What Is Vapour Pressure Of Ethanol . Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some common liquids. 131 rows ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the.
from www.chegg.com
Vapor and saturation pressure for some common liquids. It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. How do you calculate the vapor pressure of ethanol? Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. Uses formula = + [10] log 10 of ethanol vapor pressure vs.
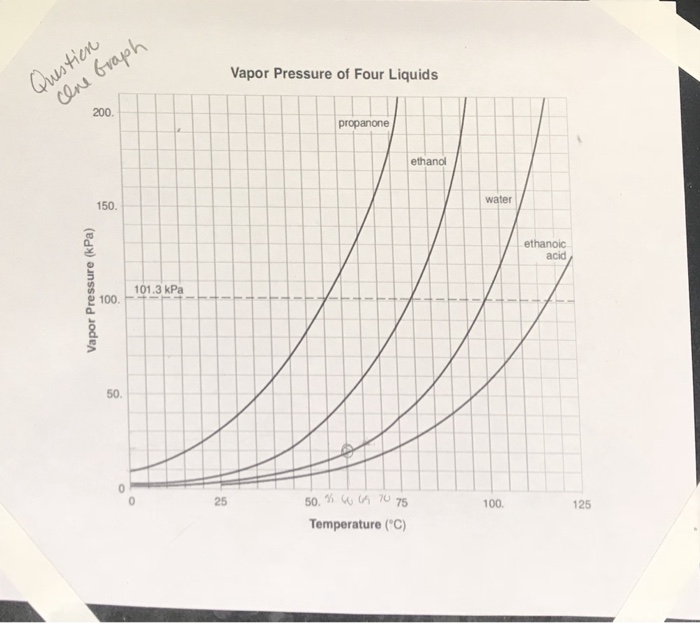
Vapor Pressure of Four Liquids 200 propanone ethanol
What Is Vapour Pressure Of Ethanol It occurs at equilibrium, i.e., when the molecules are both vaporizing. Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. It occurs at equilibrium, i.e., when the molecules are both vaporizing. Uses formula = + [10] log 10 of ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol?
From www.youtube.com
The vapour pressure of ethanol and methanol are `44.0 mm` and `88.0 mm What Is Vapour Pressure Of Ethanol How do you calculate the vapor pressure of ethanol? The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. It occurs at equilibrium, i.e., when the molecules are both vaporizing. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. Vapor. What Is Vapour Pressure Of Ethanol.
From askfilo.com
The vapour pressure of ethanol is 115 torr at 34.9°C. if AHvap of ethanol.. What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. It occurs at equilibrium, i.e., when the molecules are both vaporizing. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
SOLVED The figure below shows the vapor pressures of ether, ethanol What Is Vapour Pressure Of Ethanol The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. 131 rows ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. Uses formula = + [10] log 10 of ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol?. What Is Vapour Pressure Of Ethanol.
From mavink.com
Ethanol Vapor Pressure Chart What Is Vapour Pressure Of Ethanol How do you calculate the vapor pressure of ethanol? Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. Uses formula = + [10] log 10 of ethanol vapor pressure vs. 131 rows ethanol vapor pressure vs. It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of ethanol. What Is Vapour Pressure Of Ethanol.
From www.bartleby.com
Answered What is the vapor pressure of ethanol… bartleby What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
Vapor Pressure of GasolineEthanol Blends Download Scientific Diagram What Is Vapour Pressure Of Ethanol The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. It occurs at equilibrium, i.e., when the molecules are both vaporizing. How do you calculate the. What Is Vapour Pressure Of Ethanol.
From brainly.com
At which temperature is the vapor pressure of ethanol equal to 80. kPa What Is Vapour Pressure Of Ethanol The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. 131 rows ethanol vapor pressure vs. It occurs at equilibrium, i.e.,. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
SOLVED At 20.0*C,the vapor pressure of ethanol is 45.0 torr;and the What Is Vapour Pressure Of Ethanol Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. How do you calculate the vapor pressure of ethanol? Uses formula = + [10] log 10 of ethanol vapor pressure vs. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as. What Is Vapour Pressure Of Ethanol.
From www.chegg.com
Solved Vapor pressure The vapor pressure of ethanol is 115 What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. It. What Is Vapour Pressure Of Ethanol.
From people.umass.edu
Chem112 Fall06 Sec1 PRS Q and A What Is Vapour Pressure Of Ethanol 131 rows ethanol vapor pressure vs. Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Vapor and saturation pressure for some common liquids. It occurs at equilibrium, i.e., when the molecules are both vaporizing. How do you calculate the vapor pressure. What Is Vapour Pressure Of Ethanol.
From mavink.com
Ethanol Vapor Pressure Chart What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor and saturation pressure for some common liquids. How do you. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
SOLVED This graph shows how the vapor pressure of three liquids varies What Is Vapour Pressure Of Ethanol How do you calculate the vapor pressure of ethanol? The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. It occurs. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
Vapor pressure of methanol and ethanol as a function of temperature What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. 131 rows ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol? The vapor pressure of a liquid is defined as the pressure exerted by the molecules that.. What Is Vapour Pressure Of Ethanol.
From chem.libretexts.org
Chapter 11.4 Vapor Pressure Chemistry LibreTexts What Is Vapour Pressure Of Ethanol 131 rows ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some common liquids. Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of ethanol increases with temperature,. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
From the plot of vapor pressures vs temperature above, estimate the What Is Vapour Pressure Of Ethanol Vapor and saturation pressure for some common liquids. How do you calculate the vapor pressure of ethanol? Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Uses formula = + [10] log 10 of ethanol vapor. What Is Vapour Pressure Of Ethanol.
