Properties Of Alkaline Earth Metals Increases Going Down The Group . Reactivity of alkaline earth metals varies going down the group. Summarizing the trend down the group. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. They react with water to produce an alkaline metal hydroxide solution. Density is also affected by the packing structure of the. Group 1 alkali metals explaining reactivity. The reactivity increases down the group as the atomic radii increase there is more shielding. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The nuclear attraction decreases and. The group 1 elements are all soft, reactive metals with low melting points. As you go down the group, the density of the alkali earth metals drops and then increases; The group 1 elements, also known as the alkali metals, all react vigorously with water to.
from www.numerade.com
The group 1 elements are all soft, reactive metals with low melting points. Summarizing the trend down the group. The nuclear attraction decreases and. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. Reactivity of alkaline earth metals varies going down the group. Density is also affected by the packing structure of the. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. The group 1 elements, also known as the alkali metals, all react vigorously with water to. They react with water to produce an alkaline metal hydroxide solution.
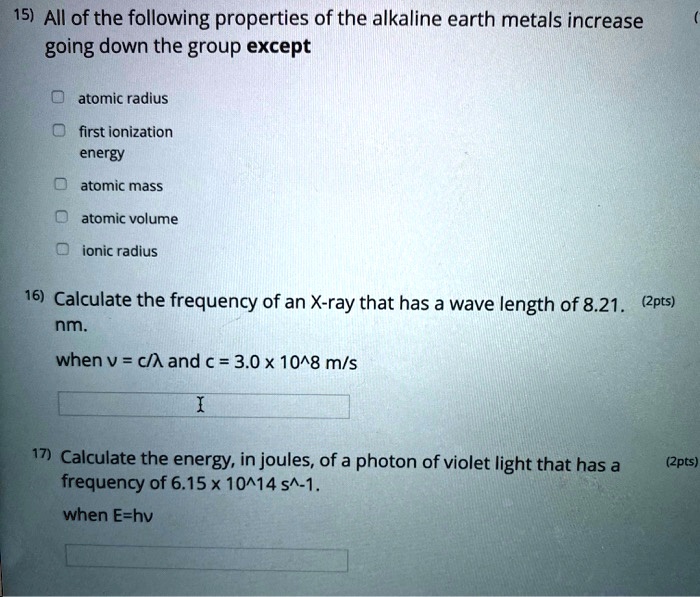
SOLVED 15) All of the following properties of the alkaline earth
Properties Of Alkaline Earth Metals Increases Going Down The Group They react with water to produce an alkaline metal hydroxide solution. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. The group 1 elements are all soft, reactive metals with low melting points. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Density is also affected by the packing structure of the. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. Summarizing the trend down the group. The group 1 elements, also known as the alkali metals, all react vigorously with water to. As you go down the group, the density of the alkali earth metals drops and then increases; They react with water to produce an alkaline metal hydroxide solution. The reactivity increases down the group as the atomic radii increase there is more shielding. Group 1 alkali metals explaining reactivity. Reactivity of alkaline earth metals varies going down the group. The nuclear attraction decreases and.
From elchoroukhost.net
Alkali Metals Periodic Table Properties Elcho Table Properties Of Alkaline Earth Metals Increases Going Down The Group As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Summarizing the trend down the group. Reactivity of alkaline earth metals varies going down the group. The reactivity increases down the group as. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From byjus.com
40.Why does the solubility of alkaline earth metal hydroxides in water Properties Of Alkaline Earth Metals Increases Going Down The Group The group 1 elements, also known as the alkali metals, all react vigorously with water to. The nuclear attraction decreases and. The group 1 elements are all soft, reactive metals with low melting points. Density is also affected by the packing structure of the. As you go down the group, the density of the alkali earth metals drops and then. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From scienceinfo.com
Comparison of properties of Alkali and Alkaline Earth Metals Properties Of Alkaline Earth Metals Increases Going Down The Group As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The group 1 elements, also known as the alkali metals, all react vigorously with water to. Reactivity of alkaline earth metals varies going down the group. Summarizing the trend down the group. Density is also affected. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.thoughtco.com
What Are the Properties of the Alkaline Earth Metals? Properties Of Alkaline Earth Metals Increases Going Down The Group Summarizing the trend down the group. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The reactivity increases down the group as the atomic radii increase there is more shielding. As the metal atoms increase in size, any bonding electron pair becomes farther from the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From thechemistrynotes.com
Trends of the Properties of Group 2 (Alkaline Earth) Metals Properties Of Alkaline Earth Metals Increases Going Down The Group Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Density is also affected by the packing structure of the. Reactivity of alkaline earth metals varies going down the group. They react with water to produce an alkaline metal hydroxide solution. The group 1 elements, also known as the alkali metals, all react vigorously with. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From slideplayer.com
Metals and Non Metals. ppt download Properties Of Alkaline Earth Metals Increases Going Down The Group Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Density is also affected by the packing structure of the. Group 1 alkali metals explaining reactivity. The group 1 elements, also known as the alkali metals, all react vigorously with water to. Summarizing the trend down the group. Reactivity of alkaline earth metals varies going. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Properties Of Alkaline Earth Metals Increases Going Down The Group As you go down the group, the density of the alkali earth metals drops and then increases; As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The reactivity increases down the group as the atomic radii increase there is more shielding. As the metal atoms. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.slideserve.com
PPT ELEMENTS CHEMICAL & PHYSICAL PROPERTIES PowerPoint Presentation Properties Of Alkaline Earth Metals Increases Going Down The Group They react with water to produce an alkaline metal hydroxide solution. Summarizing the trend down the group. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. As you go down the group, the density of the alkali earth metals drops and then. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From byjus.com
Alkaline Earth Metals Occurrence and Extraction,Physical Properties Properties Of Alkaline Earth Metals Increases Going Down The Group As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. Density is also affected by the packing structure of the. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Summarizing the trend down the group. The group 1 elements, also. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.slideserve.com
