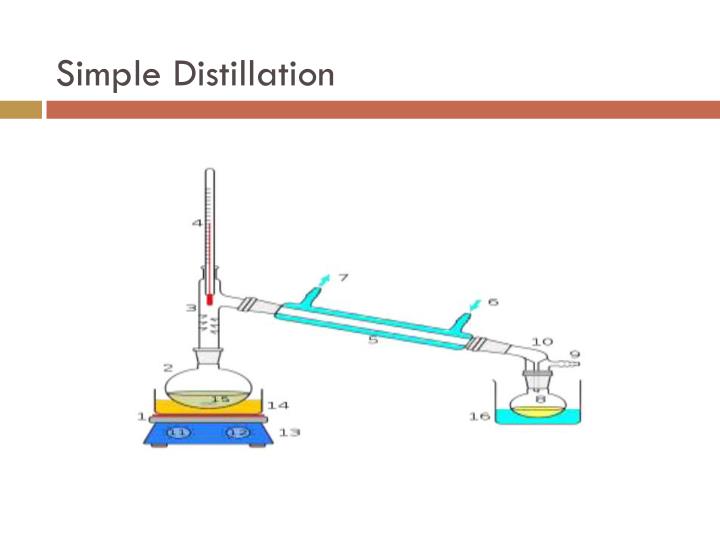
Purpose Of Distillation Is To . Simple distillation is a procedure by which two liquids with different boiling points can be separated. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for commercial purposes. Simple distillation (the procedure outlined below) can be used effectively to.
from www.slideserve.com
Simple distillation is a procedure by which two liquids with different boiling points can be separated. This process is also used for commercial purposes. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Simple distillation (the procedure outlined below) can be used effectively to. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid.
PPT DISTILLATION LAB 2 PowerPoint Presentation ID2871506
Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. This process is also used for commercial purposes. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Simple distillation (the procedure outlined below) can be used effectively to. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture.
From cekkmchw.blob.core.windows.net
Purpose Of A Distillation Unit at Elizabeth Powell blog Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation (the procedure outlined below) can be used effectively to. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for. Purpose Of Distillation Is To.
From www.theengineeringconcepts.com
Types of Distillation The Engineering Concepts Purpose Of Distillation Is To Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation (the procedure outlined below) can be used effectively to. This process. Purpose Of Distillation Is To.
From www.teachoo.com
What are the Properties of Mixtures? Teachoo Concepts Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. This process is also used for. Purpose Of Distillation Is To.
From www.peoplesbourbonreview.com
What is Distillation and How is Liquor Made? The People's Bourbon Review Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for commercial purposes. Simple distillation (the procedure outlined below) can be used. Purpose Of Distillation Is To.
From www.slideserve.com
PPT Separation Techniques Grade 10 Chemistry PowerPoint Presentation Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. This process is also used for commercial purposes. Simple. Purpose Of Distillation Is To.
From www.geeksforgeeks.org
Separation by Distillation Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. The. Purpose Of Distillation Is To.
From www.chegg.com
Solved STEAM DISTILLATION The purpose of this experiment is Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used by chemists. Purpose Of Distillation Is To.
From eartheclipse.com
What is Distillation? Purpose, Types and Various Examples of Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation (the procedure outlined below) can be used effectively to. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used by chemists to purify compounds in solution. Purpose Of Distillation Is To.
From easywayscience78.blogspot.com
Distillation Easy way to learn science Purpose Of Distillation Is To Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for commercial purposes. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation refers to the selective boiling and subsequent condensation. Purpose Of Distillation Is To.
From www.chegg.com
Solved 7. What is the purpose of Distillation? 8. What does Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. The vapors condense on this cool surface, and. Purpose Of Distillation Is To.
From www.slideserve.com
PPT DISTILLATION LAB 2 PowerPoint Presentation ID2871506 Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids,. Purpose Of Distillation Is To.
From sciencing.com
What Is the Purpose of Simple Distillation? Sciencing Purpose Of Distillation Is To Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Simple distillation (the procedure outlined below) can be used effectively to. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a. Purpose Of Distillation Is To.
From zamcopter.web.fc2.com
SIMPLE DISTILLATION Purpose Of Distillation Is To Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation (the procedure outlined below) can be used effectively to. This process is also used for commercial purposes. Distillation is. Purpose Of Distillation Is To.
From glossary.periodni.com
Distillation Chemistry Dictionary & Glossary Purpose Of Distillation Is To Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for commercial purposes. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Simple. Purpose Of Distillation Is To.
