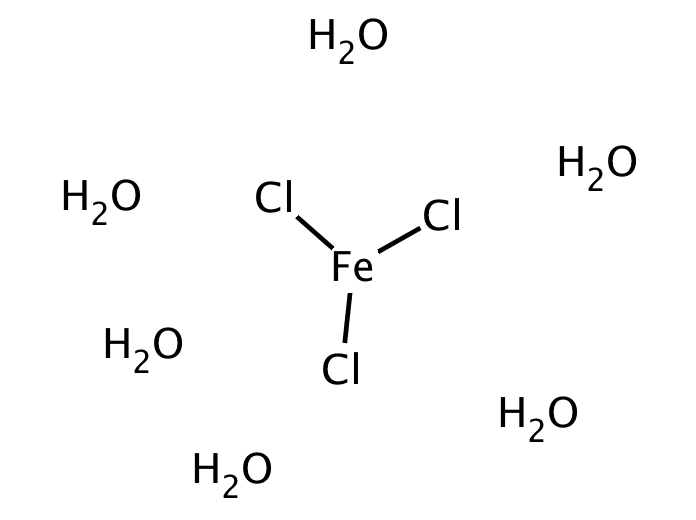
Iron + Chlorine Gives Iron Iii Chloride . This happens because chlorine is a powerful. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Write a balanced chemical equation for the following word equation: 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron + chlorine → iron (iii) chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial.
from www.smartscience.co.th
First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Write a balanced chemical equation for the following word equation: Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. 2 iron + 3 chlorine = 2 iron(iii) chloride : This happens because chlorine is a powerful. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron + chlorine → iron (iii) chloride
Iron(III) chloride hexahydrate smartscience
Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron + chlorine → iron (iii) chloride This happens because chlorine is a powerful. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. 2 iron + 3 chlorine = 2 iron(iii) chloride : Write a balanced chemical equation for the following word equation:
From www.bartleby.com
Answered 24. Solid iron and chlorine gas react… bartleby Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below;. Iron + Chlorine Gives Iron Iii Chloride.
From testbook.com
Iron (III) Chloride Formula Know Structure, Preparation,& Uses Iron + Chlorine Gives Iron Iii Chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. This happens because chlorine is a powerful. Iron + chlorine → iron (iii) chloride Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that. Iron + Chlorine Gives Iron Iii Chloride.
From www.youtube.com
How to write the formula for iron (III) chloride YouTube Iron + Chlorine Gives Iron Iii Chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. This happens because chlorine is a powerful. Write a balanced chemical equation for the following word equation: First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron (iii) chloride,. Iron + Chlorine Gives Iron Iii Chloride.
From www.smartscience.co.th
Iron(III) chloride hexahydrate smartscience Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous. Iron + Chlorine Gives Iron Iii Chloride.
From www.toppr.com
moleculal 101WUR I WIL 43. In chemical analysis of a compound, 3.438g Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride This happens because chlorine is a powerful. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown. Iron + Chlorine Gives Iron Iii Chloride.
From www.researchgate.net
What is the chemical difference between these iron III chloride solutions? Iron + Chlorine Gives Iron Iii Chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. This happens because chlorine is a powerful. Write a. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVEDThe reaction of iron metal and chlorine ga… Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Write a balanced chemical equation for the following word equation: 2 iron + 3 chlorine = 2 iron(iii) chloride : First,. Iron + Chlorine Gives Iron Iii Chloride.
From www.bartleby.com
Answered The Iron metal reacts with chlorine gas… bartleby Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron + chlorine → iron (iii) chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; When iron. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVEDThe reaction of iron metal and chlorine gas to give iron(III Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron (iii) chloride, commonly known as ferric chloride,. Iron + Chlorine Gives Iron Iii Chloride.
From www.youtube.com
Iron(III) chloride YouTube Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride Write a balanced chemical equation for the following word equation: First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) chloride, commonly known as ferric chloride,. Iron + Chlorine Gives Iron Iii Chloride.
From www.slideserve.com
PPT Today is Friday (!), March 14 th , 2014 PowerPoint Presentation Iron + Chlorine Gives Iron Iii Chloride Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Write a balanced chemical equation for the following word equation: This happens because chlorine is a powerful. When. Iron + Chlorine Gives Iron Iii Chloride.
From www.slideserve.com
PPT Binary Molecular PowerPoint Presentation, free download ID4566789 Iron + Chlorine Gives Iron Iii Chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. This happens because chlorine is a powerful. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron + chlorine → iron (iii) chloride When iron is heated. Iron + Chlorine Gives Iron Iii Chloride.
From www.glentham.com
Iron(III) chloride, anhydrous (CAS 7705080) Glentham Life Sciences Iron + Chlorine Gives Iron Iii Chloride Write a balanced chemical equation for the following word equation: Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron(ii) chloride then reacts with chlorine to yield. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVEDUse the law of constant composition to complete the table Iron + Chlorine Gives Iron Iii Chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) oxide reacts. Iron + Chlorine Gives Iron Iii Chloride.
From www.youtube.com
Iron(iii) Chloride Preparation YouTube Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride This happens because chlorine is a powerful. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. When iron is heated in a stream of dry chlorine the product is iron. Iron + Chlorine Gives Iron Iii Chloride.
From www.sciencephoto.com
Anhydrous iron (III) chloride Stock Image C009/9429 Science Photo Iron + Chlorine Gives Iron Iii Chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. 2 iron + 3 chlorine = 2 iron(iii) chloride : When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Write a. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVED In the reaction to form iron(III) chloride, 10.0 grams of iron Iron + Chlorine Gives Iron Iii Chloride When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen. Iron + Chlorine Gives Iron Iii Chloride.
