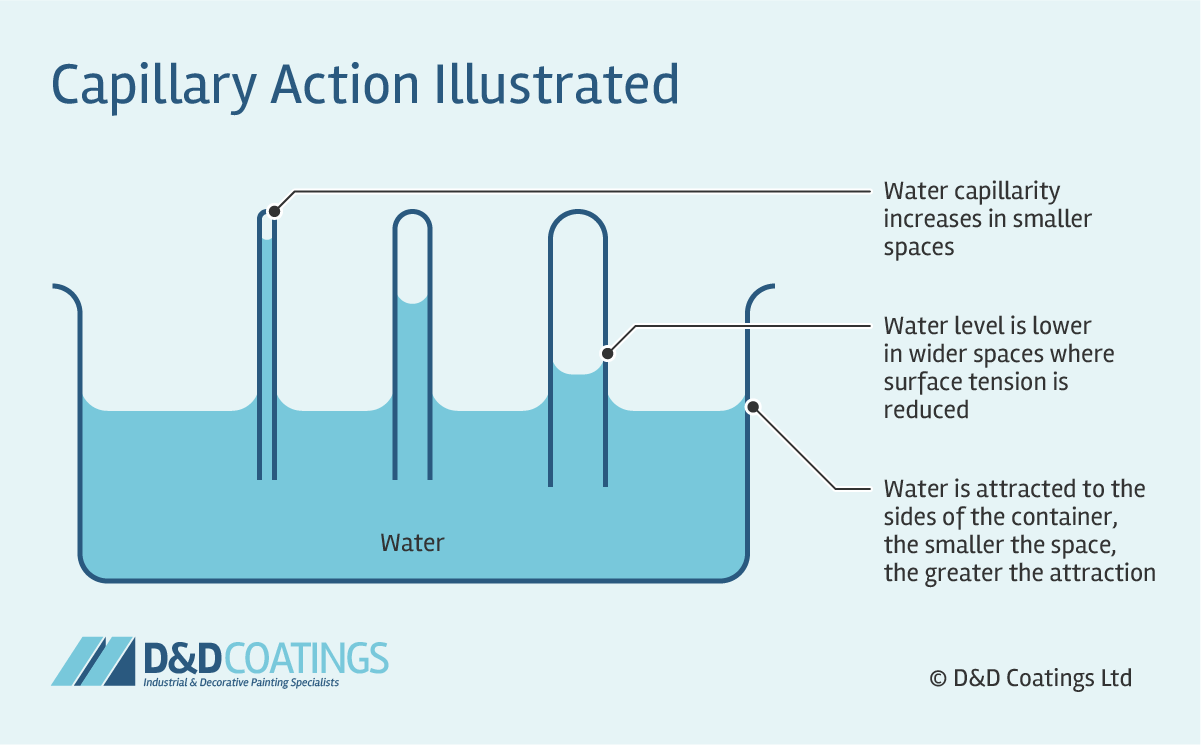
Why Does Water Rise In A Glass Tube . Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. Inside a thin glass tube, the adhesive. A meniscus is the curved surface at the top of a column of liquid. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It is curving downward, like mercury in a glass tube. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. A convex meniscus is what you see when certain liquids, like mercury,. It's a very popular experiment , from elementary school : In a science class, this liquid is usually water or some sort of aqueous solution,. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. This occurs when cohesion is stronger than adhesion. Capillary action causes the rise of damp.
from www.ddcoatings.co.uk
Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. It is curving downward, like mercury in a glass tube. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. This occurs when cohesion is stronger than adhesion. In a science class, this liquid is usually water or some sort of aqueous solution,. Capillary action causes the rise of damp. It's a very popular experiment , from elementary school : A meniscus is the curved surface at the top of a column of liquid. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces.
What is Capillary Action?
Why Does Water Rise In A Glass Tube Capillary action causes the rise of damp. Capillary action causes the rise of damp. It's a very popular experiment , from elementary school : Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. A convex meniscus is what you see when certain liquids, like mercury,. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. This occurs when cohesion is stronger than adhesion. It is curving downward, like mercury in a glass tube. Inside a thin glass tube, the adhesive. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. A meniscus is the curved surface at the top of a column of liquid. In a science class, this liquid is usually water or some sort of aqueous solution,.
From team-cartwright.com
Rising Water Experiment Magic Water Science Experiment Why Does Water Rise In A Glass Tube In a science class, this liquid is usually water or some sort of aqueous solution,. Inside a thin glass tube, the adhesive. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. A meniscus is the curved surface at the top of a column of. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Rise in liquid in Capillary Tube Surface Tension 12th Physics YouTube Why Does Water Rise In A Glass Tube Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It is curving downward, like mercury in a glass tube. Capillary action causes the rise of damp. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. Capillarity. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Why Does Water Rise? Balloon in a Bottle YouTube Why Does Water Rise In A Glass Tube Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. It's a very popular experiment , from elementary. Why Does Water Rise In A Glass Tube.
