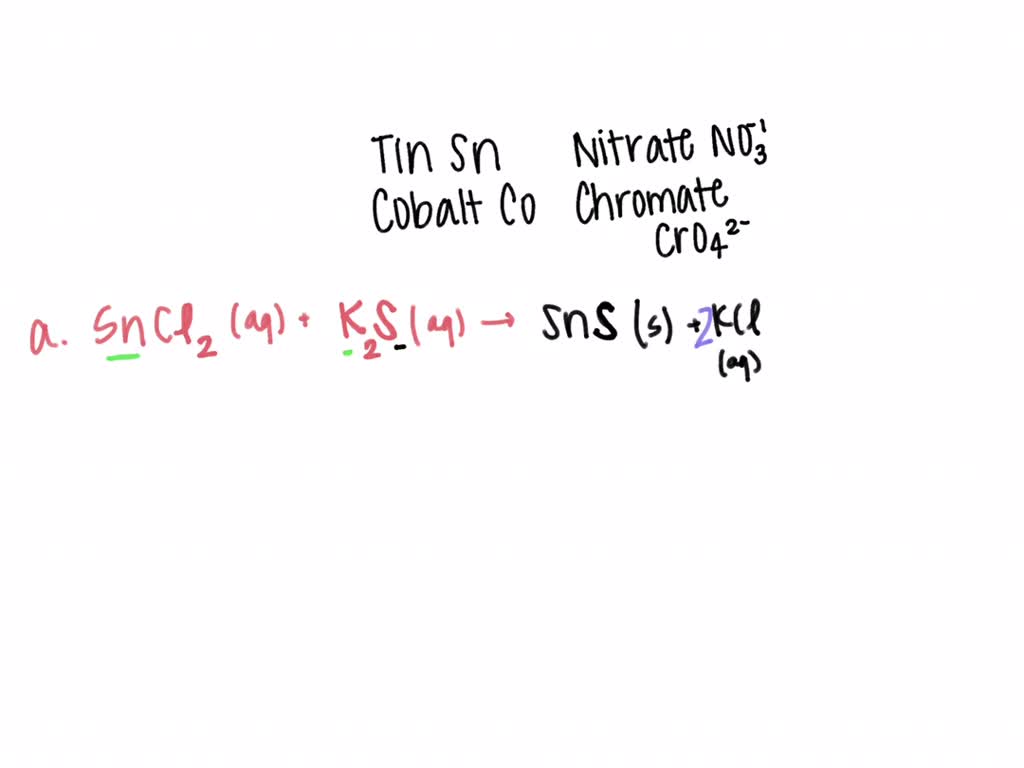Zinc Chloride And Sodium Sulfide Precipitate . a write molecular equation for the following precipitation reaction. Zinc chloride and sodium sulfide are dissolved in water. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. The balanced equation will be calculated along. enter an equation of an ionic chemical equation and press the balance button. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: Sodium chloride, sodium hydroxide, or. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a.
from www.numerade.com
enter an equation of an ionic chemical equation and press the balance button. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Sodium chloride, sodium hydroxide, or. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. The balanced equation will be calculated along. Zinc chloride and sodium sulfide are dissolved in water.
SOLVED Write balanced chemical equation for the reaction of aqueous
Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. Zinc chloride and sodium sulfide are dissolved in water. Sodium chloride, sodium hydroxide, or. enter an equation of an ionic chemical equation and press the balance button. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. The balanced equation will be calculated along. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. a write molecular equation for the following precipitation reaction.
From www.sciencephoto.com
Metal Hydroxide Precipitates Stock Image C027/9452 Science Photo Zinc Chloride And Sodium Sulfide Precipitate Zinc chloride and sodium sulfide are dissolved in water. enter an equation of an ionic chemical equation and press the balance button. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. we are considering the molecular equation for the precipitation reaction, so when we. Zinc Chloride And Sodium Sulfide Precipitate.
From questions.kunduz.com
Complete the table below by deciding whet... Physical Chemistry Zinc Chloride And Sodium Sulfide Precipitate which solution could be used to precipitate the barium ion, ba 2+, in a water sample: zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when. Zinc Chloride And Sodium Sulfide Precipitate.
From www.flickr.com
Synthetic Fluorescent Mineral Sampler Zinc Sulfide (Sphal… Flickr Zinc Chloride And Sodium Sulfide Precipitate a write molecular equation for the following precipitation reaction. Zinc chloride and sodium sulfide are dissolved in water. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m. Zinc Chloride And Sodium Sulfide Precipitate.
