How Much Heat Energy (Q) Did The Water Gain Gizmo . Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the. How much heat energy q did the water gain? Now solve for the specific heat c of copper. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Specific heat capacity can be calculated using the following equation: When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Water has a known specific heat capacity of 4 j/g °c. Use the specific heat equation to find out how much heat energy the water gained ( q). Calorimeters can be used to. Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4 j/g °c. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Water has a known specific heat capacity of 4.184 j/g °c.
from www.chegg.com
How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Use the specific heat equation to find out how much heat energy the. When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. How much heat energy q did the water gain? Use the specific heat equation to find out how much heat energy the. Now solve for the specific heat c of copper. Specific heat capacity can be calculated using the following equation: Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the water gained ( q). Calorimeters can be used to.
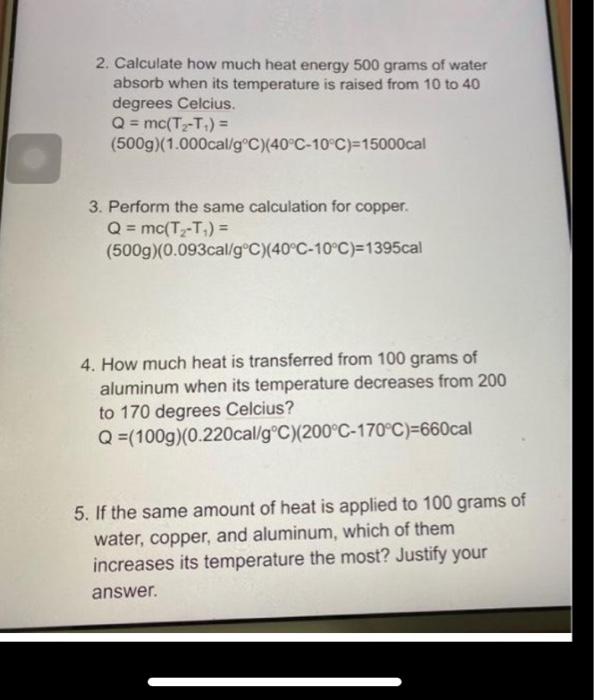
Solved 2. Calculate how much heat energy 500 grams of water
How Much Heat Energy (Q) Did The Water Gain Gizmo Specific heat capacity can be calculated using the following equation: How much heat energy q did the water gain? Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the water gained ( q). Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4.184 j/g °c. Specific heat capacity can be calculated using the following equation: Use the specific heat equation to find out how much heat energy the. Now solve for the specific heat c of copper. Calorimeters can be used to. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4 j/g °c. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ?
From www.slideserve.com
PPT Heat of Fusion PowerPoint Presentation ID2249917 How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4 j/g °c. Calorimeters can be used to. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Now solve for the specific heat c. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved 3. How much heat energy does 50.0 grams of water How Much Heat Energy (Q) Did The Water Gain Gizmo How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Now solve for the specific heat c of copper. Use the specific heat equation to find out how. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved How much heat energy must be removed from 200 g of How Much Heat Energy (Q) Did The Water Gain Gizmo Now solve for the specific heat c of copper. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the. Use the specific heat equation. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.numerade.com
SOLVED Part A How much heat energy; in kilojoules is required to How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4.184 j/g °c. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Water has a known specific heat capacity of 4 j/g °c. Use. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved How much heat energy is required to convert 39.6 g of How Much Heat Energy (Q) Did The Water Gain Gizmo How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Calorimeters can be used to. Use the specific heat equation to find out how much heat energy the.. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved 3. How can you show that the total heat absorbed (q) How Much Heat Energy (Q) Did The Water Gain Gizmo Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Calorimeters can be used to. Use the specific heat equation to find out how much heat energy the water gained ( q). Now solve for the specific heat c of copper. How much heat energy (q). How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved 2. Calculate Use the Gizmo to mix 200 g of copper at How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Specific heat capacity can be calculated using the following equation: Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy q did the water gain? Water has a known specific heat. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.youtube.com
How much heat energy is released when 5.0 g of water at 20^C changes How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Water has a known specific heat capacity of 4 j/g °c. How much heat energy q did the water gain? Use the specific. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From kunduz.com
[ANSWERED] When 41 g of water is cooled from 73 C to 40 C how much heat How Much Heat Energy (Q) Did The Water Gain Gizmo When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Use the specific heat equation to find out how much heat energy the. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From brainly.com
How much heat energy is absorbed when 7.0 moles of water boils How Much Heat Energy (Q) Did The Water Gain Gizmo Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Now solve for the specific heat c of copper. Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the water gained (. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.studocu.com
Specific Heat Worksheet Name How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Use the specific heat equation to find out how much heat energy the water gained ( q). Use the specific heat equation to find out how much heat energy the. Specific heat capacity can be calculated using the following equation: Water has a known specific heat capacity. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved How much heat energy, in kilojoules, is required to How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. When a hot object is placed in the calorimeter, heat energy is transferred from. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.toppr.com
How much heat energy is released when 5.0 g water at 42^∘C changes into How Much Heat Energy (Q) Did The Water Gain Gizmo When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Use the specific heat equation to find out how much heat energy. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From brainly.in
How much heat energy is a necessary to raisethe temperature of 5kg of How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? When a hot. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.slideserve.com
PPT 2. How much heat is required to boil 7.3g of Ammonia? (NH 3 ) ( Δ How Much Heat Energy (Q) Did The Water Gain Gizmo Now solve for the specific heat c of copper. When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Calorimeters can be used to. Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4.184. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.youtube.com
Week 1 16. How to calculate the heat absorbed when a substance How Much Heat Energy (Q) Did The Water Gain Gizmo Specific heat capacity can be calculated using the following equation: When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Use the specific heat equation to. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved Q10 This graph shows the rise in temperature as heat How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4 j/g °c. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.youtube.com