From www.youtube.com
The Rising Water Science Experiment Why Does Water Rise Up Why Does Water Rise In A Glass Tube Inside a thin glass tube, the adhesive. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. It's a very popular experiment , from elementary school : Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. A. Why Does Water Rise In A Glass Tube.
From byjus.com
When a glass capillary tube of radius 0.015 cm isdipped in water, the Why Does Water Rise In A Glass Tube When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. Capillary action causes the rise of damp. In a science class, this liquid is usually water. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Why does water rise ? Easy DIY science Experiments for kids YouTube Why Does Water Rise In A Glass Tube Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. This occurs when cohesion is stronger than adhesion. It's a very popular experiment , from elementary school :. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved 5. A 0.6 mmdiameter glass tube is inserted into Why Does Water Rise In A Glass Tube It is curving downward, like mercury in a glass tube. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. In a science class, this liquid is usually water or some sort of aqueous solution,. When you place a straw in a glass of water,. Why Does Water Rise In A Glass Tube.
From www.youtube.com
A capillary tube of radius r is immersed in water and water rises in it Why Does Water Rise In A Glass Tube This occurs when cohesion is stronger than adhesion. It is curving downward, like mercury in a glass tube. A meniscus is the curved surface at the top of a column of liquid. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. In. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Glass And Candle Experiment Why Does Water Rise? YouTube Why Does Water Rise In A Glass Tube Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It's a very popular experiment , from elementary school : Capillary action causes the rise of damp. In a science class, this liquid is usually water or some sort of aqueous solution,. A convex meniscus is what you see when certain liquids, like. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved How does the capillary rise in a glass tube depend Why Does Water Rise In A Glass Tube Capillary action causes the rise of damp. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. It is curving downward, like mercury in a glass tube. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. Inside. Why Does Water Rise In A Glass Tube.
From courses.lumenlearning.com
Water Potential Biology I Why Does Water Rise In A Glass Tube Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. In a science class, this liquid is usually water or some sort of aqueous solution,. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. Learn how the. Why Does Water Rise In A Glass Tube.
From med.libretexts.org
2.3B Water’s Cohesive and Adhesive Properties Medicine LibreTexts Why Does Water Rise In A Glass Tube Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. A meniscus is the curved surface at the top of a column of liquid. This occurs when cohesion is stronger than adhesion. When you place a straw in a glass of water, the liquid level within the straw is higher than the height. Why Does Water Rise In A Glass Tube.
From www.numerade.com
SOLVED A 0.6 mmdiameter glass tube is inserted into water as 20°C in Why Does Water Rise In A Glass Tube A convex meniscus is what you see when certain liquids, like mercury,. It is curving downward, like mercury in a glass tube. Inside a thin glass tube, the adhesive. In a science class, this liquid is usually water or some sort of aqueous solution,. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids. Why Does Water Rise In A Glass Tube.
From www.ddcoatings.co.uk
What is Capillary Action? Why Does Water Rise In A Glass Tube It's a very popular experiment , from elementary school : This occurs when cohesion is stronger than adhesion. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Water rises in a vertical capillary tube upto 10 cm . If the tube is Why Does Water Rise In A Glass Tube Inside a thin glass tube, the adhesive. A meniscus is the curved surface at the top of a column of liquid. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. When you place a straw in a glass of water, the liquid level within. Why Does Water Rise In A Glass Tube.
From www.toppr.com
A glass tube of 1.0 m length is filled with water. The water can be Why Does Water Rise In A Glass Tube Inside a thin glass tube, the adhesive. A meniscus is the curved surface at the top of a column of liquid. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. Capillarity is the combined effect of cohesive and adhesive forces that causes water and. Why Does Water Rise In A Glass Tube.
From www.youtube.com
The rising water Science experiment Why does water rise up Why Does Water Rise In A Glass Tube This occurs when cohesion is stronger than adhesion. It is curving downward, like mercury in a glass tube. A meniscus is the curved surface at the top of a column of liquid. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. Capillarity is the. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Glass And candle Experiment why Does Water Rise? XSkull Experiment🇧🇩 Why Does Water Rise In A Glass Tube A convex meniscus is what you see when certain liquids, like mercury,. This occurs when cohesion is stronger than adhesion. Capillary action causes the rise of damp. It's a very popular experiment , from elementary school : Inside a thin glass tube, the adhesive. Put a burning candle on a dish filled with water, cover the candle with an inverted. Why Does Water Rise In A Glass Tube.
From sealevel.nasa.gov
Melting Ocean Ice Affects Sea Level Unlike Ice Cubes in a Glass Why Does Water Rise In A Glass Tube It's a very popular experiment , from elementary school : This occurs when cohesion is stronger than adhesion. Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. A meniscus is the curved surface at the top of a column of liquid. In a science. Why Does Water Rise In A Glass Tube.
From frugalfun4boys.com
Candle and Rising Water Science Experiment Frugal Fun For Boys and Girls Why Does Water Rise In A Glass Tube When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. A convex meniscus is what you see when certain liquids, like mercury,. It is curving downward,. Why Does Water Rise In A Glass Tube.
