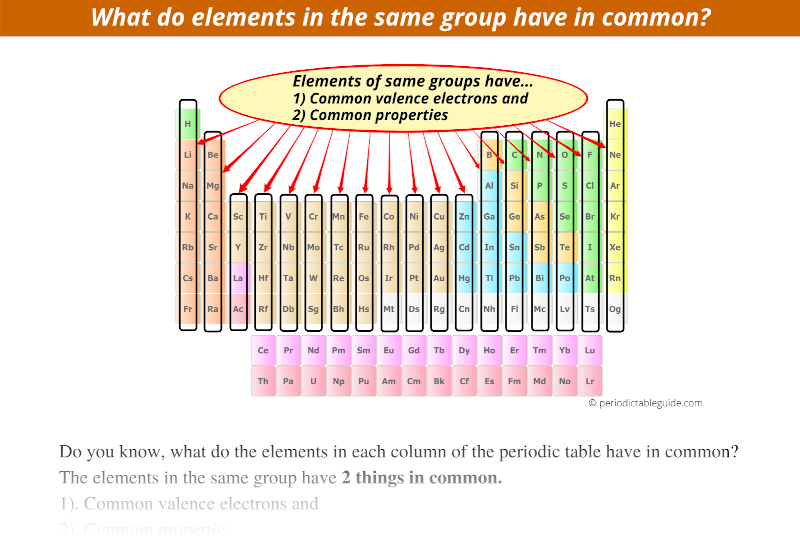
Do Elements In The Same Column Have Similar Properties . As you go across a row, the properties vary gradually from one. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the elements in each family have similar properties. All the members of a family of elements have. From left to right, the atomic number (z) of. on the periodic table, elements that have similar properties are in the same groups (vertical). group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. elements that have similar chemical properties are grouped in columns called groups (or families).
from periodictableguide.com
From left to right, the atomic number (z) of. All the members of a family of elements have. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. on the periodic table, elements that have similar properties are in the same groups (vertical). elements that have similar chemical properties are grouped in columns called groups (or families). the elements in each family have similar properties. As you go across a row, the properties vary gradually from one. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties.
What do elements in the same group have in common? (Fast)
Do Elements In The Same Column Have Similar Properties periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. As you go across a row, the properties vary gradually from one. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. elements that have similar chemical properties are grouped in columns called groups (or families). From left to right, the atomic number (z) of. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. on the periodic table, elements that have similar properties are in the same groups (vertical). the elements in each family have similar properties. the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have.
From byjus.com
Do elements in the same group have similar physical properties or not? Do Elements In The Same Column Have Similar Properties From left to right, the atomic number (z) of. As you go across a row, the properties vary gradually from one. the elements in each family have similar properties. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the periodic table is an arrangement of the elements in. Do Elements In The Same Column Have Similar Properties.
From 2dinaputri.blogspot.com
Columns On The Periodic Table Do Elements In The Same Column Have Similar Properties From left to right, the atomic number (z) of. All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the elements in each family have similar properties. on the periodic table, elements that have similar properties are in the same. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT Why do elements in the same group/family have similar properties? PowerPoint Presentation Do Elements In The Same Column Have Similar Properties the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. elements that have similar chemical properties are grouped in columns called groups (or families). All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical. Do Elements In The Same Column Have Similar Properties.
From mungfali.com
Which Two Elements Have Similar Characteristics Do Elements In The Same Column Have Similar Properties elements that have similar chemical properties are grouped in columns called groups (or families). All the members of a family of elements have. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. on the periodic table, elements that have similar properties are in the same groups (vertical). . Do Elements In The Same Column Have Similar Properties.
From powerpointban.web.fc2.com
Why do elements in the same group have similar chemical properties? Do Elements In The Same Column Have Similar Properties All the members of a family of elements have. on the periodic table, elements that have similar properties are in the same groups (vertical). group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. From left to right, the atomic number (z) of. elements that have similar chemical properties. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT Chapter 3 Atoms and the Periodic Table PowerPoint Presentation ID4793686 Do Elements In The Same Column Have Similar Properties elements that have similar chemical properties are grouped in columns called groups (or families). periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. group 1 elements (lithium, sodium, potassium,. Do Elements In The Same Column Have Similar Properties.
