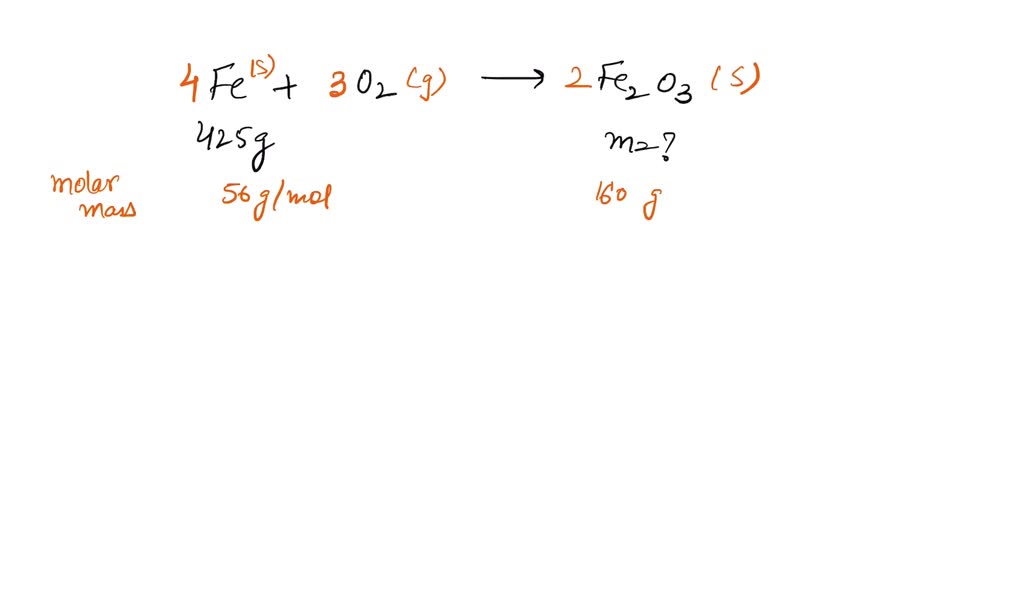Iron Iii Oxide To Iron Using Hydrogen . With a focus on hydrogen acting as a. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Thermodynamic calculations have shown that. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc.
from www.numerade.com
Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. With a focus on hydrogen acting as a. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore.
SOLVED of iron with oxygen gas and water produces iron (III) oxide (a
Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. With a focus on hydrogen acting as a. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite).
From www.bartleby.com
Answered Iron and water react to form iron(III)… bartleby Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Thermodynamic. Iron Iii Oxide To Iron Using Hydrogen.
From www.inoxia.co.uk
Buy Iron (III) Oxide at Inoxia Ltd Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Thermodynamic calculations have shown that. With a focus on hydrogen acting as a. The equilibrium diagram of iron oxides, hydrogen. Iron Iii Oxide To Iron Using Hydrogen.
From www.numerade.com
SOLVED 1. Iron reacts with oxygen to produce iron(III)oxide =. The Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. This study presents a review on the reduction of iron oxides, including. Iron Iii Oxide To Iron Using Hydrogen.
From www.chegg.com
Solved Submit Part C Solid iron III) oxide reacts with Iron Iii Oxide To Iron Using Hydrogen This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. With a focus on hydrogen acting as a. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h. Iron Iii Oxide To Iron Using Hydrogen.
From www.vrogue.co
What Is Formed In The Reaction Between Iron And Oxyge vrogue.co Iron Iii Oxide To Iron Using Hydrogen Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher.. Iron Iii Oxide To Iron Using Hydrogen.
From www.ceramic-glazes.com
IRON OXIDE Iron (III) Oxide Minium Pigments and Stains Iron Iii Oxide To Iron Using Hydrogen Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Thermodynamic calculations have shown that. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the. Iron Iii Oxide To Iron Using Hydrogen.
