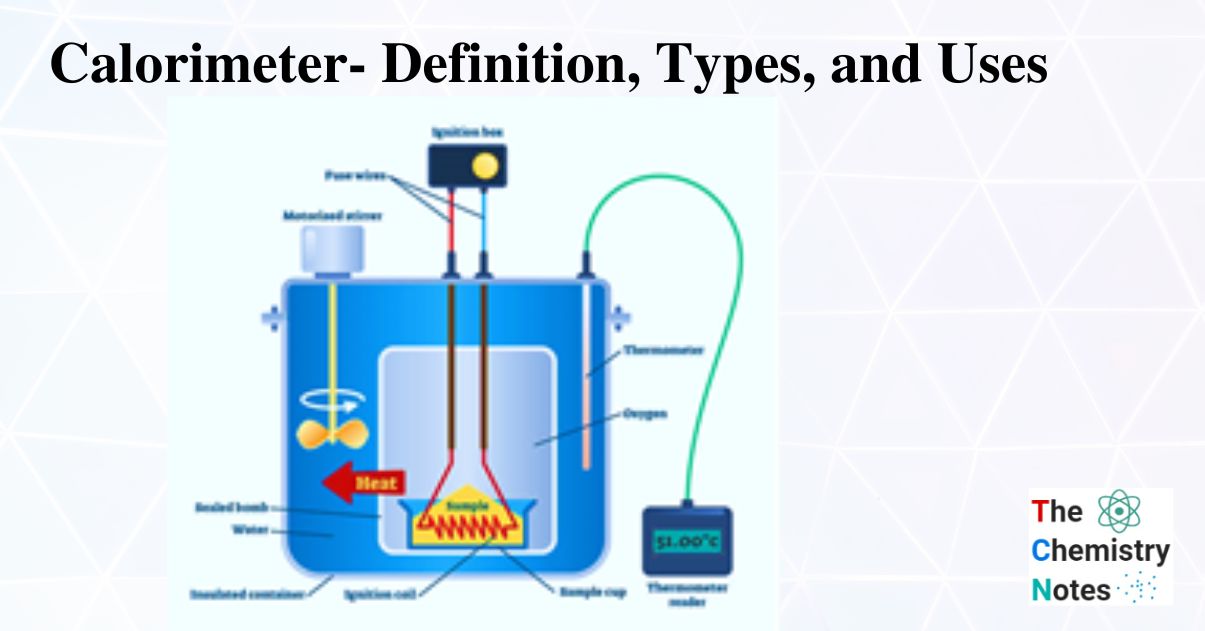
Why Is Water Used In A Calorimeter . calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). that is, the calorimeter constant is the calorimeter's heat capacitance. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. Consider dropping a hot object into cold water that has been. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,.
from thechemistrynotes.com
a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. calorimeters are designed to minimize energy exchange between the system being studied and its. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. Consider dropping a hot object into cold water that has been. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. that is, the calorimeter constant is the calorimeter's heat capacitance.
Calorimeter Definition, Types and Uses
Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. Consider dropping a hot object into cold water that has been. that is, the calorimeter constant is the calorimeter's heat capacitance. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. calorimeters are designed to minimize energy exchange between the system being studied and its. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb).
From www.slideserve.com
PPT Calorimetry PowerPoint Presentation, free download ID1875569 Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. a bomb calorimeter is used to measure,. Why Is Water Used In A Calorimeter.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Why Is Water Used In A Calorimeter one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. Consider dropping a hot object into cold water that has been. calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is used to measure, under. Why Is Water Used In A Calorimeter.
From thechemistrynotes.com
Calorimeter Definition, Types and Uses Why Is Water Used In A Calorimeter assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at. Why Is Water Used In A Calorimeter.
From psu.pb.unizin.org
Calorimetry (9.2) Chemistry 110 Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. in more recent calorimeter designs, the whole bomb, pressurized with excess. Why Is Water Used In A Calorimeter.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. water is best to use in a calorimeter because it has a high specific. Why Is Water Used In A Calorimeter.
