The Aluminum Atom Gains Electrons To Form An Ion . Learn how atoms become ions by losing or gaining electrons to achieve an octet. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. See examples of cations and anions, and how to predict their charges from the periodic table. This leaves it with a stable electron. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Find out how to predict the electron configurations and charges of ions. Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. Learn how atoms form cations and anions by gaining or losing electrons. See examples of cations and anions, and how to predict their. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms can gain or lose electrons to form ions with positive or negative charges.
from littleeagles.edu.vn
Learn how atoms form cations and anions by gaining or losing electrons. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. See examples of cations and anions, and how to predict their. This leaves it with a stable electron. Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Atoms of group 17 gain one electron and form anions with a 1− charge; Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively.
Aluminum Orbital Diagram, Electron Configuration, And Valence Electrons
The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. Atoms of group 17 gain one electron and form anions with a 1− charge; Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. See examples of cations and anions, and how to predict their charges from the periodic table. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Find out how to predict the electron configurations and charges of ions. Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. This leaves it with a stable electron. Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. See examples of cations and anions, and how to predict their. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Learn how atoms form cations and anions by gaining or losing electrons. Atoms of group 16 gain two electrons and form ions with a 2− charge,.
From www.slideserve.com
PPT Nomenclature PowerPoint Presentation, free download ID7013882 The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons to achieve an octet. Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Find out how to predict the electron configurations and charges of ions. See examples of cations and anions, and how to predict their. Aluminum typically loses 3 electrons to form. The Aluminum Atom Gains Electrons To Form An Ion.
From www.slideserve.com
PPT IV. Chemical Bonding PowerPoint Presentation, free download ID296923 The Aluminum Atom Gains Electrons To Form An Ion The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Learn how atoms become ions by losing or gaining electrons. The Aluminum Atom Gains Electrons To Form An Ion.
From www.numerade.com
SOLVED When aluminum reacts with sulfur to form an ionic compound, each metal atom loses The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms form cations and anions by gaining or losing electrons. Atoms of group 16 gain two electrons and form ions with a 2− charge,. This leaves it with a stable electron. Atoms of group 17 gain one electron and form anions with a 1− charge; Find out how to predict the electron configurations and charges of ions. See. The Aluminum Atom Gains Electrons To Form An Ion.
From aluminumgenjin.blogspot.com
Aluminum Electron Configuration For Aluminum The Aluminum Atom Gains Electrons To Form An Ion See examples of cations and anions, and how to predict their. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. Atoms. The Aluminum Atom Gains Electrons To Form An Ion.
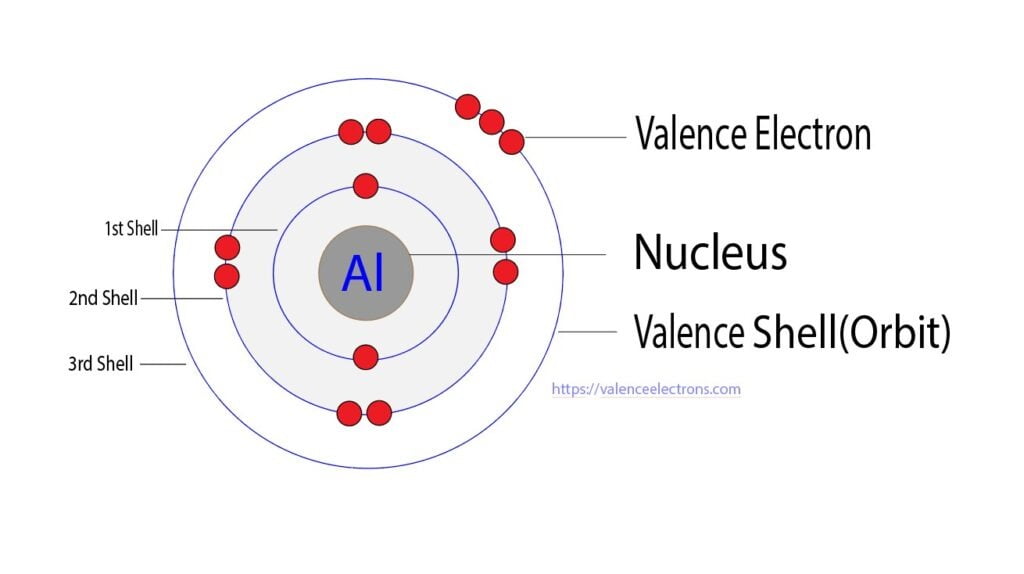
From valenceelectrons.com
Protons, Neutrons, Electrons for Aluminum (Al, Al3+) The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Atoms of group 16 gain two electrons and form ions with a 2− charge,. This leaves it with a stable electron. Aluminum typically loses 3 electrons. The Aluminum Atom Gains Electrons To Form An Ion.
