Standard Cell Potential Of Zinc And Lead . The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. Describe and relate the definitions of electrode and cell potentials. Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). “transfer” of electrons requires both a donor and. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Interpret electrode potentials in terms of relative oxidant and reductant.
from schoolbag.info
Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Describe and relate the definitions of electrode and cell potentials. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. Interpret electrode potentials in terms of relative oxidant and reductant. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell).
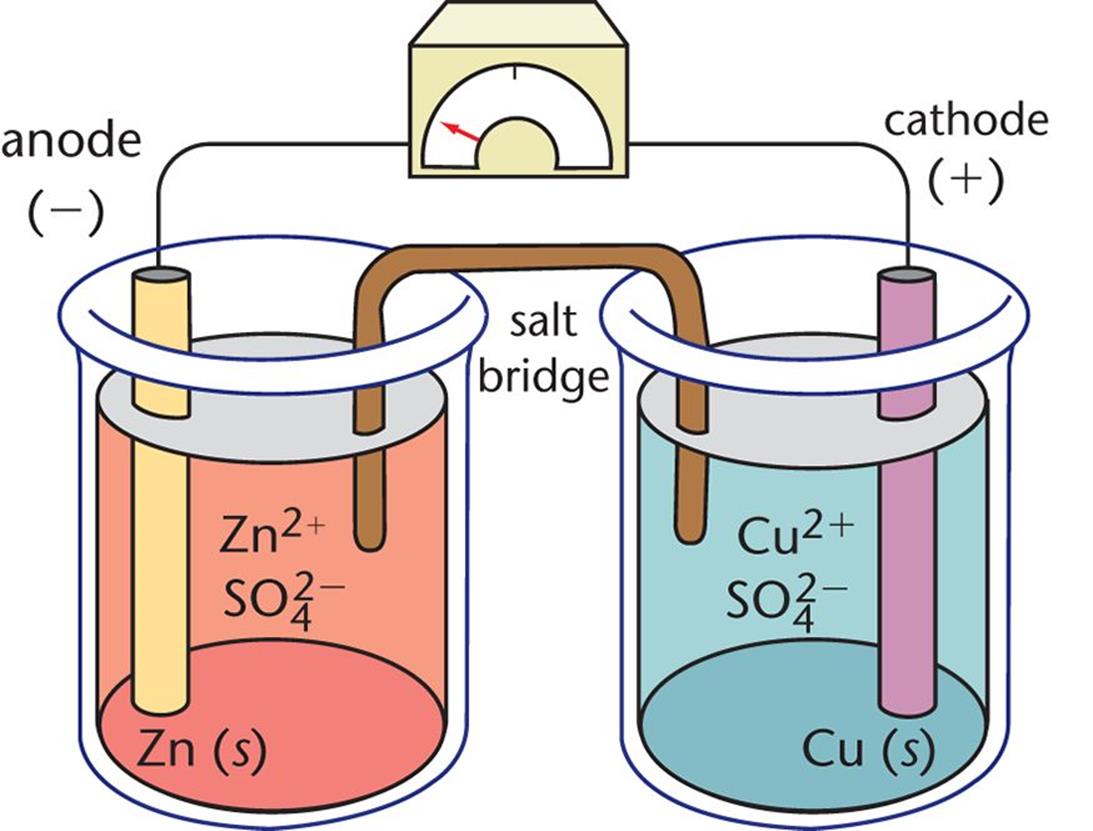
Figure 12.1. Daniell Cell In this galvanic cell, zinc is the anode and
Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. “transfer” of electrons requires both a donor and. Describe and relate the definitions of electrode and cell potentials. Interpret electrode potentials in terms of relative oxidant and reductant. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell).
From www.numerade.com
SOLVEDkalculate the standard cell potential of the following cell at Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Describe and relate the definitions of electrode and cell potentials. Only the difference between the potentials of two electrodes can be measured. “transfer” of electrons requires both a donor. Standard Cell Potential Of Zinc And Lead.
From free-images.com
Free Images standard electrode potential zinc Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Describe and relate the definitions of electrode and cell potentials. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Standard Cell Potential Of Zinc And Lead.