From www.showme.com
Write the formula for iron (III) oxide and iron (III) sulfate Iron Iii Oxide To Iron Using Hydrogen This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). During the process of iron. Iron Iii Oxide To Iron Using Hydrogen.
From www.slideserve.com
PPT Stoichiometry with a Twist PowerPoint Presentation ID1836706 Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. Thermodynamic calculations have shown that. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. During the process of iron oxide reduction, fe 2 o 3 is first reduced to. Iron Iii Oxide To Iron Using Hydrogen.
From www.youtube.com
How to write the formula for iron (III) oxide YouTube Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Thermodynamic calculations have shown that. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. The equilibrium diagram of iron oxides, hydrogen. Iron Iii Oxide To Iron Using Hydrogen.
From www.researchgate.net
Figure ED 5.10.51 Homological series of iron oxides described by the Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe. Iron Iii Oxide To Iron Using Hydrogen.
From www.numerade.com
SOLVED Iron (III) oxide reacts with carbon monoxide to produce iron Iron Iii Oxide To Iron Using Hydrogen This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. With a focus on hydrogen acting as a. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. The equilibrium diagram of. Iron Iii Oxide To Iron Using Hydrogen.
From hydrogengasgaosube.blogspot.com
Hydrogen Gas Bubbles Of Hydrogen Gas And Aqueous Iron (iii) Iron Iii Oxide To Iron Using Hydrogen Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Thermodynamic calculations have shown that. During the process of. Iron Iii Oxide To Iron Using Hydrogen.
From www.inoxia.co.uk
Buy Iron (III) Oxide at Inoxia Ltd Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. Thermodynamic calculations have. Iron Iii Oxide To Iron Using Hydrogen.
From testbook.com
Oxide Learn Structure, Preparation, Properties & Uses Iron Iii Oxide To Iron Using Hydrogen Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. With a focus on hydrogen acting as a. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h. Iron Iii Oxide To Iron Using Hydrogen.
From www.youtube.com
Redox Reaction Oxidation of Iron (II) to Iron (III) Redox Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. With a focus on hydrogen acting as a. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen.. Iron Iii Oxide To Iron Using Hydrogen.
From inoxia.co.uk
Buy Iron (III) Oxide at Inoxia Ltd Iron Iii Oxide To Iron Using Hydrogen Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. With a focus. Iron Iii Oxide To Iron Using Hydrogen.
From www.numerade.com
SOLVED of iron with oxygen gas and water produces iron (III) oxide (a Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Thermodynamic calculations have shown that. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Therefore, research efforts aim to replace these. Iron Iii Oxide To Iron Using Hydrogen.
From www.sciencephoto.com
Iron(III) Oxide Stock Image C030/8153 Science Photo Library Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Thermodynamic calculations have shown that. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Fundamental differences exist between the oxide/oxide interfaces. Iron Iii Oxide To Iron Using Hydrogen.
From www.chegg.com
Solved Iron reacts with oxygen at high temperatures to form Iron Iii Oxide To Iron Using Hydrogen Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. Thermodynamic calculations have shown that. During the process of iron oxide reduction, fe 2 o 3 is first. Iron Iii Oxide To Iron Using Hydrogen.
From www.youtube.com
How to Write the Formula for Iron (III) hydroxide YouTube Iron Iii Oxide To Iron Using Hydrogen Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. With a focus on hydrogen acting as a. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. During the process of. Iron Iii Oxide To Iron Using Hydrogen.
From cityraven.com
😀 Extraction of iron from its oxides. mining extraction of iron from Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Thermodynamic calculations have shown that. Therefore, research efforts aim to replace these. Iron Iii Oxide To Iron Using Hydrogen.
From www.inoxia.co.uk
Buy Iron (III) Oxide at Inoxia Ltd Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. Thermodynamic calculations have shown that. The equilibrium diagram of iron oxides,. Iron Iii Oxide To Iron Using Hydrogen.