From fr.slideserve.com
PPT Why do elements in the same group/family have similar properties? PowerPoint Presentation Do Elements In The Same Column Have Similar Properties As you go across a row, the properties vary gradually from one. All the members of a family of elements have. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. . Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
Unlocking the Periodic Table of Elements ppt download Do Elements In The Same Column Have Similar Properties group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. As you go across a row, the properties vary gradually from one. All the members of a family of elements have. the elements in each family have similar properties. elements that have similar chemical properties are grouped in columns. Do Elements In The Same Column Have Similar Properties.
From www.youtube.com
why do elements in the same group have similar physical and chemical properties YouTube Do Elements In The Same Column Have Similar Properties the elements in each family have similar properties. As you go across a row, the properties vary gradually from one. All the members of a family of elements have. From left to right, the atomic number (z) of. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. periodic. Do Elements In The Same Column Have Similar Properties.
From orangesciences.blogspot.com
LSS Sec 2 Periodic Table Do Elements In The Same Column Have Similar Properties elements that have similar chemical properties are grouped in columns called groups (or families). the elements in each family have similar properties. From left to right, the atomic number (z) of. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. As you go across a row, the properties. Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
Matter Notes. ppt download Do Elements In The Same Column Have Similar Properties From left to right, the atomic number (z) of. on the periodic table, elements that have similar properties are in the same groups (vertical). All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. elements that have similar chemical properties. Do Elements In The Same Column Have Similar Properties.
From www.amnh.org
How to Read the Periodic Table AMNH Do Elements In The Same Column Have Similar Properties group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. elements that have similar chemical properties are grouped in columns called groups (or families). on the periodic table, elements that. Do Elements In The Same Column Have Similar Properties.
From chemwiki.ucdavis.edu
Periodic Properties of the Elements Chemwiki Do Elements In The Same Column Have Similar Properties periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. elements that have similar chemical properties are grouped in columns called groups (or families). on the periodic table, elements that. Do Elements In The Same Column Have Similar Properties.
From hallegokecastillo.blogspot.com
In the Periodic Table Which Elements Typically Have Similar Properties Do Elements In The Same Column Have Similar Properties As you go across a row, the properties vary gradually from one. the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. elements that have similar chemical properties are grouped in columns called groups (or families). the elements in each family have similar properties. the periodic table is. Do Elements In The Same Column Have Similar Properties.
From byjus.com
Do elements in the same group have similar physical properties or not? Do Elements In The Same Column Have Similar Properties All the members of a family of elements have. As you go across a row, the properties vary gradually from one. the elements in each family have similar properties. elements that have similar chemical properties are grouped in columns called groups (or families). on the periodic table, elements that have similar properties are in the same groups. Do Elements In The Same Column Have Similar Properties.
From byjus.com
Do elements in the same group have similar physical properties or not? Do Elements In The Same Column Have Similar Properties periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. on the periodic table, elements that have similar properties are in the same groups (vertical). the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. group 1. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT The Periodic Table PowerPoint Presentation, free download ID6384783 Do Elements In The Same Column Have Similar Properties on the periodic table, elements that have similar properties are in the same groups (vertical). All the members of a family of elements have. As you go across a row, the properties vary gradually from one. the elements in each family have similar properties. periodic table, in chemistry, the organized array of all the chemical elements in. Do Elements In The Same Column Have Similar Properties.
From sciencenotes.org
Element Families on the Periodic Table Do Elements In The Same Column Have Similar Properties the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. the elements in each family have similar properties. on the periodic table, elements that have similar properties are in the same groups (vertical). All the members of a family of elements have. From left to right,. Do Elements In The Same Column Have Similar Properties.
From adelyn-has-mahoney.blogspot.com
Which Elements Have the Most Similar Chemical Properties AdelynhasMahoney Do Elements In The Same Column Have Similar Properties elements that have similar chemical properties are grouped in columns called groups (or families). All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the elements in each family have similar properties. periodic table, in chemistry, the organized array. Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
What do I need to know? Must ppt download Do Elements In The Same Column Have Similar Properties All the members of a family of elements have. From left to right, the atomic number (z) of. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. As you go across a row, the properties vary gradually from one. elements that have similar chemical properties are grouped in columns. Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
Chapter 19 Periodic table Notes ppt download Do Elements In The Same Column Have Similar Properties the elements in each family have similar properties. on the periodic table, elements that have similar properties are in the same groups (vertical). All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. As you go across a row, the. Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
Classification of Periodic Table ppt download Do Elements In The Same Column Have Similar Properties on the periodic table, elements that have similar properties are in the same groups (vertical). All the members of a family of elements have. elements that have similar chemical properties are grouped in columns called groups (or families). From left to right, the atomic number (z) of. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium). Do Elements In The Same Column Have Similar Properties.