From schoolbag.info
Figure 12.1. Daniell Cell In this galvanic cell, zinc is the anode and Standard Cell Potential Of Zinc And Lead “transfer” of electrons requires both a donor and. Interpret electrode potentials in terms of relative oxidant and reductant. Describe and relate the definitions of electrode and cell potentials. Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Chapter 17 Electrochemistry PowerPoint Presentation, free Standard Cell Potential Of Zinc And Lead Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e°. Standard Cell Potential Of Zinc And Lead.
From saylordotorg.github.io
Standard Potentials Standard Cell Potential Of Zinc And Lead Interpret electrode potentials in terms of relative oxidant and reductant. “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Only the difference between the. Standard Cell Potential Of Zinc And Lead.
From www.numerade.com
SOLVED Calculate the standard potential of the cell consisting of the Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Only the difference between the potentials of two electrodes can be measured. Describe and relate the definitions of electrode and cell potentials. The. Standard Cell Potential Of Zinc And Lead.
From app.pandai.org
Standard Electrode Potential Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. “transfer” of electrons requires both a donor and. Only the difference between the potentials of two electrodes can be measured. Interpret electrode potentials in terms of relative oxidant and. Standard Cell Potential Of Zinc And Lead.
From www.youtube.com
Electrochemistry 05 Calculating Standard Cell Potentials YouTube Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Interpret electrode potentials in terms. Standard Cell Potential Of Zinc And Lead.
From question.pandai.org
Standard Electrode Potential Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c). Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID2281210 Standard Cell Potential Of Zinc And Lead Interpret electrode potentials in terms of relative oxidant and reductant. Describe and relate the definitions of electrode and cell potentials. “transfer” of electrons requires both a donor and. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. The potential of the cell under standard conditions. Standard Cell Potential Of Zinc And Lead.
From www.toppr.com
The standard reduction potentials of Zn^2 + Zn and Cu^2 + Cu are Standard Cell Potential Of Zinc And Lead Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Describe and relate the definitions. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID2281210 Standard Cell Potential Of Zinc And Lead “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Describe and relate the definitions of electrode and cell potentials. The cell potential, \(e_{cell}\), is. Standard Cell Potential Of Zinc And Lead.
From ch302.cm.utexas.edu
stdpotsshortlist.png Standard Cell Potential Of Zinc And Lead Interpret electrode potentials in terms of relative oxidant and reductant. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. Only the difference between the potentials of. Standard Cell Potential Of Zinc And Lead.
From chem.libretexts.org
19.5 Standard Electrochemical Potentials Chemistry LibreTexts Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c). Standard Cell Potential Of Zinc And Lead.
From byjus.com
Variation of Cell Potential in ZnCu Cell Chemistry Practicals Class Standard Cell Potential Of Zinc And Lead Only the difference between the potentials of two electrodes can be measured. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). The potential of the cell under standard conditions (1 m for. Standard Cell Potential Of Zinc And Lead.
From questions-in.kunduz.com
Calculate the standard, cell potential of... Physical Chemistry Standard Cell Potential Of Zinc And Lead Interpret electrode potentials in terms of relative oxidant and reductant. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be. Standard Cell Potential Of Zinc And Lead.
From notes.pegas.is
Types of Reactions Pega's Notes Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Only the difference between the potentials of two electrodes can be measured. Describe and relate the definitions of electrode and cell potentials. “transfer” of electrons requires both a donor and. Since the tabulated standard electrode potentials are reduction potentials, the one. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID2281210 Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Since the tabulated standard electrode potentials are reduction potentials, the one which is. Standard Cell Potential Of Zinc And Lead.
From www.chemicals.co.uk
A Level Chemistry Electrodes & Electrochemical Cells Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. Only the difference between the potentials of two electrodes can be measured. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Interpret electrode potentials in terms of relative oxidant and reductant. “transfer” of electrons requires both a donor and. The. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT 14.2b Standard Cells and Cell Potential PowerPoint Presentation Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Interpret electrode potentials in. Standard Cell Potential Of Zinc And Lead.
