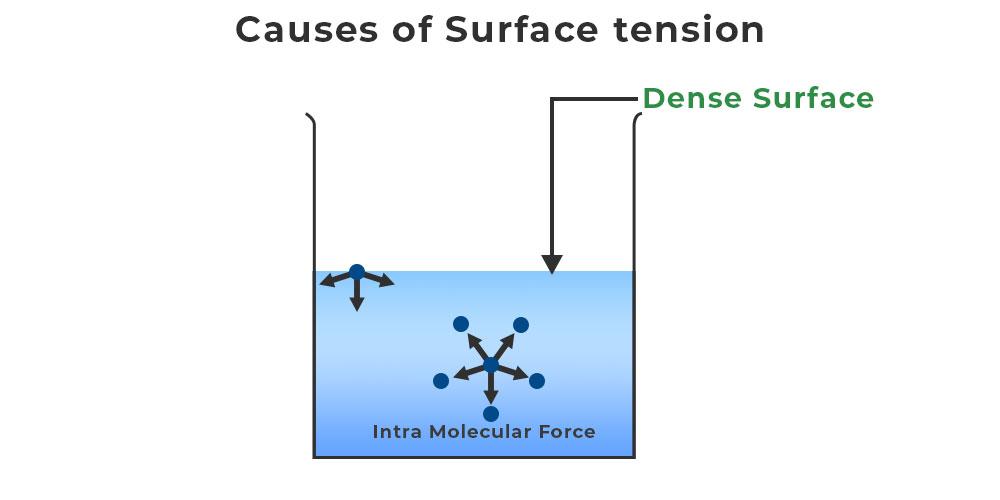
Surface Tension Of Liquid Is 5N/M . Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Effective area = 2 × 0.02 m 2 = 0.04m 2. The surface tension of a liquid is 5 n / m. The molecules on the surface of a liquid are attracted by their neighbors from the. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Surface tension of a liquid is 5 n / m. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about :
from ar.inspiredpencil.com
If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The molecules on the surface of a liquid are attracted by their neighbors from the. The surface tension of a liquid is 5 n / m. Effective area = 2 × 0.02 m 2 = 0.04m 2. Surface tension of a liquid is 5 n / m. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about :
Surface Tension Of Water Diagram
Surface Tension Of Liquid Is 5N/M Surface tension of a liquid is 5 n / m. Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The surface tension of a liquid is 5 n / m. Surface tension of a liquid is 5 n / m. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; The molecules on the surface of a liquid are attracted by their neighbors from the. Effective area = 2 × 0.02 m 2 = 0.04m 2.
From www.slideserve.com
PPT LIQUID SURFACES PowerPoint Presentation, free download ID4456243 Surface Tension Of Liquid Is 5N/M Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : The molecules on the surface of a liquid are attracted by their neighbors from the. Surface tension is a phenomenon that. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
SURFACE TENSION ON LIQUID JET BASIC OF FLUID MECHANICS 24 ANUNIVERSE 22 YouTube Surface Tension Of Liquid Is 5N/M Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Effective area = 2 × 0.02 m 2 = 0.04m 2. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension is a phenomenon that occurs due to the cohesive forces of liquid. Surface Tension Of Liquid Is 5N/M.
From 88guru.com
Viscosity And Surface Tension Definition, Applications and FAQ's 88Guru Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; The molecules on the surface of a liquid are attracted by their neighbors from the. If a film of this liquid is held on a ring of area 0.02 m 2, its. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
Chemistry 8.2b Properties of Liquids Surface Tension and Capillary Action YouTube Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Surface tension of a liquid is 5 n / m.. Surface Tension Of Liquid Is 5N/M.
From www.sciencefacts.net
Surface Tension Definition, Examples, and Unit Surface Tension Of Liquid Is 5N/M Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Effective area = 2 × 0.02 m 2 = 0.04m 2. The surface tension of a liquid is 5 n / m. Surface tension of a liquid is 5 n / m. Calculate the surface tension in flat liquid surfaces in terms of their length. Surface Tension Of Liquid Is 5N/M.