From projectopenletter.com
Zinc Sulfate And Iron Ii Bromide Precipitate Printable Form Zinc Chloride And Sodium Sulfide Precipitate zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. adding 10.0 ml of a dilute solution of zinc nitrate to 246. Zinc Chloride And Sodium Sulfide Precipitate.
From exoquepbq.blob.core.windows.net
Potassium Sulfide Zinc Chloride at Gerry Muniz blog Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Zinc chloride and sodium sulfide are dissolved in water. a write molecular equation for the following precipitation reaction. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m. Zinc Chloride And Sodium Sulfide Precipitate.
From www.youtube.com
Does sodium sulfide (Na2S) and zinc nitrate (Zn(NO3)2 precipitate Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. a typical precipitation reaction occurs when an aqueous solution of barium chloride. Zinc Chloride And Sodium Sulfide Precipitate.
From exynedagl.blob.core.windows.net
Chemical Reactions Of Zinc at Kimberly Costanzo blog Zinc Chloride And Sodium Sulfide Precipitate Sodium chloride, sodium hydroxide, or. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2. Zinc Chloride And Sodium Sulfide Precipitate.
From www.sciencephoto.com
Zinc Hydroxide Precipitate Stock Image C027/9442 Science Photo Zinc Chloride And Sodium Sulfide Precipitate The balanced equation will be calculated along. enter an equation of an ionic chemical equation and press the balance button. Zinc chloride and sodium sulfide are dissolved in water. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. which solution could be. Zinc Chloride And Sodium Sulfide Precipitate.
From www.youtube.com
Reaction of Sodium sulphide (Na2S) and zinc sulphate (ZnSO4) YouTube Zinc Chloride And Sodium Sulfide Precipitate zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Zinc chloride and sodium sulfide are dissolved in water. The balanced equation will be calculated along. . Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED CHEMICAL REACTIONS Predicting precipitation nplete the table Zinc Chloride And Sodium Sulfide Precipitate Zinc chloride and sodium sulfide are dissolved in water. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. The balanced equation will. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved (2) Write net ionic equation for each precipitation Zinc Chloride And Sodium Sulfide Precipitate Sodium chloride, sodium hydroxide, or. a write molecular equation for the following precipitation reaction. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g. Zinc Chloride And Sodium Sulfide Precipitate.
From www.youtube.com
Double displacement of ZnSO4 + BaCl2 Zinc sulphate + Barium chloride Zinc Chloride And Sodium Sulfide Precipitate but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. Zinc chloride and sodium sulfide are dissolved in water. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: zinc chloride + sodium sulfate = zinc sulfate + sodium. Zinc Chloride And Sodium Sulfide Precipitate.
From www.mdpi.com
ChemEngineering Free FullText Removal of Metals by Sulphide Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. enter an equation of an ionic chemical equation and press. Zinc Chloride And Sodium Sulfide Precipitate.
From mavink.com
Sodium Hydroxide Ph Chart Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Zinc chloride and sodium sulfide are dissolved in water. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. enter an equation. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Chloride And Sodium Sulfide Precipitate Zinc chloride and sodium sulfide are dissolved in water. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: The balanced equation will be calculated along. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. zinc. Zinc Chloride And Sodium Sulfide Precipitate.
From amudu-gowripalan.blogspot.com
amudu Magical precipitate of Chemistry Zinc Chloride And Sodium Sulfide Precipitate Zinc chloride and sodium sulfide are dissolved in water. Sodium chloride, sodium hydroxide, or. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
A solution of Iron (II) nitrate is poured into a sodium sulfide Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Sodium chloride, sodium hydroxide, or. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. Zinc chloride and sodium sulfide are. Zinc Chloride And Sodium Sulfide Precipitate.
From www.researchgate.net
(PDF) RECOVERY OF SILVER FROM CYANIDE LEACH SOLUTIONS OF A PYRITIC GOLD Zinc Chloride And Sodium Sulfide Precipitate The balanced equation will be calculated along. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. a write molecular equation for the following precipitation reaction. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is. Zinc Chloride And Sodium Sulfide Precipitate.
From brainly.com
PLEASE HELP! STAT! When aqueous solutions of cadmium chloride and Zinc Chloride And Sodium Sulfide Precipitate The balanced equation will be calculated along. a write molecular equation for the following precipitation reaction. Zinc chloride and sodium sulfide are dissolved in water. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a typical precipitation reaction occurs when an aqueous. Zinc Chloride And Sodium Sulfide Precipitate.