From saylordotorg.github.io
Calorimetry Why Is Water Used In A Calorimeter calorimeters are designed to minimize energy exchange between the system being studied and its. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded. Why Is Water Used In A Calorimeter.
From www.britannica.com
Calorimeter Definition, Uses, Diagram, & Facts Britannica Why Is Water Used In A Calorimeter Consider dropping a hot object into cold water that has been. calorimeters are designed to minimize energy exchange between the system being studied and its. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. a bomb calorimeter is used to measure, under controlled conditions, the heat. Why Is Water Used In A Calorimeter.
From www.slideserve.com
PPT Energy PowerPoint Presentation, free download ID6317093 Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). in more recent calorimeter designs, the whole bomb, pressurized with excess. Why Is Water Used In A Calorimeter.
From cider.uoregon.edu
Heat of Solution Calorimetry Simulation Dissolving Salts in Water CIDER Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. Consider dropping a hot object into cold water that has been. . Why Is Water Used In A Calorimeter.
From courses.lumenlearning.com
Calorimetry Chemistry I Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. Consider dropping a hot object into cold water that has. Why Is Water Used In A Calorimeter.
From exodnulby.blob.core.windows.net
Lab Calorimetry And Specific Heat Summary at Barbara Bailey blog Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. Consider dropping a hot object into cold water that has been. one type in widespread. Why Is Water Used In A Calorimeter.
From www.sliderbase.com
Basic Thermochemistry Presentation Chemistry Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at. Why Is Water Used In A Calorimeter.
From exoxbgvei.blob.core.windows.net
Calorimeter Laboratory at Clara Carter blog Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. calorimeters are designed to minimize energy exchange between the system being studied and its. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. in more recent calorimeter designs, the whole bomb, pressurized with. Why Is Water Used In A Calorimeter.
From cider.uoregon.edu
Calorimetry Heat Exchange Hot Metal in Cold Water Real and Computer Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. Consider dropping a hot object into cold water that has been. calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in. Why Is Water Used In A Calorimeter.
From kaffee.50webs.com
Lab Calorimetry Why Is Water Used In A Calorimeter one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. assume that the calorimeter absorbs a negligible amount of heat and, because of. Why Is Water Used In A Calorimeter.
From exosbbnfj.blob.core.windows.net
Calorimetry Ap Chemistry at Michael Faust blog Why Is Water Used In A Calorimeter assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. that is, the calorimeter constant is the calorimeter's heat capacitance. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. one type in widespread. Why Is Water Used In A Calorimeter.
From www.chemistrystudent.com
Calorimetry (ALevel) ChemistryStudent Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted. Why Is Water Used In A Calorimeter.
From www.alamy.com
Instruments for doing an experiment with heated water in a calorimeter Why Is Water Used In A Calorimeter calorimeters are designed to minimize energy exchange between the system being studied and its. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a. Why Is Water Used In A Calorimeter.
From eduinput.com
CalorimeterDefinition, History, Construction, Types, And Uses Why Is Water Used In A Calorimeter calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). that is, the calorimeter constant is the calorimeter's heat capacitance. Consider dropping a hot object into cold. Why Is Water Used In A Calorimeter.
From www.thoughtco.com
Calorimeter Definition in Chemistry Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. Consider dropping a hot object into cold water. Why Is Water Used In A Calorimeter.
From www.youtube.com
Principle of Calorimetry YouTube Why Is Water Used In A Calorimeter Consider dropping a hot object into cold water that has been. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. that is, the calorimeter constant is the calorimeter's heat capacitance. calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is. Why Is Water Used In A Calorimeter.
From dxowceosf.blob.core.windows.net
Calorimetry All Formulas at Spencer McSwain blog Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. that is, the calorimeter constant is the calorimeter's heat capacitance. Consider dropping a hot object into cold water that has been. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30. Why Is Water Used In A Calorimeter.