From mavink.com
Water Distillation Process Diagram Purpose Of Distillation Is To Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation refers. Purpose Of Distillation Is To.
From exoqonhdf.blob.core.windows.net
Purpose Of Distillation Plant at Joshua Kelley blog Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. This process is also used for commercial purposes. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of. Purpose Of Distillation Is To.
From www.theengineersperspectives.com
How Does Simple Distillation Work? The Engineer's Perspective Purpose Of Distillation Is To This process is also used for commercial purposes. Simple distillation (the procedure outlined below) can be used effectively to. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers. Purpose Of Distillation Is To.
From cekkmchw.blob.core.windows.net
Purpose Of A Distillation Unit at Elizabeth Powell blog Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures. Purpose Of Distillation Is To.
From www.yaclass.in
Simple distillation — lesson. Science CBSE, Class 9. Purpose Of Distillation Is To Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Simple distillation. Purpose Of Distillation Is To.
From www.slideserve.com
PPT What is the purpose and importance of distillation columns Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process. Purpose Of Distillation Is To.
From www.chemicals.co.uk
Distillation Of A Product From A Reaction The Chemistry Blog Purpose Of Distillation Is To Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Simple distillation (the procedure outlined below) can be used effectively to. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir. Purpose Of Distillation Is To.
From dxofngxgr.blob.core.windows.net
How Does The Condenser Work In Simple Distillation at Reba Millard blog Purpose Of Distillation Is To This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation is a procedure by which two liquids with different boiling points can be separated.. Purpose Of Distillation Is To.
From www.vedantu.com
Uses of Distillation Learn Important Terms and Concepts Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation (the procedure outlined below) can be used effectively to. The vapors. Purpose Of Distillation Is To.
From testbook.com
team Distillation Principle, Working, Advantages & Application Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Simple distillation (the procedure outlined below) can be used. Purpose Of Distillation Is To.
From ar.inspiredpencil.com
Distillation Diagram For Kids Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation refers to the selective boiling and subsequent condensation. Purpose Of Distillation Is To.
From www.youtube.com
What is Simple Distillation Separation Methods Chemistry Basics YouTube Purpose Of Distillation Is To This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture.. Purpose Of Distillation Is To.
From www.geeksforgeeks.org
Distillation Definition, Meaning, Principle, Types & Uses Purpose Of Distillation Is To Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Distillation refers. Purpose Of Distillation Is To.
From chemicaltweak.com
6 Types Of Distillation And Definition [Explained In Detail] Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation (the procedure outlined below) can be used effectively to. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Distillation is used by chemists to purify compounds in solution. Purpose Of Distillation Is To.
From fullstudy24.blogspot.com
Chemistry Distillation Full Explanation Purpose Of Distillation Is To Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. This process is also used for commercial purposes. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a. Purpose Of Distillation Is To.
From www.chegg.com
Solved 1. What is the purpose of distillation process? 2. Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented. Purpose Of Distillation Is To.
From www.nagwa.com
Lesson Distillation Nagwa Purpose Of Distillation Is To Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation (the procedure outlined below) can be used effectively to. This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists. Purpose Of Distillation Is To.
From chemistnotes.com
Distillation Definition and Types of Distillation Chemistry Notes Purpose Of Distillation Is To The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a reservoir separated from the original liquid. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Simple distillation (the procedure outlined below) can be used effectively to. This process is also used for commercial purposes. Distillation refers. Purpose Of Distillation Is To.
From www.vedantu.com
Uses of Distillation Learn Important Terms and Concepts Purpose Of Distillation Is To Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Simple distillation is a procedure by which two liquids with different boiling points can be separated. Simple distillation (the procedure outlined below) can be used effectively to. The vapors condense on this cool surface, and the condensed liquid (called the distillate) drips into a. Purpose Of Distillation Is To.
From www.expii.com
Separating Mixtures — Overview & Common Methods Expii Purpose Of Distillation Is To Simple distillation is a procedure by which two liquids with different boiling points can be separated. This process is also used for commercial purposes. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. Distillation. Purpose Of Distillation Is To.
From www.youtube.com
Distillation Definition and Technique Basic Principles and Purpose Of Distillation Is To Simple distillation (the procedure outlined below) can be used effectively to. Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or. Distillation refers to the selective boiling and subsequent condensation of a component in a liquid mixture. Distillation is used by chemists to purify compounds in solution or separating mixtures. Purpose Of Distillation Is To.