PPT 33 The Periodic Table PowerPoint Presentation, free download Properties Of Alkaline Earth Metals Increases Going Down The Group Group 1 alkali metals explaining reactivity. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The reactivity increases down the group as the atomic radii increase there is more shielding. Though the nuclear charge increases going down the group (because there are more protons), factors. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.youtube.com
The Alkali Metals Group 1 YouTube Properties Of Alkaline Earth Metals Increases Going Down The Group The nuclear attraction decreases and. The group 1 elements are all soft, reactive metals with low melting points. The group 1 elements, also known as the alkali metals, all react vigorously with water to. They react with water to produce an alkaline metal hydroxide solution. Reactivity of alkaline earth metals varies going down the group. Summarizing the trend down the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.slideserve.com
PPT Chapter 7 Periodic Properties of the Elements PowerPoint Properties Of Alkaline Earth Metals Increases Going Down The Group Summarizing the trend down the group. The reactivity increases down the group as the atomic radii increase there is more shielding. The group 1 elements, also known as the alkali metals, all react vigorously with water to. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. Because. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From slideplayer.com
Grouping the Elements. ppt download Properties Of Alkaline Earth Metals Increases Going Down The Group As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. Summarizing the trend down the group. Group 1 alkali metals explaining reactivity. The nuclear attraction decreases and. They react with water to produce an alkaline metal hydroxide solution. Density is also affected by the packing structure. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From byjus.com
Alkali Metals Properties, Electronic Configuration, Periodic Trends Properties Of Alkaline Earth Metals Increases Going Down The Group Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. Reactivity of alkaline earth metals varies going down the group. The group 1 elements, also known as the alkali metals, all react vigorously with water to. As with the alkali metals, the atomic. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.sliderbase.com
Group 1&2 Presentation Chemistry Properties Of Alkaline Earth Metals Increases Going Down The Group Density is also affected by the packing structure of the. The nuclear attraction decreases and. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. The reactivity increases down the group as the atomic radii increase there is more shielding. As with the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.numerade.com
SOLVED 15) All of the following properties of the alkaline earth Properties Of Alkaline Earth Metals Increases Going Down The Group Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. The reactivity increases down the group as the atomic radii increase there is more shielding. As you go down the group, the density of the alkali earth metals drops and then increases; The nuclear attraction decreases and. Density is also affected by the packing structure. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.savemyexams.co.uk
Trends in Group 2 The Alkaline Earth Metals (2.2.1) AQA A Level Properties Of Alkaline Earth Metals Increases Going Down The Group Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Reactivity of alkaline earth metals varies going down the group. As you go down the group, the density of the alkali earth metals drops and then increases; Density is also affected by the packing structure of the. The group 1 elements, also known as the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From selfstudypoint.in
Group 2 Element Alkaline Earth Metals Properties Of Alkaline Earth Metals Increases Going Down The Group As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The group 1 elements, also known as the alkali metals, all react vigorously with water to. As you go down the group, the density of the alkali earth metals drops and then increases; Because metallic characteristics. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From slideplayer.com
Chapter 12 Section 1 Arranging the Elements Bellwork ppt download Properties Of Alkaline Earth Metals Increases Going Down The Group Summarizing the trend down the group. They react with water to produce an alkaline metal hydroxide solution. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. The nuclear attraction decreases and. The reactivity increases down the group as the atomic radii increase. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.markedbyteachers.com
The group 2 elements are called the alkaline earth metals. The physical Properties Of Alkaline Earth Metals Increases Going Down The Group Density is also affected by the packing structure of the. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. The nuclear attraction decreases and. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. As with the alkali metals, the atomic and. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.expii.com
Alkaline Earth Metals — Overview & Properties Expii Properties Of Alkaline Earth Metals Increases Going Down The Group Density is also affected by the packing structure of the. The nuclear attraction decreases and. The group 1 elements are all soft, reactive metals with low melting points. As you go down the group, the density of the alkali earth metals drops and then increases; Summarizing the trend down the group. The reactivity increases down the group as the atomic. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.youtube.com
Group 2 Alkaline Earth Metals Properties of Matter Chemistry Properties Of Alkaline Earth Metals Increases Going Down The Group As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. They react with water to produce an alkaline metal hydroxide solution. Summarizing the trend down the group. The reactivity increases down the group as the atomic radii increase there is more shielding. The group 1 elements, also known. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.theengineeringprojects.com