From wirepartfettuccine.z21.web.core.windows.net
Aluminum Electron Dot Diagram The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Atoms of group 16 gain two electrons and form ions with a 2− charge,. See examples of cations and anions, and how to predict their charges from the periodic table. This leaves it with a stable electron. Learn how atoms can gain or. The Aluminum Atom Gains Electrons To Form An Ion.
From elchoroukhost.net
Aluminum Periodic Table Protons Neutrons Electrons Elcho Table The Aluminum Atom Gains Electrons To Form An Ion Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. See examples of cations and anions, and how to predict their. Atoms of group 17 gain one electron and form anions with a 1− charge;. The Aluminum Atom Gains Electrons To Form An Ion.
From guidediagramjeff.z13.web.core.windows.net
Aluminum Dot Diagram The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how atoms form cations and anions by gaining or losing electrons. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Find out how to. The Aluminum Atom Gains Electrons To Form An Ion.
From www.teachoo.com
Neutron Discovery, Difference and more Teachoo Concepts The Aluminum Atom Gains Electrons To Form An Ion Find out how to predict the electron configurations and charges of ions. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. See examples of. The Aluminum Atom Gains Electrons To Form An Ion.
From www.slideserve.com
PPT How do atoms form ions? PowerPoint Presentation, free download ID7021047 The Aluminum Atom Gains Electrons To Form An Ion The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. See examples of cations and anions, and how to predict their charges from the periodic table. See examples of cations and anions, and how to predict their. Learn how atoms of different elements form ions by gaining or losing electrons, and. The Aluminum Atom Gains Electrons To Form An Ion.
From www.numerade.com
SOLVED Jon Formation Element Sodium Nitrogen Aluminum Chlorine Calcium Atomic Electron The Aluminum Atom Gains Electrons To Form An Ion Find out how to predict the electron configurations and charges of ions. This leaves it with a stable electron. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. Study with quizlet and memorize flashcards containing. The Aluminum Atom Gains Electrons To Form An Ion.
From sciencenotes.org
Aluminum Atom Science Notes and Projects The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Learn how atoms become ions by losing or gaining electrons to achieve an. The Aluminum Atom Gains Electrons To Form An Ion.
From slideplayer.com
Ionic Compounds. ppt download The Aluminum Atom Gains Electrons To Form An Ion This leaves it with a stable electron. Learn how atoms form cations and anions by gaining or losing electrons. See examples of cations and anions, and how to predict their charges from the periodic table. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Study with quizlet and memorize flashcards containing terms like 1) in. The Aluminum Atom Gains Electrons To Form An Ion.
From elchoroukhost.net
Aluminum Periodic Table Protons Neutrons And Electrons Elcho Table The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. This leaves it with a stable electron. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Find out how to predict the electron configurations and charges of ions. Aluminum typically loses 3 electrons to form. The Aluminum Atom Gains Electrons To Form An Ion.
