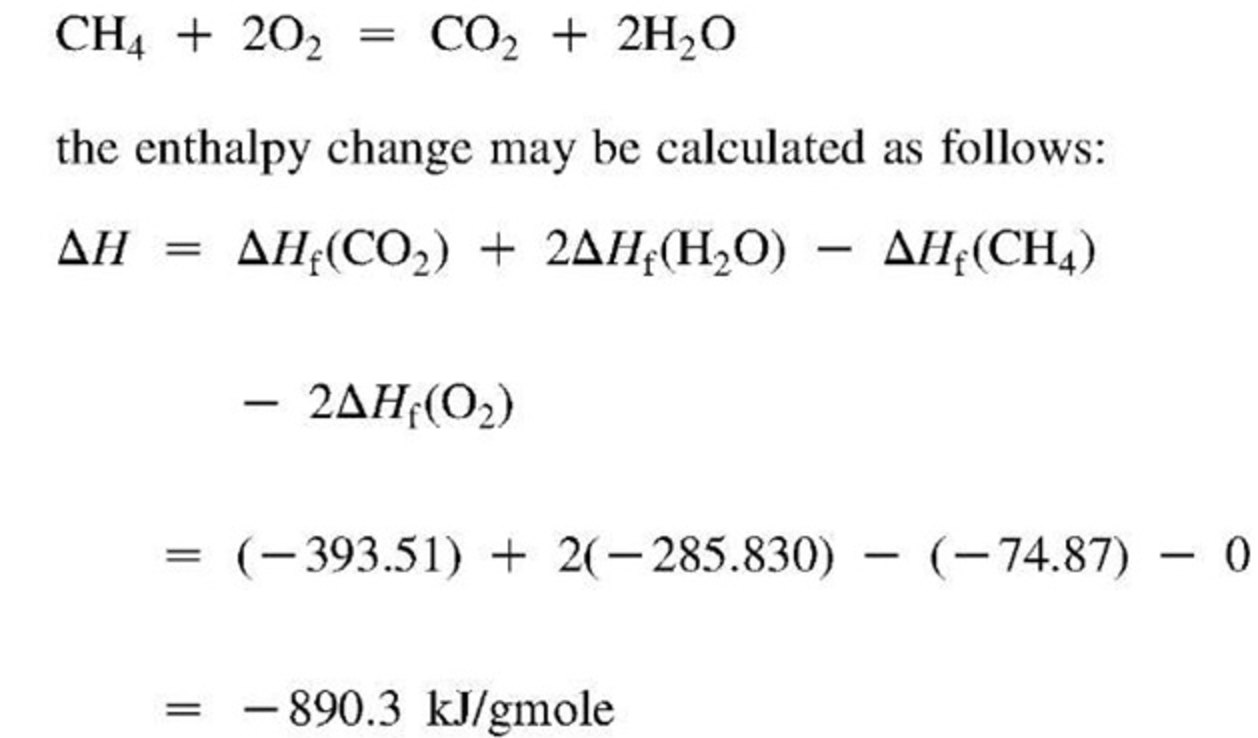Standard Heat Of Formation Chemistry Definition . Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard.
from materiallistgaskell.z21.web.core.windows.net
The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one.
Heat Of Formation Equations
Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance.
From www.chemistrylearner.com
Heat (Enthalpy) of Reaction Definition, Examples, & Formula Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is defined as the change in enthalpy when. Standard Heat Of Formation Chemistry Definition.
From mungfali.com
Standard Enthalpy Of Formation Equation Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is defined as the change in enthalpy when. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT Energy Transformations PowerPoint Presentation, free download Standard Heat Of Formation Chemistry Definition The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy. Standard Heat Of Formation Chemistry Definition.
From www.youtube.com
Standard Enthalpy of Formation and Formation Reactions OpenStax Standard Heat Of Formation Chemistry Definition The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. 193 rows in chemistry and thermodynamics, the standard. Standard Heat Of Formation Chemistry Definition.
From brunofuga.adv.br
Standard Enthalpy Of Formation Definition, Table, Equation, 46 OFF Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is. Standard Heat Of Formation Chemistry Definition.
From slideplayer.com
Chapter 10 “Thermochemistry” ppt download Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Heat of formation, the amount of heat absorbed or evolved when one. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT STANDARD HEAT OF FORMATION ΔH 0 f or ΔH θ f PowerPoint Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard enthalpy of formation is defined as the change in enthalpy. Standard Heat Of Formation Chemistry Definition.
From www.showme.com
11.4Calculating Heat ChangesStandard Heats of Formation (II) Science Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of. Standard Heat Of Formation Chemistry Definition.
From bceweb.org
Heat Of Formation Chart A Visual Reference of Charts Chart Master Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change. Standard Heat Of Formation Chemistry Definition.
From slideplayer.com
Chemistry ppt download Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound. Standard Heat Of Formation Chemistry Definition.
