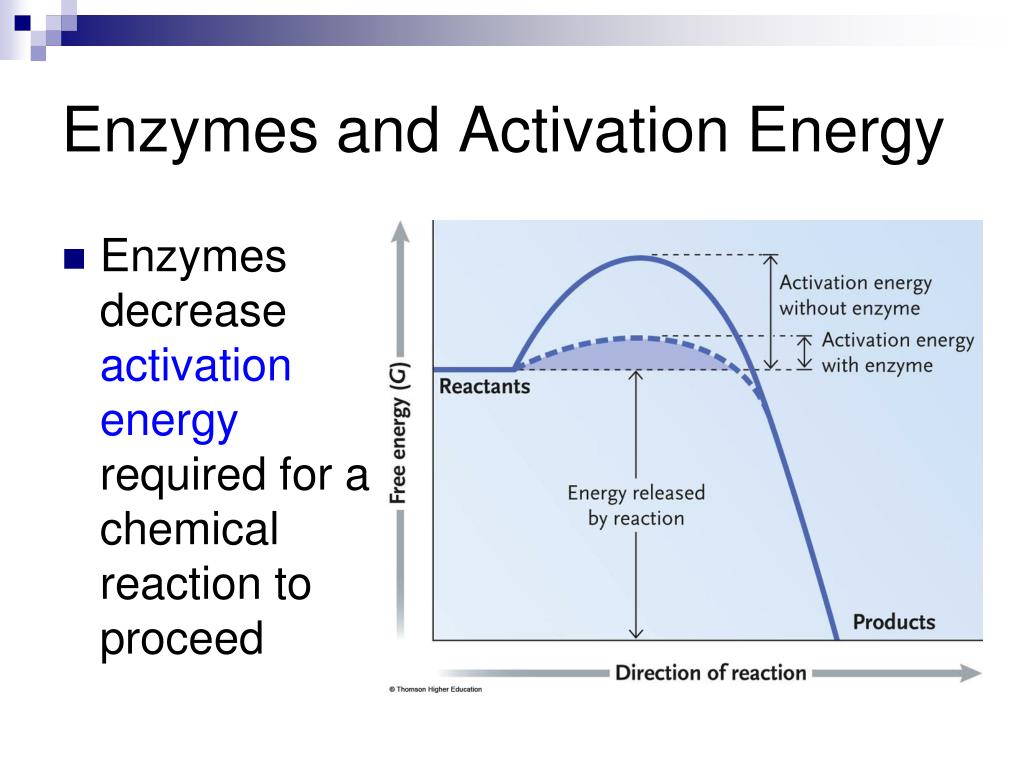
How Do Enzymes Lower Activation Energy Of A Chemical Reaction . Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. Enzymes lower the activation energy necessary to transform a reactant into a product. Enzymes can lower the activation energy of a chemical reaction in three ways. Enzymatic action can aid this process. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). One of the ways the activation energy is lowered is having the. They do this by lowering the activation energy of the. In chemistry, a catalyst is a chemical that drives a reaction forward. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. Enzymes are biological catalysts that speed up chemical reactions in living organisms. How do enzymes speed up biochemical reactions so dramatically? On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. This complex lowers the reaction's activation energy and promotes.
from www.slideserve.com
On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. In chemistry, a catalyst is a chemical that drives a reaction forward. Enzymatic action can aid this process. One of the ways the activation energy is lowered is having the. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. Enzymes can lower the activation energy of a chemical reaction in three ways. They do this by lowering the activation energy of the. Enzymes are biological catalysts that speed up chemical reactions in living organisms. This complex lowers the reaction's activation energy and promotes. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward.
PPT Energy, Enzymes, and Biological Reactions PowerPoint Presentation
How Do Enzymes Lower Activation Energy Of A Chemical Reaction On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. Enzymes can lower the activation energy of a chemical reaction in three ways. Enzymes lower the activation energy necessary to transform a reactant into a product. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). Enzymes are biological catalysts that speed up chemical reactions in living organisms. How do enzymes speed up biochemical reactions so dramatically? In chemistry, a catalyst is a chemical that drives a reaction forward. Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. One of the ways the activation energy is lowered is having the. They do this by lowering the activation energy of the. This complex lowers the reaction's activation energy and promotes. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. Enzymatic action can aid this process. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward.
