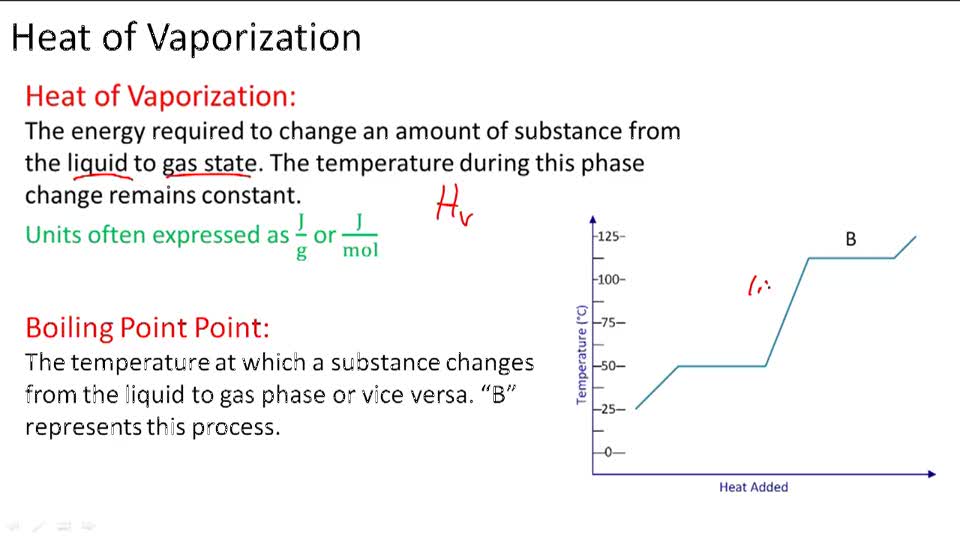
Standard Heat Of Vaporization . The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. It is sometimes called enthalpy of vaporization or latent heat of vaporization.
from www.ck12.org
The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change.
Heats of Vaporization and Condensation CK12 Foundation
Standard Heat Of Vaporization Water has a heat of vaporization value of 40.65 kj/mol. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. Water has a heat of vaporization value of 40.65 kj/mol.
From www.youtube.com
ALEKS Calculating Enthalpy of Vaporization from Vapor Pressure YouTube Standard Heat Of Vaporization The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the. Standard Heat Of Vaporization.
From www.youtube.com
Boiling Point from Heat of Vaporization (Example) YouTube Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. Water has a heat of vaporization value of 40.65 kj/mol. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization.. Standard Heat Of Vaporization.
From www.numerade.com
SOLVEDThe standard enthalpy change of vaporization, Δvap H^θ, of Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of hydrogen bonding present between water molecules, a high input. Standard Heat Of Vaporization.
From www.youtube.com
Calculating Heat of vaporization YouTube Standard Heat Of Vaporization The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called. Standard Heat Of Vaporization.
From education-portal.com
Heat of Vaporization Definition & Equation Video & Lesson Transcript Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat which. Standard Heat Of Vaporization.
From digital.library.unt.edu
Vapor pressures and calculated heats of vaporization of concentrated Standard Heat Of Vaporization The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called. Standard Heat Of Vaporization.
From www.slideserve.com
PPT Chapter 13 PowerPoint Presentation, free download ID3206797 Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. Water has a heat of vaporization value of 40.65 kj/mol. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance. Standard Heat Of Vaporization.
From www.ck12.org
Heat of vaporization Example 1 ( Video ) Chemistry CK12 Foundation Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat of vaporization (also called the enthalpy of vaporization). Standard Heat Of Vaporization.
From engineerexcel.com
Heat of Vaporization Explained EngineerExcel Standard Heat Of Vaporization Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. It is sometimes. Standard Heat Of Vaporization.
From www.researchgate.net
Standard enthalpy of vaporization at 298.15 K as a function of the Standard Heat Of Vaporization The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. Water has a heat of vaporization value of 40.65 kj/mol. It is sometimes. Standard Heat Of Vaporization.
From www.researchgate.net
Standard enthalpy of vaporization at 298.15 K as a function of the Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas. Standard Heat Of Vaporization.
From www.vrogue.co
Latent Heat Of Vaporization And Fusion Definition Tea vrogue.co Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The heat which a solid absorbs when it melts is. Standard Heat Of Vaporization.
From www.researchgate.net
Standardstate specific heat ratio of carbon dioxide and water vapor Standard Heat Of Vaporization The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. The molar heat of vaporization \(\left(. Standard Heat Of Vaporization.
From www.slideserve.com
PPT Thermochemistry PowerPoint Presentation, free download ID6319219 Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance. Standard Heat Of Vaporization.