From www.youtube.com
Copper (II) Sulfate + Sodium Carbonate = (Double Displacement with Zinc Chloride And Sodium Sulfide Precipitate a write molecular equation for the following precipitation reaction. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Sodium chloride, sodium hydroxide, or. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved solution A solution B Does a precipitate form when A Zinc Chloride And Sodium Sulfide Precipitate enter an equation of an ionic chemical equation and press the balance button. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. Sodium chloride, sodium hydroxide, or. Zinc chloride and sodium sulfide are dissolved in water. we are considering the molecular equation for the. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Chloride And Sodium Sulfide Precipitate a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. a write molecular equation for the following precipitation reaction. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. which solution could be used. Zinc Chloride And Sodium Sulfide Precipitate.
From www.researchgate.net
(PDF) Temperature Dependent Synthesis of Zinc Sulphide (ZnS Zinc Chloride And Sodium Sulfide Precipitate a write molecular equation for the following precipitation reaction. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. Sodium chloride, sodium hydroxide, or. Zinc chloride and sodium sulfide are dissolved in water. we are considering the molecular equation for the precipitation reaction,. Zinc Chloride And Sodium Sulfide Precipitate.
From userdatarheumatics.z21.web.core.windows.net
Chemical Equation For Salt Dissolved In Water Zinc Chloride And Sodium Sulfide Precipitate The balanced equation will be calculated along. Sodium chloride, sodium hydroxide, or. Zinc chloride and sodium sulfide are dissolved in water. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. a write molecular equation for the following precipitation reaction. enter an equation of. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved Complete The Table Below By Deciding Whether A Pre... Zinc Chloride And Sodium Sulfide Precipitate a write molecular equation for the following precipitation reaction. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. we are considering. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED Decide whether a precipitate forms. What is the empirical Zinc Chloride And Sodium Sulfide Precipitate a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. Sodium chloride, sodium hydroxide, or. The balanced equation will be calculated along. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. a write molecular equation. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved Predicting precipitation Complete the table below by Zinc Chloride And Sodium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. Sodium chloride, sodium hydroxide, or. enter an equation of an ionic chemical equation and press the balance button. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with. Zinc Chloride And Sodium Sulfide Precipitate.
From www.shutterstock.com
79 Residual Volume Images, Stock Photos & Vectors Shutterstock Zinc Chloride And Sodium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. enter an equation of an ionic chemical equation and press the balance button. which solution could be used to precipitate the barium ion, ba 2+, in a water sample: we are considering. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED Write balanced chemical equation for the reaction of aqueous Zinc Chloride And Sodium Sulfide Precipitate which solution could be used to precipitate the barium ion, ba 2+, in a water sample: Sodium chloride, sodium hydroxide, or. zinc chloride + sodium sulfate = zinc sulfate + sodium chloride zncl2 + na2so4 = znso4 + nacl is a double displacement. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed. Zinc Chloride And Sodium Sulfide Precipitate.
From woelen.homescience.net
Science made alive Chemistry/Solutions Zinc Chloride And Sodium Sulfide Precipitate a write molecular equation for the following precipitation reaction. The balanced equation will be calculated along. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED Complete the table below by deciding whether precipitate forms Zinc Chloride And Sodium Sulfide Precipitate enter an equation of an ionic chemical equation and press the balance button. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. a write molecular equation for the following precipitation reaction. The balanced equation will be calculated along. adding 10.0 ml of a dilute solution of. Zinc Chloride And Sodium Sulfide Precipitate.
From www.chegg.com
Solved Complete the table below by deciding whether a Zinc Chloride And Sodium Sulfide Precipitate adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. a write molecular equation for the following precipitation reaction. but when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a. we. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED Does precipitate form when A and B are mixed? empirical formula Zinc Chloride And Sodium Sulfide Precipitate The balanced equation will be calculated along. we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. Zinc chloride and sodium sulfide are dissolved in water. enter an equation of an ionic chemical equation and press the balance button. a write molecular equation for. Zinc Chloride And Sodium Sulfide Precipitate.
From www.numerade.com
SOLVED The substance that would be expected to form a precipitate Zinc Chloride And Sodium Sulfide Precipitate Sodium chloride, sodium hydroxide, or. a write molecular equation for the following precipitation reaction. The balanced equation will be calculated along. Zinc chloride and sodium sulfide are dissolved in water. adding 10.0 ml of a dilute solution of zinc nitrate to 246 ml of 2.00 m sodium sulfide produced 0.279 g of a precipitate. enter an equation. Zinc Chloride And Sodium Sulfide Precipitate.
From www.coursehero.com
[Solved] 5. Precipitation Reaction. Sodium sulfide is commonly used to Zinc Chloride And Sodium Sulfide Precipitate we are considering the molecular equation for the precipitation reaction, so when we see this precipitation reaction, so when we look at this. a typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. but when two ions find each other that form an insoluble compound, they suddenly fall. Zinc Chloride And Sodium Sulfide Precipitate.