis Of A Commercial Antacid Then, from this, we can calculate how much acid reacted with the. Back titration is different from the direct. A technique called back titration is used to determine how much stomach acid is neutralized. The reactions above are reversible, which. Compare the active ingredients in two different. The number of moles of acid that can be neutralized by a single. Back Titration Analysis Of A Commercial Antacid.
From www.studocu.com
Lab Report Analysis of an Antacid Analysis of an Antacid By Jonathan Back Titration Analysis Of A Commercial Antacid Perform a titration with naoh to figure out the amount of excess hcl. Back titration is different from the direct. A technique called back titration is used to determine how much stomach acid is neutralized. The reactions above are reversible, which. Compare the active ingredients in two different. Then, from this, we can calculate how much acid reacted with the.. Back Titration Analysis Of A Commercial Antacid.