From www.slideserve.com
PPT 2 Combustion and Thermochemistry PowerPoint Presentation, free Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. Water has a heat of vaporization value of 40.65 kj/mol. As a result of the network. Standard Heat Of Vaporization.
From engineerexcel.com
Heat of Vaporization Explained EngineerExcel Standard Heat Of Vaporization The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance. Standard Heat Of Vaporization.
From www.ck12.org
Heats of Vaporization and Condensation CK12 Foundation Standard Heat Of Vaporization The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The heat of vaporization is the enthalpy change when a unit mass of a substance. Standard Heat Of Vaporization.
From www.chegg.com
Solved 0. Calculate the standard heat of vaporization, ΔHvap Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat which. Standard Heat Of Vaporization.
From www.toppr.com
Standard enthalpy of vaporization of water at 1 atm pressure and 100^oC Standard Heat Of Vaporization The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted. Standard Heat Of Vaporization.
From sciencenotes.org
Clausius Clapeyron Equation Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. Water has a heat. Standard Heat Of Vaporization.
From www.numerade.com
SOLVED The standard enthalpy of vaporization (Hvap) of ethanol Standard Heat Of Vaporization Water has a heat of vaporization value of 40.65 kj/mol. It is sometimes called enthalpy of vaporization or latent heat of vaporization. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization.. Standard Heat Of Vaporization.
From www.numerade.com
SOLVED8. The standard enthalpy of vaporization of a liquid is 5.581 Standard Heat Of Vaporization Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. It is sometimes called enthalpy of vaporization or latent heat of vaporization. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is. Standard Heat Of Vaporization.
From slidecourse.blogspot.com
Latent Heat Of Vaporization Equation Slide Course Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of hydrogen bonding present between water molecules, a high input. Standard Heat Of Vaporization.
From www.slideshare.net
Enthalpy of vaporization of liquid Standard Heat Of Vaporization The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. As a result of the network of. Standard Heat Of Vaporization.
From www.youtube.com
Heat of Vaporization from Vapor Pressure (Example) YouTube Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization is the enthalpy change when a unit mass. Standard Heat Of Vaporization.
From www.coursehero.com
[Solved] The standard enthalpy of vaporization of a liquid is 36.8 kJ Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance. Standard Heat Of Vaporization.
From www.chegg.com
Solved 17. The standard enthalpy of vaporization of Standard Heat Of Vaporization The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. Water has a heat of vaporization value of 40.65 kj/mol. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat of vaporization (also called the enthalpy of vaporization) is the heat. Standard Heat Of Vaporization.
From www.youtube.com
Standard enthalpy of vaporization \( \Delta \mathrm{H}^{\circ} \) f Standard Heat Of Vaporization It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required. Standard Heat Of Vaporization.
From www.researchgate.net
Standard enthalpy of vaporization at 298.15 K as a function of the Standard Heat Of Vaporization The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant temperature and pressure. The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. As a result of the network of hydrogen bonding present between water molecules,. Standard Heat Of Vaporization.
From www.numerade.com
A sample of 2.0 mol CH3OH(g) is condensed isothermally and reversibly Standard Heat Of Vaporization The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. It is sometimes called enthalpy of vaporization or latent heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted. Standard Heat Of Vaporization.
From material-properties.org
Latent Heat of Vaporization of Chemical Elements Material Properties Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually. Standard Heat Of Vaporization.
From chemistrytalk.org
Heat of Fusion Explained ChemTalk Standard Heat Of Vaporization The heat of vaporization (also called the enthalpy of vaporization) is the heat required to induce this phase change. As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. The heat of. Standard Heat Of Vaporization.
From www.chegg.com
Solved 2.(40) a) The standard enthalpy of vaporization of Standard Heat Of Vaporization The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. Water has a heat of vaporization value of 40.65 kj/mol. The heat of vaporization is the enthalpy change when a unit mass of a substance changes its state from liquid to gas at a constant. Standard Heat Of Vaporization.
From www.chegg.com
Solved 1. Explain why the standard enthalpy of vaporization, Standard Heat Of Vaporization The heat which a solid absorbs when it melts is called the enthalpy of fusion or heat of fusion and is usually quoted on a molar. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance is the heat absorbed by one mole of that substance as it is. The heat of vaporization (also called the enthalpy of. Standard Heat Of Vaporization.
From quizlet.com
Using the Clapeyron equation, estimate the enthalpy of vapor Quizlet Standard Heat Of Vaporization As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor, an energy requirement called the heat of vaporization. Water has a heat of vaporization value of 40.65 kj/mol. The molar heat of vaporization \(\left( \delta h_\text{vap} \right)\) of a substance. Standard Heat Of Vaporization.