From www.learnable.education
Year 11 Chemistry Practical Investigation Calorimetry Experiment Why Is Water Used In A Calorimeter water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place,. Why Is Water Used In A Calorimeter.
From courses.lumenlearning.com
Calorimetry Introductory Chemistry Lecture & Lab Why Is Water Used In A Calorimeter calorimeters are designed to minimize energy exchange between the system being studied and its. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the. Why Is Water Used In A Calorimeter.
From chem.libretexts.org
12 Calorimetry and Hess's Law (Experiment) Chemistry LibreTexts Why Is Water Used In A Calorimeter assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. calorimeters are designed to minimize energy exchange between the system being studied and its. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. one type in. Why Is Water Used In A Calorimeter.
From www.scienceabc.com
Molar Heat Capacity Definition, Formula, Equation, Calculation Why Is Water Used In A Calorimeter calorimeters are designed to minimize energy exchange between the system being studied and its. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). Consider dropping a hot object into cold water that has been. that is, the calorimeter constant is. Why Is Water Used In A Calorimeter.
From byjus.com
In a calorimeter (water equivalent=40 g) are 200 g of water and 50 g of Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). that is, the calorimeter constant is the calorimeter's heat capacitance. calorimeters are designed to minimize energy exchange between the system being studied and its. in more recent calorimeter designs, the. Why Is Water Used In A Calorimeter.
From studyadvertiser.z21.web.core.windows.net
How To Use A Calorimeter Stepbystep Why Is Water Used In A Calorimeter in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. Consider dropping a hot object into cold water that has been. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. that is, the calorimeter constant. Why Is Water Used In A Calorimeter.
From www.bartleby.com
A 0.500g sample of KCl is added to 50.0 g of water in a calorimeter Why Is Water Used In A Calorimeter Consider dropping a hot object into cold water that has been. in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. assume that the calorimeter absorbs a negligible amount of. Why Is Water Used In A Calorimeter.
From exozspczc.blob.core.windows.net
Calorimeter Experiment Introduction at Jose Evans blog Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). assume that the calorimeter absorbs a negligible amount of heat and, because of the large volume of water, the specific heat of. that is, the calorimeter constant is the calorimeter's heat. Why Is Water Used In A Calorimeter.
From pressbooks.calstate.edu
3.1 Calorimetry Nutrition and Physical Fitness Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. that is, the calorimeter constant is the calorimeter's heat capacitance. . Why Is Water Used In A Calorimeter.
From www.youtube.com
Thermal Properties of Matter Class 11 Physics Calorimetry Principle Why Is Water Used In A Calorimeter that is, the calorimeter constant is the calorimeter's heat capacitance. water is best to use in a calorimeter because it has a high specific heat, however, other liquids such as. calorimeters are designed to minimize energy exchange between the system being studied and its. assume that the calorimeter absorbs a negligible amount of heat and, because. Why Is Water Used In A Calorimeter.
From pathwaystochemistry.com
Calorimetry Pathways to Chemistry Why Is Water Used In A Calorimeter in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. Consider dropping a hot object into cold water that has been. one type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid,. water is best to use. Why Is Water Used In A Calorimeter.
From faqguide.co
What does a calorimeter do? Explained by FAQGuide Why Is Water Used In A Calorimeter in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). one type in widespread use, called a bomb calorimeter, basically consists of an enclosure. Why Is Water Used In A Calorimeter.
From dxofdzrlj.blob.core.windows.net
Types Of Calorimeter at Joanna Johnson blog Why Is Water Used In A Calorimeter a bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb). in more recent calorimeter designs, the whole bomb, pressurized with excess pure oxygen (typically at 30 standard. calorimeters are designed to minimize energy exchange between the system being studied and its.. Why Is Water Used In A Calorimeter.