From www.slideserve.com
PPT EnZyMeS PowerPoint Presentation, free download ID2065739 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. One of the ways the activation energy is lowered is having the. They do this by lowering the activation energy of the. Enzymes lower the activation energy necessary to transform a reactant into a product. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. This complex lowers. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Enzymes PowerPoint Presentation, free download ID4283724 How Do Enzymes Lower Activation Energy Of A Chemical Reaction This complex lowers the reaction's activation energy and promotes. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). How do enzymes speed up biochemical reactions so dramatically? Enzymes are biological catalysts that speed up chemical reactions in living organisms. Like all catalysts , enzymes work by lowering the activation energy. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Energy, Enzymes, and Biological Reactions PowerPoint Presentation How Do Enzymes Lower Activation Energy Of A Chemical Reaction How do enzymes speed up biochemical reactions so dramatically? This complex lowers the reaction's activation energy and promotes. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. One of the ways the activation energy is lowered is having the. Enzymatic action can aid this process. Enzymes are chemical catalysts that accelerate chemical reactions. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From telegra.ph
How do enzymes lower activation energy of chemical reactions Telegraph How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes lower the activation energy necessary to transform a reactant into a product. Enzymatic action can aid this process. Enzymes can lower the activation energy of a chemical reaction in three ways. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. In chemistry, a catalyst is a chemical that drives a reaction forward.. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.britannica.com
Activation energy Definition & Facts Britannica How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). In chemistry, a catalyst is a chemical that drives a reaction forward. Like all catalysts , enzymes work by lowering the activation energy of chemical. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From studylib.net
Enzymes Lower Activation Energy How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes can lower the activation energy of a chemical reaction in three ways. How do enzymes speed up biochemical reactions so dramatically? On the left is a reaction that is not catalyzed by an enzyme (red), and on. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Ch 4 Energy and Cellular Metabolism PowerPoint Presentation How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes lower the activation energy necessary to transform a reactant into a product. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. How do enzymes speed up biochemical reactions so dramatically? This complex lowers the reaction's activation energy and promotes. Enzymatic action can aid this process. Enzymes are biological. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.pinterest.com
Catalyst speeds up a chemical reaction by lowering the activation How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. This complex lowers the reaction's activation energy and promotes. Enzymes lower the activation energy necessary to transform a reactant into a product. They do this by lowering the activation energy of the. Like all catalysts , enzymes work by lowering the activation energy of. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.genome.gov
Enzyme How Do Enzymes Lower Activation Energy Of A Chemical Reaction One of the ways the activation energy is lowered is having the. They do this by lowering the activation energy of the. This complex lowers the reaction's activation energy and promotes. Enzymes lower the activation energy necessary to transform a reactant into a product. Catalysts lower the activation energy, which is the amount of energy required for reactants to form. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From fixportillobadgered.z21.web.core.windows.net
Energy Diagram Activation Energy How Do Enzymes Lower Activation Energy Of A Chemical Reaction On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. In chemistry, a catalyst is a chemical that drives a reaction forward. They do this by lowering the activation energy of the. Enzymes are. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Endergonic and Exergonic Reactions PowerPoint Presentation ID How Do Enzymes Lower Activation Energy Of A Chemical Reaction They do this by lowering the activation energy of the. In chemistry, a catalyst is a chemical that drives a reaction forward. One of the ways the activation energy is lowered is having the. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. How do enzymes speed up biochemical. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From present5.com
Enzyme Structure classification and mechanism of action How Do Enzymes Lower Activation Energy Of A Chemical Reaction On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. One of the ways the activation energy is lowered is having the. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). Enzymes are biological catalysts that speed up chemical reactions. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.savemyexams.com
Activation Energy Skills HL IB Biology Revision Notes 2025 Save My How Do Enzymes Lower Activation Energy Of A Chemical Reaction How do enzymes speed up biochemical reactions so dramatically? Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). They do this by lowering the activation energy of the. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. Enzymes lower the activation energy necessary. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.sliderbase.com
Enzymes. A Cell's Catalysts Presentation Biology How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. This complex lowers the reaction's activation energy and promotes. Enzymes are biological catalysts that speed. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT HOW ENZYMES WORK PowerPoint Presentation, free download ID6954410 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). One of the ways the activation energy is lowered is having the. Enzymes are biological catalysts that speed up chemical reactions in living organisms. Enzymes can lower the activation energy of a chemical reaction in three ways. Enzymes lower the activation. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Metabolism PowerPoint Presentation, free download ID1153506 How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. Enzymes lower the activation energy necessary to transform a reactant into a product. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. Catalysts lower the activation energy, which is the amount of energy required for reactants to. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT CHAPTER 3 PowerPoint Presentation, free download ID1361511 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes can lower the activation energy of a chemical reaction in three ways. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes are biological catalysts that speed up chemical reactions in living organisms. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. On the left. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From thebiologs.blogspot.com
The BioLogs CAPE 1 Enzymes and Enzyme Activity How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). One of the ways the activation energy is lowered is having the. This complex lowers the reaction's activation energy and promotes. In chemistry, a catalyst is a chemical that drives a reaction forward. On the left is a reaction that is. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.onlinebiologynotes.com
