Does Air Pressure Increase Or Decrease With Temperature . For example, if air pressure increases, the temperature must increase. As the number of molecules increases, they exert more pressure on a surface, and the total. Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. The number of air molecules above a surface determines air pressure. In the case of the ideal. Density is the ratio of the mass to the. I'm trying to reconcile a paradox regarding air pressure and temperature. On the one hand, compressing air heats it up, while. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. Ideal gas law (pv = nrt): It also explains why air gets colder at higher. This law states that the pressure (p) of a fixed. If air pressure decreases, the temperature decreases.
from engineerexcel.com
As the number of molecules increases, they exert more pressure on a surface, and the total. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. If air pressure decreases, the temperature decreases. The number of air molecules above a surface determines air pressure. Density is the ratio of the mass to the. For example, if air pressure increases, the temperature must increase. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. It also explains why air gets colder at higher. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. Ideal gas law (pv = nrt):
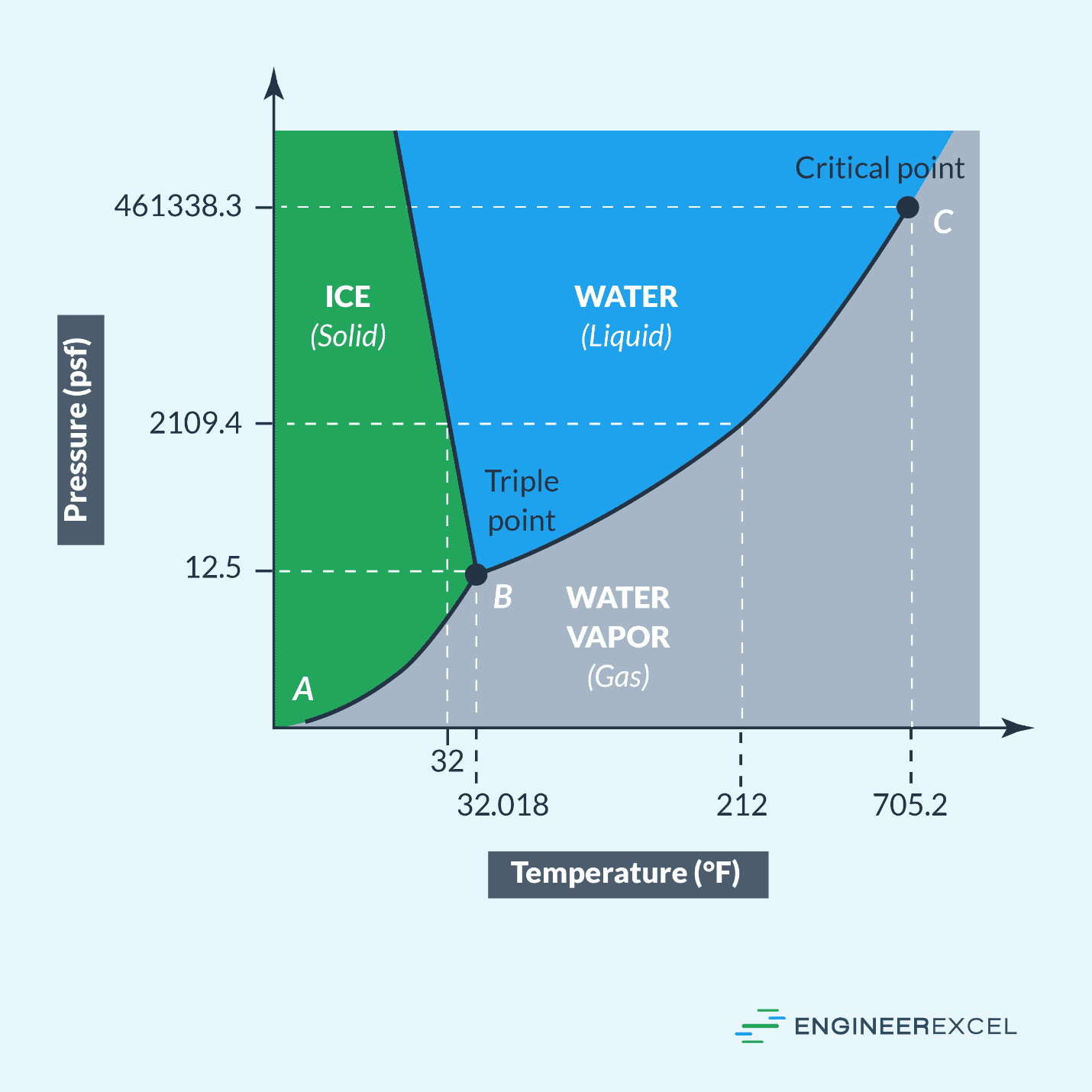
Pressure Temperature Graphs Explained EngineerExcel
Does Air Pressure Increase Or Decrease With Temperature In reality, most compression take place by reducing volume. Density is the ratio of the mass to the. If air pressure decreases, the temperature decreases. As the number of molecules increases, they exert more pressure on a surface, and the total. In the case of the ideal. Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. For example, if air pressure increases, the temperature must increase. This law states that the pressure (p) of a fixed. The number of air molecules above a surface determines air pressure. I'm trying to reconcile a paradox regarding air pressure and temperature. In reality, most compression take place by reducing volume. It also explains why air gets colder at higher. Ideal gas law (pv = nrt):
From www.slideserve.com
PPT Chapter 15 The Atmosphere PowerPoint Presentation, free download Does Air Pressure Increase Or Decrease With Temperature Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. Ideal gas law (pv = nrt): It also explains why air gets colder at higher. In reality, most compression take place by reducing volume. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Equilibrium PowerPoint Presentation, free download ID6271487 Does Air Pressure Increase Or Decrease With Temperature I'm trying to reconcile a paradox regarding air pressure and temperature. As the number of molecules increases, they exert more pressure on a surface, and the total. If air pressure decreases, the temperature decreases. For example, if air pressure increases, the temperature must increase. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. Density. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Unit 4 Phases of Matter (Chapters 1314) PowerPoint Presentation Does Air Pressure Increase Or Decrease With Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. I'm trying to reconcile a paradox regarding air pressure and temperature. This law states that the pressure (p) of a fixed. The number of air molecules above a surface determines air pressure. If air pressure decreases, the temperature. Does Air Pressure Increase Or Decrease With Temperature.
From www.teachoo.com
Effect of Temperature to Change State of Matter Teachoo Science Does Air Pressure Increase Or Decrease With Temperature Density is the ratio of the mass to the. In the case of the ideal. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. This law states that the pressure (p) of a fixed. If air pressure decreases, the temperature decreases. The number of air molecules above. Does Air Pressure Increase Or Decrease With Temperature.