From www.youtube.com
Why do elements in the same group have similar physical and chemical properties? YouTube Do Elements In The Same Column Have Similar Properties group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. As you go across a row, the properties vary gradually from one. All the members of a family of elements have. From left to right, the atomic number (z) of. the periodic table is an arrangement of the elements in. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT Why do elements in the same group/family have similar properties? PowerPoint Presentation Do Elements In The Same Column Have Similar Properties the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the elements in each family have similar properties. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar. Do Elements In The Same Column Have Similar Properties.
From brainly.in
The diagram shows two early attempts at the periodic table. In both cases elements with similar Do Elements In The Same Column Have Similar Properties the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. All the members of a family of elements have. elements that have similar chemical properties are grouped in columns called groups (or families). As you go across a row, the properties vary gradually from one. the. Do Elements In The Same Column Have Similar Properties.
From www.researchgate.net
Periodic table Elements display similar properties at regular periods... Download Scientific Do Elements In The Same Column Have Similar Properties on the periodic table, elements that have similar properties are in the same groups (vertical). From left to right, the atomic number (z) of. All the members of a family of elements have. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the vertical columns on the periodic. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT Introduction to the Periodic Table A Comprehensive Overview PowerPoint Presentation ID Do Elements In The Same Column Have Similar Properties the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. on the periodic table, elements that have similar properties are in the same groups (vertical). periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. All the members of a family. Do Elements In The Same Column Have Similar Properties.
From slideplayer.com
Periodic Table Basics. ppt download Do Elements In The Same Column Have Similar Properties the elements in each family have similar properties. All the members of a family of elements have. the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. . Do Elements In The Same Column Have Similar Properties.
From www.sliderbase.com
Charged Atoms Ions Do Elements In The Same Column Have Similar Properties the vertical columns on the periodic table are called groups or families because of their similar chemical behavior. the elements in each family have similar properties. As you go across a row, the properties vary gradually from one. on the periodic table, elements that have similar properties are in the same groups (vertical). From left to right,. Do Elements In The Same Column Have Similar Properties.
From www.slideserve.com
PPT Why do elements in the same group/family have similar properties? PowerPoint Presentation Do Elements In The Same Column Have Similar Properties on the periodic table, elements that have similar properties are in the same groups (vertical). elements that have similar chemical properties are grouped in columns called groups (or families). group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. All the members of a family of elements have. . Do Elements In The Same Column Have Similar Properties.
From mungfali.com
Periodic Table Of Elements Electronegativity Do Elements In The Same Column Have Similar Properties All the members of a family of elements have. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. the elements in each family have similar properties. the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. . Do Elements In The Same Column Have Similar Properties.
From www.haikudeck.com
Chemistry Periodic Table by James Rowland Do Elements In The Same Column Have Similar Properties periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. From left to right, the atomic number (z) of. group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. the vertical columns on the periodic table are called groups or families. Do Elements In The Same Column Have Similar Properties.
From www.youtube.com
Elements with similar properties tend to be in the same column. Valence electrons! YouTube Do Elements In The Same Column Have Similar Properties the periodic table is an arrangement of the elements in order of their atomic numbers so that elements with similar properties. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. All the members of a family of elements have. on the periodic table, elements that have similar properties. Do Elements In The Same Column Have Similar Properties.
From chemistrymsq10.blogspot.com
Grade10 CHAPTER1 THE PERIODIC TABLE SEMESTER 1 Do Elements In The Same Column Have Similar Properties group 1 elements (lithium, sodium, potassium, rubidium, cesium and francium) have similar physical properties like, metallic luster, softness,. As you go across a row, the properties vary gradually from one. From left to right, the atomic number (z) of. on the periodic table, elements that have similar properties are in the same groups (vertical). elements that have. Do Elements In The Same Column Have Similar Properties.
From periodictableguide.com
What do elements in the same group have in common? (Fast) Do Elements In The Same Column Have Similar Properties on the periodic table, elements that have similar properties are in the same groups (vertical). the elements in each family have similar properties. All the members of a family of elements have. From left to right, the atomic number (z) of. the vertical columns on the periodic table are called groups or families because of their similar. Do Elements In The Same Column Have Similar Properties.