Enzymes Properties and Mechanism of enzyme action Online Biology Notes How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. In chemistry, a catalyst is a chemical that drives a reaction forward. This complex lowers the reaction's activation energy and promotes. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes can lower the activation energy of a chemical reaction in three ways. Enzymes lower the activation. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT 6.2 Enzymes and Chemical Reactions pages 156160 PowerPoint How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes can lower the activation energy of a chemical reaction in three ways. How do enzymes speed up biochemical reactions so dramatically? Enzymes are biological catalysts that speed up chemical reactions in living organisms. Enzymes are chemical catalysts that accelerate chemical reactions at physiological. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From bettergop.weebly.com
How Do Enzymes Lower Activation Energy bettergop How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). How do enzymes speed up biochemical reactions so dramatically? In chemistry, a catalyst is a chemical that drives a. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Enzymes as Biological Catalysts PowerPoint Presentation, free How Do Enzymes Lower Activation Energy Of A Chemical Reaction They do this by lowering the activation energy of the. In chemistry, a catalyst is a chemical that drives a reaction forward. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes are biological catalysts that speed up chemical reactions in living organisms. This complex lowers the reaction's activation energy and promotes. On. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Enzymes PowerPoint Presentation, free download ID1936659 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. In chemistry, a catalyst is a chemical that drives a reaction forward. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. How do. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Metabolic Reactions and Enzymes PowerPoint Presentation, free How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. They do this by lowering the activation energy of the. Enzymatic action can aid this process. Enzymes are chemical catalysts that. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT ENZYMES PowerPoint Presentation, free download ID1171133 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes lower the activation energy necessary to transform a reactant into a product. They do this by lowering the activation energy of the. Catalysts also lower the kinetic barrier, which is needed to drive a reaction forward and backward. Enzymes can lower the activation energy of a chemical reaction in three ways. One of the ways the activation energy is. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From kenya-khurst.blogspot.com
Catalysts Lower the Activation Energy of a Reaction by How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. How do enzymes speed up biochemical reactions so dramatically? This complex lowers the reaction's activation energy and promotes. Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. One of the ways the activation energy is lowered is having the. Enzymes lower the activation. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Enzymes Biological Catalysts PowerPoint Presentation ID5736986 How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. They do this by lowering the activation energy of the. Enzymes are biological catalysts that speed up chemical reactions in living organisms. Enzymes lower the activation energy necessary to transform a reactant into a product. This complex lowers the reaction's activation energy and promotes.. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www2.nau.edu
Enzymes and Reaction Rates How Do Enzymes Lower Activation Energy Of A Chemical Reaction Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). They do this by lowering the activation energy of the. How do enzymes speed up biochemical reactions so dramatically? Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. On the left is a reaction that is. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From telegra.ph
How do enzymes lower activation energy of a reaction Telegraph How Do Enzymes Lower Activation Energy Of A Chemical Reaction Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. They do this by lowering the activation energy of the. Enzymes lower the activation energy necessary to transform a reactant into a product. This complex lowers the reaction's activation energy and promotes. Enzymes can lower the activation energy of a chemical reaction in three ways. Enzymes. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From animalia-life.club
Enzyme Activation Energy Graph How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. Enzymes can lower the activation energy of a chemical reaction in three ways. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). Enzymes are biological catalysts that speed up chemical reactions in living organisms. One of the ways. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Enzymes Just because a reaction is spontaneous, that does not How Do Enzymes Lower Activation Energy Of A Chemical Reaction This complex lowers the reaction's activation energy and promotes. On the left is a reaction that is not catalyzed by an enzyme (red), and on the right is one. In chemistry, a catalyst is a chemical that drives a reaction forward. Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. One of the ways the. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.expii.com
What Are Enzymes? — Definition & Overview Expii How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. They do this by lowering the activation energy of the. Catalysts lower the activation energy, which is the amount of energy required for reactants to form products (figure 1). Enzymes lower the activation energy necessary to transform a reactant into a product. Catalysts also lower the kinetic barrier, which is needed to drive a. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From btccasting.weebly.com
Enzymes Lower The Activation Energy Of A Reaction btccasting How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. How do enzymes speed up biochemical reactions so dramatically? One of the ways the activation energy is lowered is having the. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. On the left is a reaction that is not catalyzed by. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT ENZYMES PowerPoint Presentation, free download ID6886656 How Do Enzymes Lower Activation Energy Of A Chemical Reaction In chemistry, a catalyst is a chemical that drives a reaction forward. Enzymes can lower the activation energy of a chemical reaction in three ways. Like all catalysts , enzymes work by lowering the activation energy of chemical reactions. Enzymes are biological catalysts that speed up chemical reactions in living organisms. Enzymatic action can aid this process. How do enzymes. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.
From www.slideserve.com
PPT Chemical reactions and enzymes PowerPoint Presentation, free How Do Enzymes Lower Activation Energy Of A Chemical Reaction Enzymatic action can aid this process. Enzymes are chemical catalysts that accelerate chemical reactions at physiological temperatures by lowering their activation energy. How do enzymes speed up biochemical reactions so dramatically? They do this by lowering the activation energy of the. Enzymes can lower the activation energy of a chemical reaction in three ways. On the left is a reaction. How Do Enzymes Lower Activation Energy Of A Chemical Reaction.