From www.vrogue.co
What Are The Importances Of Surface Tension And Visco vrogue.co Surface Tension Of Liquid Is 5N/M If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : The molecules on the surface of a liquid are attracted by their neighbors from the. The surface tension of a liquid is 5 n / m. Surface tension is a phenomenon that occurs due to the cohesive forces of. Surface Tension Of Liquid Is 5N/M.
From stock.adobe.com
illustration of physics, Surface tension of water, the cohesive forces between water molecules Surface Tension Of Liquid Is 5N/M Surface tension of a liquid is 5 n / m. The molecules on the surface of a liquid are attracted by their neighbors from the. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The surface tension. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
A square frame of side 2 cm is dipped in a liquid. A membrane is formed when taken out, force Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The molecules on the surface of a liquid are attracted by their neighbors. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
The surface tension of a liquid is 5 N/m . If a film is held on a ring of area 0.02 m^2 . its Surface Tension Of Liquid Is 5N/M Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The surface tension of a liquid is 5 n / m. Effective area = 2 × 0.02 m 2 = 0.04m 2. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The molecules on the surface. Surface Tension Of Liquid Is 5N/M.
From www.chegg.com
Solved The surface tension of a liquid is being measured Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The surface tension of. Surface Tension Of Liquid Is 5N/M.
From www.scienceabc.com
Surface Tension Definition, Explanation, Examples And Significance Surface Tension Of Liquid Is 5N/M Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The surface tension of a liquid is 5 n / m. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension is a phenomenon that occurs due to the cohesive forces of liquid. Surface Tension Of Liquid Is 5N/M.
From www.meritnation.com
Surface tension acting on the liquid depends upon mass of the liquid, pressure of the liquid and Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. The molecules on the surface of a liquid are attracted by their neighbors from the. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If its thin film is made in a ring of area 0.02 m 2, then its. Surface Tension Of Liquid Is 5N/M.
From www.slideserve.com
PPT Chapter 15 PowerPoint Presentation, free download ID245553 Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. The surface tension of a liquid is 5 n / m. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Surface tension of a liquid is 5 n / m. If its thin film is made. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
Surface Tension of Water (Mechanics lab.) YouTube Surface Tension Of Liquid Is 5N/M Surface tension of a liquid is 5 n / m. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : The surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface. Surface Tension Of Liquid Is 5N/M.
From www.slideserve.com
PPT PHYSIC PowerPoint Presentation, free download ID4511585 Surface Tension Of Liquid Is 5N/M The molecules on the surface of a liquid are attracted by their neighbors from the. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Surface tension of a liquid is 5 n / m. Effective area = 2 × 0.02 m 2 = 0.04m 2. Surface tension is a phenomenon that. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
Surface Tension on Liquid Jet formula derivation with 3d animation YouTube Surface Tension Of Liquid Is 5N/M If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Surface tension of a liquid is 5 n / m. The surface tension of a liquid is 5 n / m. Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Cohesive forces between. Surface Tension Of Liquid Is 5N/M.
From ar.inspiredpencil.com
Surface Tension Of Water Diagram Surface Tension Of Liquid Is 5N/M If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The surface tension of a liquid is 5 n / m. Effective area = 2 × 0.02 m 2 =. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
A square frame of side 2 cm IZ of side 2 cm is dipped in a liquid. ormed when taken out, force Surface Tension Of Liquid Is 5N/M If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Cohesive forces. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
Surface Tension of given Liquid (Water) by Jaeger's Method YouTube Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : The molecules on the surface of. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
SRTMUN_BScFY_MECHANICSUNIT2_SURFACE TENSION_PART8Surface Tension of a liquid by FERGUSON Surface Tension Of Liquid Is 5N/M The molecules on the surface of a liquid are attracted by their neighbors from the. Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Effective area = 2 × 0.02 m 2 = 0.04m 2. Surface tension of a liquid is 5 n / m. If a film of this liquid is held on. Surface Tension Of Liquid Is 5N/M.