From www.youtube.com
Why temperature decreases with altitude?why_temperature_decreases_with Does Air Pressure Increase Or Decrease With Temperature If air pressure decreases, the temperature decreases. Density is the ratio of the mass to the. I'm trying to reconcile a paradox regarding air pressure and temperature. In the case of the ideal. In reality, most compression take place by reducing volume. On the one hand, compressing air heats it up, while. As the number of molecules increases, they exert. Does Air Pressure Increase Or Decrease With Temperature.
From med.libretexts.org
2.3 Gaseous Exchange Mechanism Medicine LibreTexts Does Air Pressure Increase Or Decrease With Temperature Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. For example, if air pressure increases, the temperature must increase. If air pressure decreases, the temperature decreases. On the one hand,. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Aim What are factors of Air Pressure? PowerPoint Presentation Does Air Pressure Increase Or Decrease With Temperature As the number of molecules increases, they exert more pressure on a surface, and the total. For example, if air pressure increases, the temperature must increase. On the one hand, compressing air heats it up, while. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. Since air pressure is proportional to the density of. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideshare.net
Air pressure and weather Does Air Pressure Increase Or Decrease With Temperature On the one hand, compressing air heats it up, while. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. It also explains why air gets colder at higher. As the number of molecules increases, they exert more pressure on a surface, and the total. If air pressure. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Air Pressure and Wind PowerPoint Presentation, free download ID Does Air Pressure Increase Or Decrease With Temperature I'm trying to reconcile a paradox regarding air pressure and temperature. It also explains why air gets colder at higher. In reality, most compression take place by reducing volume. As the number of molecules increases, they exert more pressure on a surface, and the total. The number of air molecules above a surface determines air pressure. If air pressure decreases,. Does Air Pressure Increase Or Decrease With Temperature.
From pressbooks.bccampus.ca
LABORATORY 2 HEAT AND TEMPERATURE IN THE ATMOSPHERE Physical Does Air Pressure Increase Or Decrease With Temperature In reality, most compression take place by reducing volume. Density is the ratio of the mass to the. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. I'm trying to reconcile a paradox regarding air pressure and temperature. It also explains why air gets colder at higher.. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Air Pressure and Wind PowerPoint Presentation, free download ID Does Air Pressure Increase Or Decrease With Temperature In reality, most compression take place by reducing volume. The number of air molecules above a surface determines air pressure. As the number of molecules increases, they exert more pressure on a surface, and the total. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. In january, when air temperatures average 20.7. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideshare.net
Air pressure Does Air Pressure Increase Or Decrease With Temperature I'm trying to reconcile a paradox regarding air pressure and temperature. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. It also explains why air gets colder at higher. Density is the ratio of the mass to the. Ideal gas law (pv = nrt): In january, when air temperatures average 20.7 c. Does Air Pressure Increase Or Decrease With Temperature.