From www.youtube.com
when iron rusts, solid iron reacts with gaseous oxygen to form solid Iron Iii Oxide To Iron Using Hydrogen Thermodynamic calculations have shown that. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. With a focus on hydrogen acting as a. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe. Iron Iii Oxide To Iron Using Hydrogen.
From www.pw.live
Iron III Oxide Formula, Structure, Properties, Uses Iron Iii Oxide To Iron Using Hydrogen Thermodynamic calculations have shown that. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. With a focus on hydrogen acting as a. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is. Iron Iii Oxide To Iron Using Hydrogen.
From ijmmm.ustb.edu.cn
Hydrogenbased direct reduction of iron oxide at 700°C Heterogeneity Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. With a focus on hydrogen acting as a. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. The equilibrium diagram. Iron Iii Oxide To Iron Using Hydrogen.
From www.youtube.com
14 Reactions of Iron oxides (3rd year secondary) YouTube Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. With a focus on hydrogen acting as a. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Thermodynamic calculations have shown. Iron Iii Oxide To Iron Using Hydrogen.
From testbook.com
Iron(III) Oxide Formula Know Its Preparation, Properties & Uses Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Fundamental differences exist. Iron Iii Oxide To Iron Using Hydrogen.
From www.coursehero.com
[Solved] Iron (III) oxide is formed when iron combines with oxygen in Iron Iii Oxide To Iron Using Hydrogen This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. With a focus on hydrogen acting as a. Thermodynamic calculations have shown that. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition. Iron Iii Oxide To Iron Using Hydrogen.
From meaningkosh.com
Iron 3 Oxide MeaningKosh Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen.. Iron Iii Oxide To Iron Using Hydrogen.
From www.inoxia.co.uk
Buy Iron (III) Oxide at Inoxia Ltd Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2,. Iron Iii Oxide To Iron Using Hydrogen.
From www.numerade.com
SOLVED Calculate ΔH° in kJ/mol for the reaction of solid iron (III Iron Iii Oxide To Iron Using Hydrogen Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. Therefore, research efforts aim to replace these reductants with sustainably. Iron Iii Oxide To Iron Using Hydrogen.
From www.sciencephoto.com
Iron (III) oxide in water and in hydrochloric acid Stock Image C036 Iron Iii Oxide To Iron Using Hydrogen During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Thermodynamic calculations have shown that. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). This study presents a review on the reduction of iron oxides, including the influence of. Iron Iii Oxide To Iron Using Hydrogen.
From www.showme.com
Iron III oxide Science, Chemistry, Chemical Bonds ShowMe Iron Iii Oxide To Iron Using Hydrogen The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. With a focus on hydrogen acting as a. This study presents a review on the reduction of iron oxides, including the influence of process parameters, iron oxide, etc. During the process of iron oxide reduction, fe 2 o 3 is first. Iron Iii Oxide To Iron Using Hydrogen.
From www.mdpi.com
Metals Free FullText Hydrogen Ironmaking How It Works Iron Iii Oxide To Iron Using Hydrogen With a focus on hydrogen acting as a. During the process of iron oxide reduction, fe 2 o 3 is first reduced to fe 3 o 4 by h 2, and when the reduction temperature is higher. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite).. Iron Iii Oxide To Iron Using Hydrogen.
From www.numerade.com
SOLVED Calculate the mass of Iron(III) oxide (FezO;) that contains a Iron Iii Oxide To Iron Using Hydrogen Thermodynamic calculations have shown that. Therefore, research efforts aim to replace these reductants with sustainably produced hydrogen. Fundamental differences exist between the oxide/oxide interfaces (e.g., hematite/magnetite and magnetite/wüstite). With a focus on hydrogen acting as a. The equilibrium diagram of iron oxides, hydrogen and carbon monoxide shows the equilibrium gas composition to reduce iron ore. This study presents a review. Iron Iii Oxide To Iron Using Hydrogen.