From www.slideserve.com
PPT Surface Tension PowerPoint Presentation, free download ID3106425 Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. The molecules on the surface of a liquid are attracted by their neighbors from the. Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Surface. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
The surface tension of liquid is 5 N/m. If a thin of area 0.04 m^2 is formed on a loop, then its Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : The surface tension of a liquid is 5 n / m. Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. Cohesive forces. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
The surface tension of a liquid is 5 N/m If a film is held on a ring of area 0.02 ms^2 its Surface Tension Of Liquid Is 5N/M Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. The molecules on the surface of a liquid are attracted by their neighbors from the. The surface tension of a liquid is 5 n /. Surface Tension Of Liquid Is 5N/M.
From www.doubtnut.com
[Marathi] The surface tension of a liquid is 0.5 N/m. if a liquid film Surface Tension Of Liquid Is 5N/M The molecules on the surface of a liquid are attracted by their neighbors from the. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : If its thin film is. Surface Tension Of Liquid Is 5N/M.
From www.numerade.com
The surface tension of a liquid is 5 N/m. If a thin film of the area 0.02 m2 is formed on a loop Surface Tension Of Liquid Is 5N/M If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : The surface tension of a liquid is 5 n / m. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Cohesive forces between molecules cause the. Surface Tension Of Liquid Is 5N/M.
From mungfali.com
Surface Tension Chart Surface Tension Of Liquid Is 5N/M Effective area = 2 × 0.02 m 2 = 0.04m 2. Surface tension of a liquid is 5 n / m. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : If a film of this liquid is held on a ring of area 0.02 m 2, its surface. Surface Tension Of Liquid Is 5N/M.
From www.scienceabc.com
Why Does Water Feel Like Concrete When You Belly Flop Into It? » ScienceABC Surface Tension Of Liquid Is 5N/M Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The molecules on the surface of a liquid are attracted by their neighbors from the. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Effective area = 2 × 0.02 m 2 =. Surface Tension Of Liquid Is 5N/M.
From www.slideserve.com
PPT States of Matter PowerPoint Presentation, free download ID6036532 Surface Tension Of Liquid Is 5N/M Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. The molecules on the surface of a liquid are attracted by their neighbors from the. The surface tension of a liquid is 5 n / m. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Effective area. Surface Tension Of Liquid Is 5N/M.
From www.sliderbase.com
Properties of Liquids Presentation Chemistry Surface Tension Of Liquid Is 5N/M If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If its thin film is made in a ring of. Surface Tension Of Liquid Is 5N/M.
From ar.inspiredpencil.com
Surface Tension Of Water Molecules Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. The molecules on the surface of a liquid are attracted by their neighbors from the. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Calculate the surface tension in flat liquid surfaces in terms of their. Surface Tension Of Liquid Is 5N/M.
From www.toppr.com
The surface tension of a liquid is 5 N/m If a film is held on a ring of area 0.02 ms^2 its Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Surface tension is a phenomenon that occurs due to the. Surface Tension Of Liquid Is 5N/M.
From askfilo.com
A liquid has surface tension 5 N/m. If a thin film of this liquid is pres.. Surface Tension Of Liquid Is 5N/M If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : Surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. Calculate the surface tension in flat liquid surfaces in terms. Surface Tension Of Liquid Is 5N/M.
From www.slideserve.com
PPT Physical Pharmacy SURFACE TENSION PowerPoint Presentation, free download ID6578004 Surface Tension Of Liquid Is 5N/M The surface tension of a liquid is 5 n / m. Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If a film of this liquid is held on a ring of area 0.02 m 2, its surface energy is about : If its thin film is made in a ring. Surface Tension Of Liquid Is 5N/M.
From www.youtube.com
Determination of surface tension of Liquid by using stalagmometer information with viva Surface Tension Of Liquid Is 5N/M Surface tension is a phenomenon that occurs due to the cohesive forces of liquid molecules. If its thin film is made in a ring of area 0.02 m 2, then its surface energy will be : Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. If a film of this liquid. Surface Tension Of Liquid Is 5N/M.
From www.numerade.com
SOLVED f an object exerts a force of 1n and its surface tension on the water is measured to be Surface Tension Of Liquid Is 5N/M The molecules on the surface of a liquid are attracted by their neighbors from the. Calculate the surface tension in flat liquid surfaces in terms of their length and external force applied; Surface tension of a liquid is 5 n / m. The surface tension of a liquid is 5 n / m. If its thin film is made in. Surface Tension Of Liquid Is 5N/M.