From www.pinterest.com
Atmosphere characteristics, air pressure Molecule diagram, Earth and Does Air Pressure Increase Or Decrease With Temperature It also explains why air gets colder at higher. In reality, most compression take place by reducing volume. This law states that the pressure (p) of a fixed. If air pressure decreases, the temperature decreases. For example, if air pressure increases, the temperature must increase. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is. Does Air Pressure Increase Or Decrease With Temperature.
From www.acurite.com
Atmospheric Pressure AcuRite Does Air Pressure Increase Or Decrease With Temperature The number of air molecules above a surface determines air pressure. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. If air pressure decreases, the temperature decreases. Ideal gas law (pv = nrt): I'm trying to reconcile a paradox regarding air pressure and temperature. In june, when. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT The Ideal Gas Equation PowerPoint Presentation, free download Does Air Pressure Increase Or Decrease With Temperature As the number of molecules increases, they exert more pressure on a surface, and the total. This law states that the pressure (p) of a fixed. Density is the ratio of the mass to the. If air pressure decreases, the temperature decreases. Since air pressure is proportional to the density of the air, a decrease in density results in a. Does Air Pressure Increase Or Decrease With Temperature.
From www.nesscopressure.com.au
What is air pressure? Nessco Pressure Systems Does Air Pressure Increase Or Decrease With Temperature If air pressure decreases, the temperature decreases. For example, if air pressure increases, the temperature must increase. In reality, most compression take place by reducing volume. Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. In june, when the average air temperature is 27.6c (81.7f), the average. Does Air Pressure Increase Or Decrease With Temperature.
From techiescientist.com
Does Density Change With Temperature? Techiescientist Does Air Pressure Increase Or Decrease With Temperature This law states that the pressure (p) of a fixed. It also explains why air gets colder at higher. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. In reality, most compression take place by reducing volume. As the number of molecules increases, they exert more pressure. Does Air Pressure Increase Or Decrease With Temperature.
From brainly.in
, 1. Does air temperature increase or decrease as you go up in the Does Air Pressure Increase Or Decrease With Temperature On the one hand, compressing air heats it up, while. If air pressure decreases, the temperature decreases. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. Ideal gas law (pv = nrt): For example, if air pressure increases, the temperature must increase. In january, when air temperatures average 20.7 c (69f), the. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Chapter 4 Atmospheric Pressure and Wind PowerPoint Presentation Does Air Pressure Increase Or Decrease With Temperature It also explains why air gets colder at higher. Density is the ratio of the mass to the. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. This law states that the. Does Air Pressure Increase Or Decrease With Temperature.
From climate.ncsu.edu
Structure of the Atmosphere North Carolina Climate Office Does Air Pressure Increase Or Decrease With Temperature If air pressure decreases, the temperature decreases. In reality, most compression take place by reducing volume. On the one hand, compressing air heats it up, while. It also explains why air gets colder at higher. Ideal gas law (pv = nrt): If you had a way to increase pressure with no volume change, then yes, temperature would increase by the. Does Air Pressure Increase Or Decrease With Temperature.
From canvas.santarosa.edu
3.7 Atmospheric Pressure GEOG4 Physical Geography Section 4931 Does Air Pressure Increase Or Decrease With Temperature If air pressure decreases, the temperature decreases. This law states that the pressure (p) of a fixed. In the case of the ideal. I'm trying to reconcile a paradox regarding air pressure and temperature. Density is the ratio of the mass to the. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb.. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Chapter 18 PowerPoint Presentation, free download ID1757364 Does Air Pressure Increase Or Decrease With Temperature If air pressure decreases, the temperature decreases. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. This law states that the pressure (p) of a fixed. In reality, most compression take place by reducing volume. Density is. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Characteristics of the Atmosphere PowerPoint Presentation, free Does Air Pressure Increase Or Decrease With Temperature Density is the ratio of the mass to the. Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. As the number of molecules increases, they exert more pressure on a surface, and the total. On the one hand, compressing air heats it up, while. It also explains. Does Air Pressure Increase Or Decrease With Temperature.