From spmchemistry.blog.onlinetuition.com.my
Formation of Ion SPM Chemistry The Aluminum Atom Gains Electrons To Form An Ion Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. See examples of cations and anions, and how to predict their. Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms form cations and anions by gaining or losing electrons. Learn how atoms become ions by losing or gaining. The Aluminum Atom Gains Electrons To Form An Ion.
From www.numerade.com
SOLVED When an aluminum atom (Al) an aluminum ion (Ap+), it three electrons and its The Aluminum Atom Gains Electrons To Form An Ion The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Atoms of group 17 gain one electron and form anions with a 1− charge; Find out how to predict the electron configurations and charges of ions. Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose. The Aluminum Atom Gains Electrons To Form An Ion.
From brainly.com
The aluminum atom electrons to form an ion. The ion that is formed is The Aluminum Atom Gains Electrons To Form An Ion Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. This leaves it with a stable electron. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Learn how atoms of different elements form ions. The Aluminum Atom Gains Electrons To Form An Ion.
From newtondesk.com
Aluminium Al (Element 13) of Periodic Table Elements FlashCards The Aluminum Atom Gains Electrons To Form An Ion Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. See examples of cations and anions, and how to predict their charges from the periodic table. Aluminum typically loses 3 electrons to form a positive ion with a 3+. The Aluminum Atom Gains Electrons To Form An Ion.
From www.slideserve.com
PPT Chapter 2 Atoms, Molecules, and Ions PowerPoint Presentation, free download ID5479793 The Aluminum Atom Gains Electrons To Form An Ion This leaves it with a stable electron. Atoms of group 16 gain two electrons and form ions with a 2− charge,. See examples of cations and anions, and how to predict their charges from the periodic table. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Study with quizlet and. The Aluminum Atom Gains Electrons To Form An Ion.
From www.slideshare.net
Understanding The Chemistry Of Atoms to Ions The Aluminum Atom Gains Electrons To Form An Ion Atoms of group 16 gain two electrons and form ions with a 2− charge,. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. This leaves it with a stable electron. Study with quizlet and memorize. The Aluminum Atom Gains Electrons To Form An Ion.
From www.alamy.com
3d render of atom structure of aluminum isolated over white background Protons are represented The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. This leaves it with a stable electron. Atoms of group 16 gain two electrons and form ions with a 2− charge,. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Find out how. The Aluminum Atom Gains Electrons To Form An Ion.
From spmkimia.onlinetuition.com.my
Pembentukan Ion Positif Nota Ulangkaji Kimia SPM Tingkatan 4/Tingkatan 5 The Aluminum Atom Gains Electrons To Form An Ion Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Atoms of group 16 gain two electrons and form ions with a 2− charge,. This leaves it with a stable electron. Learn how atoms form cations and anions by. The Aluminum Atom Gains Electrons To Form An Ion.
From www.slideserve.com
PPT How do atoms form ions? PowerPoint Presentation, free download ID7021047 The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Find out how to predict the electron configurations and charges of ions. This leaves it with a stable electron. See examples of cations and anions, and how to predict their. Atoms of group 17 gain one electron and form anions with a 1− charge;. The Aluminum Atom Gains Electrons To Form An Ion.
From www.goodscience.com.au
Formation of Ions and Ionic Compounds Good Science The Aluminum Atom Gains Electrons To Form An Ion The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Atoms of group 17 gain one electron and form anions with a 1− charge; Find out how to predict the electron configurations and charges of ions. Learn how atoms of different elements form ions by gaining or losing electrons, and how. The Aluminum Atom Gains Electrons To Form An Ion.
From brokeasshome.com
Periodic Table Which Groups Of Elements Tend To Form Positive Ions The Aluminum Atom Gains Electrons To Form An Ion Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. See examples of cations and anions, and how to predict their. Learn how atoms of different elements form ions by. The Aluminum Atom Gains Electrons To Form An Ion.