Periodic Table of Elements Definition, Groups & Trends The Properties Of Alkaline Earth Metals Increases Going Down The Group Group 1 alkali metals explaining reactivity. The nuclear attraction decreases and. The group 1 elements, also known as the alkali metals, all react vigorously with water to. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. The group 1 elements are all soft, reactive metals with low melting points. They react with water to. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From studylib.net
Alkaline Earth Metals Properties Of Alkaline Earth Metals Increases Going Down The Group Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. Summarizing the trend down the group. Group 1 alkali metals explaining reactivity. They react with water to produce an alkaline metal hydroxide solution. The group 1 elements, also known as the alkali metals,. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.nagwa.com
Question Video Identifying the Property of Alkali Metals From a List Properties Of Alkaline Earth Metals Increases Going Down The Group They react with water to produce an alkaline metal hydroxide solution. Reactivity of alkaline earth metals varies going down the group. The group 1 elements, also known as the alkali metals, all react vigorously with water to. The group 1 elements are all soft, reactive metals with low melting points. The reactivity increases down the group as the atomic radii. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.slideserve.com
PPT Elements and their Properties PowerPoint Presentation, free Properties Of Alkaline Earth Metals Increases Going Down The Group Group 1 alkali metals explaining reactivity. The group 1 elements are all soft, reactive metals with low melting points. The reactivity increases down the group as the atomic radii increase there is more shielding. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. They react. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From brainly.in
Compare the properties of alkali metals with alkaline earth metals Properties Of Alkaline Earth Metals Increases Going Down The Group Density is also affected by the packing structure of the. The reactivity increases down the group as the atomic radii increase there is more shielding. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. As you go down the group, the density. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Properties Of Alkaline Earth Metals Increases Going Down The Group Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. Though the nuclear charge increases going down the group (because there are more protons), factors such as an increased shielding effect and a larger distance between the. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.markedbyteachers.com
The group 2 elements are called the alkaline earth metals. The physical Properties Of Alkaline Earth Metals Increases Going Down The Group The nuclear attraction decreases and. They react with water to produce an alkaline metal hydroxide solution. As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. Group 1 alkali metals explaining reactivity. Density is also affected by the packing structure of the. As the metal atoms. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.sliderbase.com
Element Classes Presentation Chemistry Properties Of Alkaline Earth Metals Increases Going Down The Group Summarizing the trend down the group. They react with water to produce an alkaline metal hydroxide solution. The reactivity increases down the group as the atomic radii increase there is more shielding. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. Density is also affected by the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.britannica.com
Alkalineearth metal chemical element Britannica Properties Of Alkaline Earth Metals Increases Going Down The Group Summarizing the trend down the group. As you go down the group, the density of the alkali earth metals drops and then increases; As with the alkali metals, the atomic and ionic radii of the alkaline earth metals increase smoothly from be to ba, and the ionization. The nuclear attraction decreases and. The reactivity increases down the group as the. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From byjus.com
Alkaline Earth Metals General Characteristics of Oxides, Hallides Properties Of Alkaline Earth Metals Increases Going Down The Group Group 1 alkali metals explaining reactivity. Density is also affected by the packing structure of the. The group 1 elements, also known as the alkali metals, all react vigorously with water to. Reactivity of alkaline earth metals varies going down the group. They react with water to produce an alkaline metal hydroxide solution. Though the nuclear charge increases going down. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From slideplayer.com
Chapter 7 Periodic Properties of the Elements ppt download Properties Of Alkaline Earth Metals Increases Going Down The Group As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. As you go down the group, the density of the alkali earth metals drops and then increases; The reactivity increases down the group as the atomic radii increase there is more shielding. The group 1 elements, also known. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.slideserve.com
PPT Where are the alkali metals? PowerPoint Presentation ID5525447 Properties Of Alkaline Earth Metals Increases Going Down The Group The group 1 elements, also known as the alkali metals, all react vigorously with water to. As the metal atoms increase in size, any bonding electron pair becomes farther from the metal nucleus, and so is less strongly. As you go down the group, the density of the alkali earth metals drops and then increases; Though the nuclear charge increases. Properties Of Alkaline Earth Metals Increases Going Down The Group.
From www.askiitians.com
Alkaline Earth Metals Study Material for IIT JEE askIITians Properties Of Alkaline Earth Metals Increases Going Down The Group As you go down the group, the density of the alkali earth metals drops and then increases; The group 1 elements, also known as the alkali metals, all react vigorously with water to. Summarizing the trend down the group. Because metallic characteristics of alkaline earth metals increases when going down the group, reactivity also. The group 1 elements are all. Properties Of Alkaline Earth Metals Increases Going Down The Group.