From www.youtube.com
vapour pressure,definition,unit, factors YouTube What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol? It occurs at equilibrium, i.e., when the molecules are both vaporizing. 131 rows ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. Uses formula = + [10] log 10 of ethanol. What Is Vapour Pressure Of Ethanol.
From www.chegg.com
Solved What is the vapor pressure of ethanol (in mmHg ) at What Is Vapour Pressure Of Ethanol Vapor and saturation pressure for some common liquids. How do you calculate the vapor pressure of ethanol? It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. 131 rows ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
The measurements of the saturation vapor pressure of ethanol at low What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. Vapor and saturation pressure for some common liquids. It occurs at equilibrium, i.e., when the molecules are both vaporizing. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. The vapor. What Is Vapour Pressure Of Ethanol.
From byjus.com
A 1 2 mixture (by mole) of ethanol and propanol exerts a vapour What Is Vapour Pressure Of Ethanol It occurs at equilibrium, i.e., when the molecules are both vaporizing. 131 rows ethanol vapor pressure vs. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Uses formula = + [10] log 10 of ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
Vapor pressure of methanol and ethanol as a function of temperature What Is Vapour Pressure Of Ethanol Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. It occurs at equilibrium, i.e., when the molecules are both vaporizing. Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance. What Is Vapour Pressure Of Ethanol.
From askfilo.com
The vapor pressure of ethanol is 100mmHg at 34.9∘C. What is its vapor pre.. What Is Vapour Pressure Of Ethanol Vapor and saturation pressure for some common liquids. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. It. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
The vapor pressure of ethanol vs. the normal boilingpoint temperature What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
SOLVED vapor pressure of ethanol, CH,CH,OH, at 40.0 PCis 17.88 kPa. If What Is Vapour Pressure Of Ethanol It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Uses formula = + [10] log 10 of ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol? The vapor pressure of ethanol increases with temperature, indicating that more ethanol. What Is Vapour Pressure Of Ethanol.
From www.chegg.com
The vapor pressure of ethanol (C_2H_5OH) at 20 degree What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. Uses formula = + [10] log 10 of ethanol vapor pressure vs. It occurs at equilibrium, i.e., when the. What Is Vapour Pressure Of Ethanol.
From mavink.com
Ethanol Vapor Pressure Chart What Is Vapour Pressure Of Ethanol Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor and saturation pressure for some common liquids. Uses formula = + [10] log 10 of ethanol vapor pressure vs. 131 rows ethanol vapor pressure vs. How. What Is Vapour Pressure Of Ethanol.
From moonshinedistiller.com
Controlling Your Heat and Boiler Temperature vs. Vaporization What Is Vapour Pressure Of Ethanol 131 rows ethanol vapor pressure vs. Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol?. What Is Vapour Pressure Of Ethanol.
From guides.hostos.cuny.edu
Chapter 3 Solids and Liquids CHE 110 Introduction to Chemistry What Is Vapour Pressure Of Ethanol 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor and saturation pressure for some common liquids. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol? It occurs at equilibrium,. What Is Vapour Pressure Of Ethanol.
From www.youtube.com
The vapour pressure of ethanol is 115 torr at \( 34.9^{\circ} \math What Is Vapour Pressure Of Ethanol It occurs at equilibrium, i.e., when the molecules are both vaporizing. How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the. What Is Vapour Pressure Of Ethanol.
From sciencenotes.org
Vapor Pressure Definition and How to Calculate It What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some common liquids. The vapor pressure of ethanol increases with temperature, indicating. What Is Vapour Pressure Of Ethanol.
From www.researchgate.net
Vapourliquid equilibrium of ethanolwater showing distillation steps What Is Vapour Pressure Of Ethanol 131 rows ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. How do you calculate the vapor pressure of ethanol? It occurs at equilibrium, i.e., when the molecules. What Is Vapour Pressure Of Ethanol.
From www.chegg.com
Vapor Pressure of Four Liquids 200 propanone ethanol What Is Vapour Pressure Of Ethanol How do you calculate the vapor pressure of ethanol? Vapor and saturation pressure for some common liquids. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Uses formula = +. What Is Vapour Pressure Of Ethanol.
From www.youtube.com
The vapour pressures of ethanol and methanol are `44.5` and `88.7 mm Hg What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. Vapor and saturation pressure for some common liquids. It occurs at equilibrium, i.e., when the molecules are both vaporizing. 131 rows ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol? The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape. What Is Vapour Pressure Of Ethanol.
From www.toppr.com
The vapour pressures of ethanol and methanol are 44.5 mm and 88.7 mm Hg What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of ethanol increases with temperature, indicating that more ethanol molecules escape into the. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. It occurs at equilibrium, i.e., when the molecules are both vaporizing. How do you calculate the. What Is Vapour Pressure Of Ethanol.
From www.numerade.com
SOLVED32. Use the vapour pressure curves illustrated here to answer What Is Vapour Pressure Of Ethanol Uses formula = + [10] log 10 of ethanol vapor pressure vs. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor pressure is the pressure exerted by the vapor molecules of a substance in a closed system. 131 rows ethanol vapor pressure vs. It occurs at equilibrium, i.e., when the molecules are. What Is Vapour Pressure Of Ethanol.
From www.chegg.com
Solved What is the vapor pressure of ethanol (in mmHg) at What Is Vapour Pressure Of Ethanol It occurs at equilibrium, i.e., when the molecules are both vaporizing. The vapor pressure of a liquid is defined as the pressure exerted by the molecules that. Vapor and saturation pressure for some common liquids. Uses formula = + [10] log 10 of ethanol vapor pressure vs. How do you calculate the vapor pressure of ethanol? Vapor pressure is the. What Is Vapour Pressure Of Ethanol.