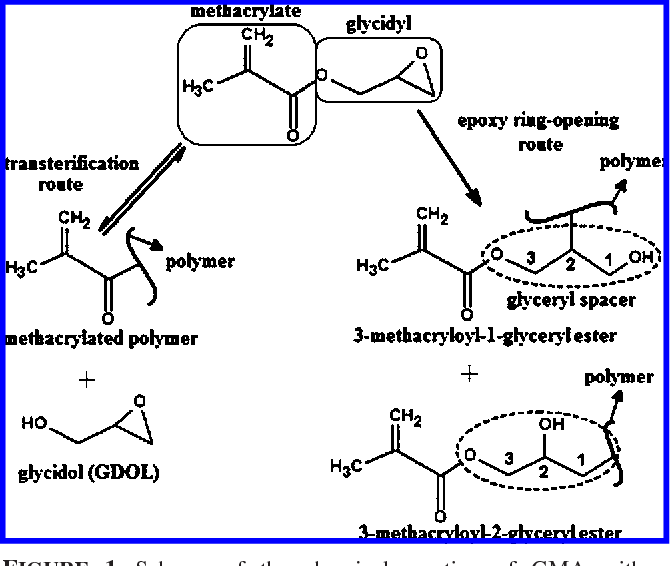
Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid . The polymers were modified through epoxy functional groups in two steps: The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with.
from www.semanticscholar.org
The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour.
Reaction of glycidyl methacrylate at the hydroxyl and carboxylic groups
Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour.
From www.researchgate.net
Fig. S1. 1 H NMR spectrum for for... Download Scientific Diagram Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.mdpi.com
IJMS Free FullText Polymorphism in SelfAssembled Structures of 9 Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Figure 4 from Reaction of glycidyl methacrylate at the hydroxyl and Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Figure 9 from Reaction of glycidyl methacrylate at the hydroxyl and Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From sielc.com
9Anthracenecarboxylic acid SIELC Technologies Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.frontiersin.org
Frontiers Photocrosslinkable natural polymers in tissue engineering Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From sielc.com
Glycidyl methacrylate SIELC Technologies Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(a) The structures of 9methyl anthracene (9MA), 9anthracene Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Structures of (a) anthracene and (b)(d) the three isomers of Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From pubs.acs.org
Reaction of Glycidyl Methacrylate at the Hydroxyl and Carboxylic Groups Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. The polymers were modified through epoxy functional groups in. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Crystallographic and structural refinement parameters of 9anthracene Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From testpubschina.acs.org
Crystal Structure of the 9AnthraceneCarboxylic Acid Photochemical Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.guidechem.com
Anthracene9carboxylic acid 723626 wiki Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.chemicalbook.com
Anthracene9carboxylic acid(723626) IR2 spectrum Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(a) The structures of 9methyl anthracene (9MA), 9anthracene Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From vdocuments.mx
Modification of copolymers using nucleophilic reactions between Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(PDF) Modification of copolymers using nucleophilic reactions between Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From alchetron.com
Glycidyl methacrylate Alchetron, The Free Social Encyclopedia Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. FLexibility of these functionalisations, coupled with the commercial availability. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.mdpi.com
Crystals Free FullText Mechanochemical Syntheses of Isostructural Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Scheme1 Polymerization of glycidyl methacrylate with succinic Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Reaction of glycidyl methacrylate at the hydroxyl and carboxylic groups Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Reaction of glycidyl methacrylate at the hydroxyl and carboxylic groups Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.guidechem.com
Anthracene9carboxylic acid 723626 wiki Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(a) Structures of 9methyl anthracene (9MA), 9anthracene carboxylic Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Figure 1 from Reaction of glycidyl methacrylate at the hydroxyl and Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(PDF) Modification of copolymers using nucleophilic reactions between Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
(a) Structures of 9methyl anthracene (9MA), 9anthracene carboxylic Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.fishersci.fi
Glycidyl methacrylate, 97, stabilized, ACROS Organics™ 100g, Glass Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Figure 7 from Reaction of glycidyl methacrylate at the hydroxyl and Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Scheme 1. HNC/ONC glycidyl methacrylate (GMA) grafting. Download Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The polymers were modified through epoxy functional groups in two steps: FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Scheme 2 Illustration of side reaction possible between the epoxy Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The polymers were modified through epoxy functional groups in two steps: Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From eureka.patsnap.com
Synthesis method of 9anthracene carboxylic acid Eureka Patsnap Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.researchgate.net
Scheme 1. HNC/ONC glycidyl methacrylate (GMA) grafting. Download Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid The present paper reports the synthesis, spectroscopic and thermal characterization of glycidyl methacrylate. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. The polymers were modified through epoxy functional groups in. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Figure 6 from Reaction of glycidyl methacrylate at the hydroxyl and Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.
From www.semanticscholar.org
Reaction of glycidyl methacrylate at the hydroxyl and carboxylic groups Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with. FLexibility of these functionalisations, coupled with the commercial availability of glycidyl methacrylate monomer, its controlled polymerisation behaviour. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (gma) polymers with nucleophilic reagent. The polymers were modified through. Glycidyl Methacrylate And 9-Anthracene Carboxylic Acid.