From app.pandai.org
Chemical Formulae Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. 2 iron + 3 chlorine = 2 iron(iii) chloride. Iron + Chlorine Gives Iron Iii Chloride.
From www.bartleby.com
The reaction of iron metal and chlorine gas to give iron(III) chloride Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron(ii) chloride then reacts. Iron + Chlorine Gives Iron Iii Chloride.
From slideplayer.com
Chapter 7 Compounds and Their Bonds ppt download Iron + Chlorine Gives Iron Iii Chloride Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; This happens because chlorine is a powerful. Iron + chlorine → iron (iii) chloride Write a balanced chemical equation for the following word equation: First, iron scrap is. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVEDMany metals react with halogens to give metal halides. For Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : This happens because chlorine is a powerful. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii). Iron + Chlorine Gives Iron Iii Chloride.
From www.youtube.com
Lewis Structure of Iron (III) Chloride, FeCl3 YouTube Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron + chlorine → iron (iii) chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. 2 iron + 3 chlorine = 2 iron(iii) chloride : Write a balanced. Iron + Chlorine Gives Iron Iii Chloride.
From www.slideserve.com
PPT Prentice Hall Chemistry (c) 2005 PowerPoint Presentation ID6658952 Iron + Chlorine Gives Iron Iii Chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron + chlorine → iron (iii) chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : This happens because chlorine is a powerful. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. First, iron scrap is. Iron + Chlorine Gives Iron Iii Chloride.
From www.alamy.com
Ironchlorine reaction. Iron (Fe) producing flames as it reacts with Iron + Chlorine Gives Iron Iii Chloride When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. This happens because chlorine is a powerful. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron + chlorine → iron (iii) chloride. Iron + Chlorine Gives Iron Iii Chloride.
From fphoto.photoshelter.com
science chemistry compound iron iii chloride Fundamental Photographs Iron + Chlorine Gives Iron Iii Chloride Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; This happens because chlorine is a powerful. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. 2 iron + 3 chlorine = 2 iron(iii) chloride : When. Iron + Chlorine Gives Iron Iii Chloride.
From fphoto.photoshelter.com
science chemistry compound iron iii chloride Fundamental Photographs Iron + Chlorine Gives Iron Iii Chloride Iron + chlorine → iron (iii) chloride Write a balanced chemical equation for the following word equation: Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then. Iron + Chlorine Gives Iron Iii Chloride.
From icsechemistry16.blogspot.com
Preparation of Iron (III) chloride (Ferric chloride) Iron + Chlorine Gives Iron Iii Chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron + chlorine → iron (iii) chloride Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Write a balanced chemical equation for the following word equation: When iron is heated in a stream of dry chlorine the product. Iron + Chlorine Gives Iron Iii Chloride.
From www.chegg.com
Solved 82. The reaction of iron metal and chlorine gas to Iron + Chlorine Gives Iron Iii Chloride Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Write a balanced chemical equation for the following word equation: When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and. Iron + Chlorine Gives Iron Iii Chloride.
From www.difference.wiki
Iron II Chloride vs. Iron III Chloride What’s the Difference? Iron + Chlorine Gives Iron Iii Chloride When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. This happens because chlorine is a powerful. First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. Iron + chlorine → iron (iii) chloride. Iron + Chlorine Gives Iron Iii Chloride.
From www.numerade.com
SOLVEDIron(III) chloride is produced by the reaction of iron and Iron + Chlorine Gives Iron Iii Chloride First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. This happens because chlorine. Iron + Chlorine Gives Iron Iii Chloride.
From www.youtube.com
How to Write the Formula for Iron (III) chloride YouTube Iron + Chlorine Gives Iron Iii Chloride Write a balanced chemical equation for the following word equation: First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas,. Iron + Chlorine Gives Iron Iii Chloride.
From favpng.com
Iron(III) Chloride Iron(III) Oxide Chlorine, PNG, 1100x759px, Ironiii Iron + Chlorine Gives Iron Iii Chloride Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; First, iron scrap is dissolved in the melt at 600 °c and oxidized to iron(ii) chloride by iron(iii) chloride. This happens because chlorine is a powerful. Write a balanced chemical equation for the following word equation: Iron + chlorine. Iron + Chlorine Gives Iron Iii Chloride.
From www.sciencephoto.com
Ironchlorine reaction Stock Image C011/4535 Science Photo Library Iron + Chlorine Gives Iron Iii Chloride Write a balanced chemical equation for the following word equation: Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron(ii) chloride then reacts with chlorine to yield. Iron + Chlorine Gives Iron Iii Chloride.
From www.askdifference.com
Iron II Chloride vs. Iron III Chloride — What’s the Difference? Iron + Chlorine Gives Iron Iii Chloride 2 iron + 3 chlorine = 2 iron(iii) chloride : Iron(ii) chloride then reacts with chlorine to yield iron(iii) chloride, which. When iron is heated in a stream of dry chlorine the product is iron (iii) chloride. Write a balanced chemical equation for the following word equation: This happens because chlorine is a powerful. Iron + chlorine → iron (iii). Iron + Chlorine Gives Iron Iii Chloride.
From www.slideserve.com
PPT Review of Atomic Model PowerPoint Presentation, free download Iron + Chlorine Gives Iron Iii Chloride Write a balanced chemical equation for the following word equation: Iron (iii) chloride, commonly known as ferric chloride, is an important inorganic compound that plays a significant role in numerous industrial. Iron (iii) oxide reacts with chlorine gas to give iron (iii) chloride and oxygen gas, according to the equation shown below; Iron(ii) chloride then reacts with chlorine to yield. Iron + Chlorine Gives Iron Iii Chloride.