From askfilo.com
CHEMISTRY Example Calculate the standard heat of formation of carbon di.. Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard enthalpy of formation. Standard Heat Of Formation Chemistry Definition.
From lessonlibnurselings.z21.web.core.windows.net
Explain Heats Of Formation Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation. Standard Heat Of Formation Chemistry Definition.
From cepbtpfh.blob.core.windows.net
Standard Heat Of Formation Hydrogen Peroxide at John Ahmed blog Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole. Standard Heat Of Formation Chemistry Definition.
From mungfali.com
Standard Heat Formation Chart Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. The standard enthalpy of formation is defined as the change in enthalpy when one mole. Standard Heat Of Formation Chemistry Definition.
From joizhadcr.blob.core.windows.net
Standard Enthalpy Of Formation Hcl Aq at Denise Lewis blog Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. The standard enthalpy of formation is a measure of the energy released or consumed when one. Standard Heat Of Formation Chemistry Definition.
From materiallistgaskell.z21.web.core.windows.net
Heat Of Formation Equations Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of. Standard Heat Of Formation Chemistry Definition.
From www.youtube.com
5.1 Define Standard State, Enthalpy change of formation and combustion Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of formation. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT THERMOCHEMISTRY PowerPoint Presentation, free download ID4499046 Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is. Standard Heat Of Formation Chemistry Definition.
From rayb78.github.io
Heat Of Formation Chart Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of. Standard Heat Of Formation Chemistry Definition.
From printablevascelomgm.z13.web.core.windows.net
How To Determine The Heat Of Formation Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard enthalpy of formation is defined as the change in enthalpy. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT Thermochemistry PowerPoint Presentation, free download ID314871 Standard Heat Of Formation Chemistry Definition The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. Heat of formation, the amount of heat absorbed. Standard Heat Of Formation Chemistry Definition.
From www.coursehero.com
[Solved] 1. All of the following compounds have a standard heat of Standard Heat Of Formation Chemistry Definition A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. 193 rows in chemistry and thermodynamics, the standard. Standard Heat Of Formation Chemistry Definition.
From www.chem.fsu.edu
CHM1045 Enthalpy Lecture Standard Heat Of Formation Chemistry Definition The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of. Standard Heat Of Formation Chemistry Definition.
From duanerafanan.blogspot.com
DUANE HESS'S LAW Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. Heat of formation, the amount of heat absorbed or evolved when one mole. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT CHAPTER 4 PowerPoint Presentation ID3180471 Standard Heat Of Formation Chemistry Definition A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. 193 rows in chemistry and thermodynamics, the standard. Standard Heat Of Formation Chemistry Definition.
From www.slideserve.com
PPT Hess’s Law PowerPoint Presentation, free download ID6793985 Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the. Standard Heat Of Formation Chemistry Definition.
From materialdbhutchins.z21.web.core.windows.net
Formula Of Heat Of Formation Standard Heat Of Formation Chemistry Definition Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. The standard enthalpy of formation is a measure of the energy. Standard Heat Of Formation Chemistry Definition.
From quizzlistreplevies.z13.web.core.windows.net
Heat Of Formation Equations Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Heat of formation, the amount of heat absorbed or evolved when one mole. Standard Heat Of Formation Chemistry Definition.
From lessonlibnurselings.z21.web.core.windows.net
Heat Of Formation Formula Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard enthalpy of. Standard Heat Of Formation Chemistry Definition.
From classdbmarshall.z13.web.core.windows.net
Heat Of Formation Formula Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under standard. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right). Standard Heat Of Formation Chemistry Definition.
From general.chemistrysteps.com
Standard Enthalpies of Formation Chemistry Steps Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. 193 rows in chemistry. Standard Heat Of Formation Chemistry Definition.
From slideplayer.com
Heat in Changes of State and Calculating Heat of Reaction ppt download Standard Heat Of Formation Chemistry Definition The standard heat of formation \left (\delta h^\text {o}_\text {f} \right) is the enthalpy change associated with the formation of one. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of. Standard Heat Of Formation Chemistry Definition.
From lessonluft.z19.web.core.windows.net
Heat Of Formation Chart Standard Heat Of Formation Chemistry Definition 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. A standard enthalpy of formation δh °f δ h ° f is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from free. The standard heat of formation \left. Standard Heat Of Formation Chemistry Definition.
From www.coursehero.com
[Solved] 1. All of the following compounds have a standard heat of Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard heat of formation \left (\delta h^\text {o}_\text {f} \right). Standard Heat Of Formation Chemistry Definition.
From www.showme.com
Standard heat of formation Science, Chemistry, thermochemistry ShowMe Standard Heat Of Formation Chemistry Definition The standard enthalpy of formation is defined as the change in enthalpy when one mole of a compound is formed from its elements in their. Heat of formation, the amount of heat absorbed or evolved when one mole of a compound is formed from its constituent elements, each substance. The standard enthalpy of formation is a measure of the energy. Standard Heat Of Formation Chemistry Definition.