From www.benjamin-mills.com
Electron arrangements The Aluminum Atom Gains Electrons To Form An Ion See examples of cations and anions, and how to predict their. Atoms of group 17 gain one electron and form anions with a 1− charge; Atoms of group 16 gain two electrons and form ions with a 2− charge,. Find out how to predict the electron configurations and charges of ions. Learn how atoms of different elements form ions by. The Aluminum Atom Gains Electrons To Form An Ion.
From www.alamy.com
Aluminium (Al). Diagram of the nuclear composition, electron configuration, chemical data, and The Aluminum Atom Gains Electrons To Form An Ion Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. See examples of cations and anions, and how to predict their. The Aluminum Atom Gains Electrons To Form An Ion.
From general.chemistrysteps.com
Electron Configurations of Ions Chemistry Steps The Aluminum Atom Gains Electrons To Form An Ion Atoms of group 16 gain two electrons and form ions with a 2− charge,. Atoms of group 17 gain one electron and form anions with a 1− charge; See examples of cations and anions, and how to predict their. Learn how atoms become ions by losing or gaining electrons to achieve an octet. This leaves it with a stable electron.. The Aluminum Atom Gains Electrons To Form An Ion.
From igcse-chemistry-2017.blogspot.com
IGCSE Chemistry 2017 1.37 Understand How Ions are formed by Electron Loss or Gain The Aluminum Atom Gains Electrons To Form An Ion Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. Learn how atoms of different elements form ions by gaining or losing electrons, and how they combine to form ionic. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Atoms of group 17 gain. The Aluminum Atom Gains Electrons To Form An Ion.
From www.breakingatom.com
Ions of Elements The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms can gain or lose electrons to form ions with positive or negative charges. See examples of cations and anions, and how to predict their. This leaves it with a stable electron. Atoms of group 17 gain one electron and form anions with a 1− charge; Learn how atoms form cations and anions by gaining or losing electrons.. The Aluminum Atom Gains Electrons To Form An Ion.
From www.youtube.com
How to find Protons & Electrons for the Aluminum ion (Al 3+) YouTube The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons to achieve an octet. See examples of cations and anions, and how to predict their. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Aluminum typically loses 3 electrons to form a positive ion with a 3+ charge. Learn how. The Aluminum Atom Gains Electrons To Form An Ion.
From periodictable.me
Aluminum Valence Electrons Aluminum Valency (Al) with Dot Diagram The Aluminum Atom Gains Electrons To Form An Ion The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Atoms of group 16 gain two electrons and form ions with a 2− charge,. See examples of cations and anions, and how to predict their. See examples of cations and anions, and how to predict their charges from the periodic table.. The Aluminum Atom Gains Electrons To Form An Ion.
From littleeagles.edu.vn
Aluminum Orbital Diagram, Electron Configuration, And Valence Electrons The Aluminum Atom Gains Electrons To Form An Ion Atoms of group 17 gain one electron and form anions with a 1− charge; See examples of cations and anions, and how to predict their. Find out how to predict the electron configurations and charges of ions. The charge of aluminum ion is +3 or 3+, and it is formed by losing three electrons from an. Learn how atoms become. The Aluminum Atom Gains Electrons To Form An Ion.
From valenceelectrons.com
Protons, Neutrons, Electrons for Aluminum (Al, Al3+) The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms become ions by losing or gaining electrons, and how to name and represent them. Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. Learn how atoms can gain or lose electrons to form ions with positive or negative charges. Learn how atoms form cations and. The Aluminum Atom Gains Electrons To Form An Ion.
From slideplayer.com
Notes Ionic Bonds 1. Key Concept Ionic bonds form when electrons are ppt download The Aluminum Atom Gains Electrons To Form An Ion Learn how atoms form cations and anions by gaining or losing electrons. Find out how to predict the electron configurations and charges of ions. Learn how atoms become ions by losing or gaining electrons to achieve an octet. Study with quizlet and memorize flashcards containing terms like 1) in ionic compounds, _____ lose their valence electrons to form positively. The. The Aluminum Atom Gains Electrons To Form An Ion.