From courses.lumenlearning.com
Layers of the Atmosphere Physical Geography Does Air Pressure Increase Or Decrease With Temperature Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. If air pressure decreases, the temperature decreases. In the case of the ideal. It also explains why air gets colder at higher. If you had a way to increase pressure with no volume change, then yes, temperature would. Does Air Pressure Increase Or Decrease With Temperature.
From www.youtube.com
Pressure Altitude vs Density Altitude Private Pilot Knowledge Test Does Air Pressure Increase Or Decrease With Temperature In the case of the ideal. This law states that the pressure (p) of a fixed. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. If you had a way to increase pressure with no volume change,. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Chapter 9 PowerPoint Presentation ID1275640 Does Air Pressure Increase Or Decrease With Temperature The number of air molecules above a surface determines air pressure. On the one hand, compressing air heats it up, while. In reality, most compression take place by reducing volume. If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. I'm trying to reconcile a paradox regarding air. Does Air Pressure Increase Or Decrease With Temperature.
From www.researchgate.net
1 The atmospheric density and pressure distribution. Download Does Air Pressure Increase Or Decrease With Temperature Density is the ratio of the mass to the. If air pressure decreases, the temperature decreases. For example, if air pressure increases, the temperature must increase. This law states that the pressure (p) of a fixed. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. In reality, most compression take place by reducing volume.. Does Air Pressure Increase Or Decrease With Temperature.
From www.eoas.ubc.ca
UBC ATSC 113 Layers in the Standard Atmosphere Does Air Pressure Increase Or Decrease With Temperature On the one hand, compressing air heats it up, while. Density is the ratio of the mass to the. This law states that the pressure (p) of a fixed. Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. In reality, most compression take place by reducing volume.. Does Air Pressure Increase Or Decrease With Temperature.
From courses.lumenlearning.com
Phase Changes Physics Does Air Pressure Increase Or Decrease With Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. Density is the ratio of the mass to the. I'm trying to reconcile a paradox regarding air pressure and temperature. In the case. Does Air Pressure Increase Or Decrease With Temperature.
From letstalkscience.ca
Weather Temperature Let's Talk Science Does Air Pressure Increase Or Decrease With Temperature In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. The number of air molecules above a surface determines air pressure. In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. I'm trying to reconcile a paradox regarding air pressure and temperature. It also explains why air gets. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT The Atmosphere in Motion PowerPoint Presentation ID1718980 Does Air Pressure Increase Or Decrease With Temperature If you had a way to increase pressure with no volume change, then yes, temperature would increase by the ideal gas law. If air pressure decreases, the temperature decreases. Ideal gas law (pv = nrt): For example, if air pressure increases, the temperature must increase. As the number of molecules increases, they exert more pressure on a surface, and the. Does Air Pressure Increase Or Decrease With Temperature.
From ar.inspiredpencil.com
Air Pressure Diagram For Kids Does Air Pressure Increase Or Decrease With Temperature In january, when air temperatures average 20.7 c (69f), the average pressure is 1019.1 mb. It also explains why air gets colder at higher. Density is the ratio of the mass to the. The number of air molecules above a surface determines air pressure. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0. Does Air Pressure Increase Or Decrease With Temperature.
From www.slideserve.com
PPT Air Pressure PowerPoint Presentation, free download ID5368729 Does Air Pressure Increase Or Decrease With Temperature Since air pressure is proportional to the density of the air, a decrease in density results in a decrease in air pressure. It also explains why air gets colder at higher. Density is the ratio of the mass to the. As the number of molecules increases, they exert more pressure on a surface, and the total. For example, if air. Does Air Pressure Increase Or Decrease With Temperature.
From www.dreamstime.com
Atmospheric Pressure Example with Lower and Higher Altitude Outline Does Air Pressure Increase Or Decrease With Temperature As the number of molecules increases, they exert more pressure on a surface, and the total. It also explains why air gets colder at higher. In the case of the ideal. In june, when the average air temperature is 27.6c (81.7f), the average air pressure is 1015.0 mb. The number of air molecules above a surface determines air pressure. If. Does Air Pressure Increase Or Decrease With Temperature.
From engineerexcel.com
Pressure Temperature Graphs Explained EngineerExcel Does Air Pressure Increase Or Decrease With Temperature It also explains why air gets colder at higher. The number of air molecules above a surface determines air pressure. I'm trying to reconcile a paradox regarding air pressure and temperature. Density is the ratio of the mass to the. As the number of molecules increases, they exert more pressure on a surface, and the total. In june, when the. Does Air Pressure Increase Or Decrease With Temperature.