From byjus.com
Water rises to a height of 10cm in a capillary tube and mercury falls Why Does Water Rise In A Glass Tube A convex meniscus is what you see when certain liquids, like mercury,. It's a very popular experiment , from elementary school : Capillary action causes the rise of damp. In a science class, this liquid is usually water or some sort of aqueous solution,. It is curving downward, like mercury in a glass tube. This occurs when cohesion is stronger. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved = 1. Find the capillary rise in the tube for a Why Does Water Rise In A Glass Tube When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. A meniscus is the curved surface at the top of a column of liquid. Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Burning Candle Rising Water Experiment Why does water rise? YouTube Why Does Water Rise In A Glass Tube Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It is curving downward, like mercury in a glass tube. This occurs when cohesion is stronger than adhesion.. Why Does Water Rise In A Glass Tube.
From brainly.in
4. With reference to the diagram answer the following chemical K glass Why Does Water Rise In A Glass Tube A convex meniscus is what you see when certain liquids, like mercury,. In a science class, this liquid is usually water or some sort of aqueous solution,. It's a very popular experiment , from elementary school : Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved CAPILLARY EFFECT • Capillary effect is the rise or Why Does Water Rise In A Glass Tube Learn how the free energy of wetting and the gravitational work balance determine the height and shape of a liquid column in a capillary tube. This occurs when cohesion is stronger than adhesion. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It is curving downward, like mercury in a glass tube.. Why Does Water Rise In A Glass Tube.
From classnotes.org.in
Surface Tension Chemistry, Class 11, States of Matter Why Does Water Rise In A Glass Tube When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. It is curving downward, like mercury in a glass tube. Capillary action causes the rise of damp. Put a burning candle on a dish filled with water, cover the candle with an inverted. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Why does water stick to your glass when pouring? YouTube Why Does Water Rise In A Glass Tube When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It is curving downward, like mercury in a glass tube. Capillarity is the combined effect of. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved Problem 4.055 A glass tube is inserted into a flowing Why Does Water Rise In A Glass Tube Capillary action causes the rise of damp. It's a very popular experiment , from elementary school : Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. A convex meniscus is what you see when certain liquids, like mercury,. A meniscus is the curved surface at the top of a column of liquid.. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Why Does Water Rise? Candle and Glass Experiment YouTube Why Does Water Rise In A Glass Tube Inside a thin glass tube, the adhesive. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. Capillary action causes the rise of damp. In a science class, this liquid is usually water or some sort of aqueous solution,. A convex meniscus is what you see when certain liquids, like mercury,. It is. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Make Water Rise in a Jar Experiment YouTube Why Does Water Rise In A Glass Tube A convex meniscus is what you see when certain liquids, like mercury,. It's a very popular experiment , from elementary school : Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. Put a burning candle on a dish filled with water, cover the candle. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved A 0.6mmdiameter glass tube is inserted into water Why Does Water Rise In A Glass Tube Inside a thin glass tube, the adhesive. A convex meniscus is what you see when certain liquids, like mercury,. It's a very popular experiment , from elementary school : Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. In a science class, this liquid. Why Does Water Rise In A Glass Tube.
From www.researchgate.net
(a) Sketch of capillary rise in a vertical tube. (b) Sketch of the Why Does Water Rise In A Glass Tube A meniscus is the curved surface at the top of a column of liquid. This occurs when cohesion is stronger than adhesion. Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. When you place a straw in a glass of water, the liquid level within the straw is higher than the height. Why Does Water Rise In A Glass Tube.
From www.numerade.com
q1 a glass tube is attached to a water pipe as shown in fig 1 if the Why Does Water Rise In A Glass Tube Capillarity is the combined effect of cohesive and adhesive forces that causes water and other liquids to rise in thin tubes or other constricted spaces. In a science class, this liquid is usually water or some sort of aqueous solution,. It is curving downward, like mercury in a glass tube. When you place a straw in a glass of water,. Why Does Water Rise In A Glass Tube.
From www.youtube.com
Glass And Candle Experiment Why Does Water Rise? LAHORE SCIENCE CLUB Why Does Water Rise In A Glass Tube It's a very popular experiment , from elementary school : Capillary action causes the rise of damp. Inside a thin glass tube, the adhesive. When you place a straw in a glass of water, the liquid level within the straw is higher than the height of the water in the glass. In a science class, this liquid is usually water. Why Does Water Rise In A Glass Tube.
From www.chegg.com
Solved A glass tube is inserted into a flowing stream of Why Does Water Rise In A Glass Tube Put a burning candle on a dish filled with water, cover the candle with an inverted glass:. It's a very popular experiment , from elementary school : A meniscus is the curved surface at the top of a column of liquid. A convex meniscus is what you see when certain liquids, like mercury,. Capillarity is the combined effect of cohesive. Why Does Water Rise In A Glass Tube.