From courses.lumenlearning.com
17.3 Standard Reduction Potentials General College Chemistry II Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Only the difference between the potentials of two electrodes can. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation ID165903 Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. The cell potential, \(e_{cell}\), is the measure of the potential difference. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Chapter 20 PowerPoint Presentation, free download ID6976551 Standard Cell Potential Of Zinc And Lead Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. Only the difference between the potentials of two electrodes can be measured. “transfer” of electrons requires both a donor and. Describe and relate the definitions of electrode and cell potentials. The cell potential, \(e_{cell}\), is the. Standard Cell Potential Of Zinc And Lead.
From inspiritvr.com
Calculating Standard Cell Potentials Study Guide Inspirit Standard Cell Potential Of Zinc And Lead “transfer” of electrons requires both a donor and. Only the difference between the potentials of two electrodes can be measured. Describe and relate the definitions of electrode and cell potentials. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Since the tabulated standard electrode potentials are reduction potentials, the one. Standard Cell Potential Of Zinc And Lead.
From rohanfersmorrison.blogspot.com
Identify the Conditions for a Standard Electrochemical Cell. Standard Cell Potential Of Zinc And Lead Only the difference between the potentials of two electrodes can be measured. “transfer” of electrons requires both a donor and. The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for. Standard Cell Potential Of Zinc And Lead.
From www.youtube.com
Determination of Standard Electrode potential of Zinc and Copper YouTube Standard Cell Potential Of Zinc And Lead Only the difference between the potentials of two electrodes can be measured. “transfer” of electrons requires both a donor and. Interpret electrode potentials in terms of relative oxidant and reductant. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is. Standard Cell Potential Of Zinc And Lead.
From www.pveducation.org
Standard Potential PVEducation Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Only the difference between the potentials of two electrodes can. Standard Cell Potential Of Zinc And Lead.
From www.youtube.com
STANDARD CELL POTENTIAL YouTube Standard Cell Potential Of Zinc And Lead Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e°. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Chapter 20 Electrochemistry PowerPoint Presentation, free Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Interpret electrode potentials in terms of relative oxidant and reductant. Describe and relate the definitions of electrode and cell potentials. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in. Standard Cell Potential Of Zinc And Lead.
From www.chemistrystudent.com
Electrochemistry (ALevel) ChemistryStudent Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to. Standard Cell Potential Of Zinc And Lead.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID1195602 Standard Cell Potential Of Zinc And Lead Interpret electrode potentials in terms of relative oxidant and reductant. “transfer” of electrons requires both a donor and. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). The potential of the cell. Standard Cell Potential Of Zinc And Lead.
From www.toppr.com
A galvanic cell consists of a metallic. zinc plate immersed in 0.1 M Zn Standard Cell Potential Of Zinc And Lead Describe and relate the definitions of electrode and cell potentials. The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called. Only the difference between the potentials of two electrodes can be measured. The cell potential, \(e_{cell}\), is the measure. Standard Cell Potential Of Zinc And Lead.
From courses.lumenlearning.com
17.3 Standard Reduction Potentials Chemistry Standard Cell Potential Of Zinc And Lead The potential of the cell under standard conditions (1 m for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°c) is called the standard cell potential (e° cell). The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. “transfer” of electrons requires. Standard Cell Potential Of Zinc And Lead.
From www.nagwa.com
Question Video Calculating the Standard Cell Potential for a Copper Standard Cell Potential Of Zinc And Lead Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. “transfer” of electrons requires both a donor and. Interpret electrode potentials in terms of relative oxidant and reductant. Only the difference between the potentials of two electrodes can be measured. Describe and relate the definitions of. Standard Cell Potential Of Zinc And Lead.
From www.numerade.com
SOLVEDThe standard cell potential, E^∘, for the reaction of Zn(s) and Standard Cell Potential Of Zinc And Lead The cell potential, \(e_{cell}\), is the measure of the potential difference between two half cells in an electrochemical cell. Describe and relate the definitions of electrode and cell potentials. Since the tabulated standard electrode potentials are reduction potentials, the one which is most negative will need to be reversed in sign to get. “transfer” of electrons requires both a donor. Standard Cell Potential Of Zinc And Lead.