(1//2) mole of helium is contained in a container at STP how much heat How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the water gained ( q). Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. How much heat energy q did the water gain? Use. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.numerade.com
SOLVEDCalculate the heat change at 0^∘ C for each of the following How Much Heat Energy (Q) Did The Water Gain Gizmo How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Use the specific heat equation to find out how much heat energy the water gained ( q). Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From brainly.ph
When 3.0 kg of water is cooled from 80.0 C to 10.0 C, how much heat How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Use the specific heat equation. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved 2. Calculate how much heat energy 500 grams of water How Much Heat Energy (Q) Did The Water Gain Gizmo Now solve for the specific heat c of copper. Use the specific heat equation to find out how much heat energy the. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the. Water has a. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.numerade.com
SOLVED A hot pot of water is set on the counter to cool. After a few How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4 j/g °c. Specific heat capacity can be calculated using the following equation: How much heat energy q did the water gain? Use the specific heat equation to find out how much heat energy the. Now solve for the specific heat c of copper. Water has a known specific heat capacity of. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From slideplayer.com
CHAPTER 10 ENERGY. ppt download How Much Heat Energy (Q) Did The Water Gain Gizmo Calorimeters can be used to. Water has a known specific heat capacity of 4.184 j/g °c. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. Water has a known specific heat capacity of 4 j/g °c. Use the specific heat equation to find out how. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.nagwa.com
Question Video Calculating the Energy Needed to Heat a Mass by a Known How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4 j/g °c. Use the specific heat equation to find out how much heat energy the water gained ( q). How much heat energy q did the water gain? Use the specific heat equation to find out how much heat energy the. Use the specific heat equation to find out how much. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.doubtnut.com
How much heat energy must be supplied to change36 g of ice at 0^()C to How Much Heat Energy (Q) Did The Water Gain Gizmo Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? When a hot object is placed in the calorimeter, heat energy is transferred from the object. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved Q3 175 g of water was heated from 15 °C to 88 °C. How How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Now solve for the specific heat c of copper. How much heat energy q did the water gain? When a hot object is placed in the calorimeter,. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved QUESTION 9 How much heat energy must be added to How Much Heat Energy (Q) Did The Water Gain Gizmo Calorimeters can be used to. Now solve for the specific heat c of copper. How much heat energy q did the water gain? How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Use the specific heat equation to find out how much heat energy the water gained ( q).. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved How much heat energy is required to convert 14.4 g of How Much Heat Energy (Q) Did The Water Gain Gizmo When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Specific heat capacity can be calculated using the following equation: Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4.184 j/g °c. Calorimeters can be used. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From studylib.net
Heat Equation How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Use the specific heat equation to find out how much heat energy the water gained ( q). Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy q did the water. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From slideplayer.com
Chapter 10 Energy. ppt download How Much Heat Energy (Q) Did The Water Gain Gizmo Calorimeters can be used to. Water has a known specific heat capacity of 4.184 j/g °c. When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. Now solve for the specific heat c of copper. Water has a known specific heat capacity of 4 j/g °c.. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.toppr.com
How much heat energy is gained when 5kg of water at 20^ ^0C I brought How Much Heat Energy (Q) Did The Water Gain Gizmo Water has a known specific heat capacity of 4.184 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. How much heat energy (q) did the water gain (remember water has a known. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.chegg.com
Solved (3) How much heat energy is released by dissolving How Much Heat Energy (Q) Did The Water Gain Gizmo Now solve for the specific heat c of copper. Use the specific heat equation to find out how much heat energy the water gained ( q). Water has a known specific heat capacity of 4 j/g °c. Calorimeters can be used to. How much heat energy q did the water gain? Water has a known specific heat capacity of 4. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From brainly.com
how much heat energy is absorbed when 7 moles of water boils according How Much Heat Energy (Q) Did The Water Gain Gizmo Specific heat capacity can be calculated using the following equation: Water has a known specific heat capacity of 4.184 j/g °c. How much heat energy q did the water gain? Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of 4 j/g °c. Water has a known specific heat capacity of. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From www.toppr.com
How much heat energy is necessary to raise the temperature of 5 kg How Much Heat Energy (Q) Did The Water Gain Gizmo Use the specific heat equation to find out how much heat energy the. Specific heat capacity can be calculated using the following equation: Q = mc∆t in the equation q represents the amount of heat energy gained or lost (in joules), m is the mass of the. When a hot object is placed in the calorimeter, heat energy is transferred. How Much Heat Energy (Q) Did The Water Gain Gizmo.
From ar.inspiredpencil.com
Common Heat Capacities How Much Heat Energy (Q) Did The Water Gain Gizmo Calorimeters can be used to. How much heat energy (q) did the water gain (remember water has a known specific heat capacity of 4.184j/g°c) ? Specific heat capacity can be calculated using the following equation: Use the specific heat equation to find out how much heat energy the. How much heat energy q did the water gain? When a hot. How Much Heat Energy (Q) Did The Water Gain Gizmo.