List Of Off Patent Drugs . To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. Drugpatentwatch ® estimated upcoming generic entry dates. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with.
from www.pharmacompass.com
Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. Drugpatentwatch ® estimated upcoming generic entry dates. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or.
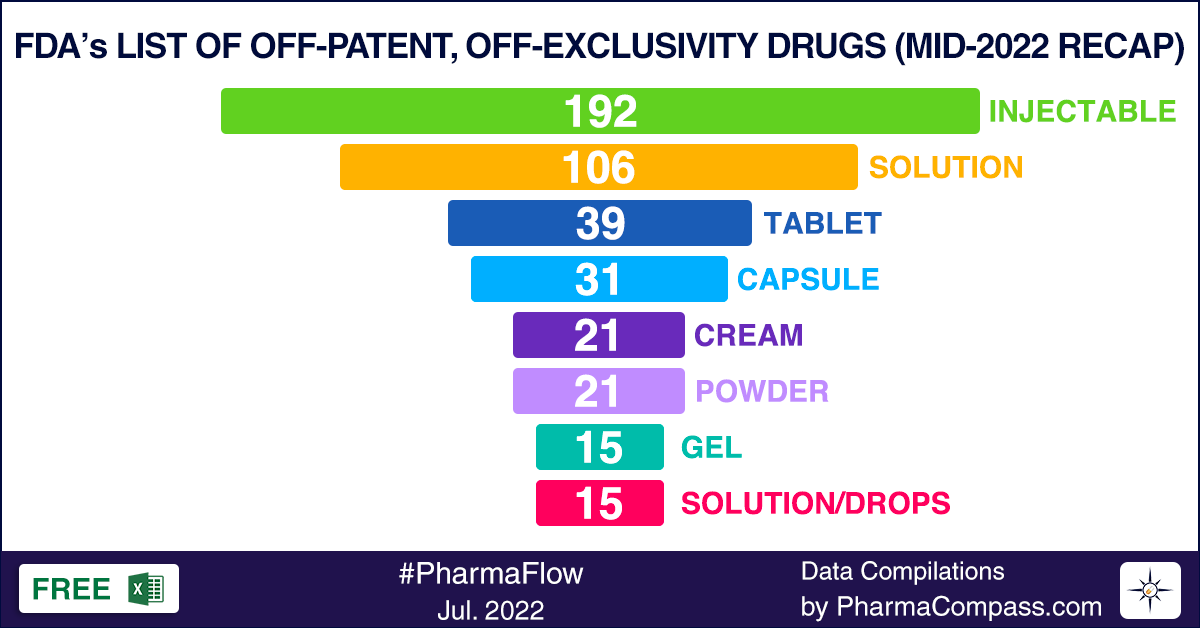
FDA’s list of offpatent, offexclusivity drugs sees steep rise in new
List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. Drugpatentwatch ® estimated upcoming generic entry dates. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with.
From www.studocu.com
CommonEmergencyDrugs COMMON EMERGENCY DRUGS DRUG NAME STOCK DOSE List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. Drugpatentwatch ® estimated upcoming generic entry dates. The fda maintains a. List Of Off Patent Drugs.
From www.researchgate.net
Top 200 prescribed drugs Download Table List Of Off Patent Drugs The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities.. List Of Off Patent Drugs.
From www.slideserve.com
PPT Division of Pediatric Drug Development PowerPoint Presentation List Of Off Patent Drugs The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Drugpatentwatch ® estimated upcoming generic entry dates. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. To improve transparency and encourage the development and submission of abbreviated. List Of Off Patent Drugs.
From www.researchgate.net
(PDF) A Review on the Drugs Getting OffPatent between the Years 2022 List Of Off Patent Drugs The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of. List Of Off Patent Drugs.
From gbu-taganskij.ru
Popular Nasal Decongestant Doesn't Actually Relieve, 57 OFF List Of Off Patent Drugs The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents. List Of Off Patent Drugs.
From www.moneycontrol.com
NLEM 2022 Govt releases new list of 384 essential medicines; 34 drugs List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated. List Of Off Patent Drugs.
From www.pharmacompass.com
FDA’s list of offpatent, offexclusivity drugs sees steep rise in new List Of Off Patent Drugs Drugpatentwatch ® estimated upcoming generic entry dates. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. The opoe list includes approved new drug applications (ndas) for drug. List Of Off Patent Drugs.
From www.semanticscholar.org
Table 1 from The Effects of Patent Expiration on Prescription Rates of List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. List of off‐patent, off‐exclusivity drugs. List Of Off Patent Drugs.
From www.koreabiomed.com
Offpatent drugs Godex, Atozet on a roll without generics List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Drugpatentwatch ® estimated upcoming generic entry dates. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for. List Of Off Patent Drugs.
From www.uspharmacist.com
New Generics in the Pipeline List Of Off Patent Drugs The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. Drugpatentwatch ® estimated upcoming generic entry dates. To improve transparency and encourage the development and submission of abbreviated. List Of Off Patent Drugs.
From www.pharmaexcipients.com
USFDA’s list of drugs that need generic alternatives Pharma Excipients List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Drugpatentwatch ® estimated upcoming generic entry dates. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for. List Of Off Patent Drugs.
From www.tandfonline.com
The impact of South Korea’s new drugpricing policy on market List Of Off Patent Drugs Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. Drugpatentwatch ® estimated upcoming generic entry dates. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs. List Of Off Patent Drugs.
From www.uspharmacist.com
Generic Drugs in the Pipeline 2017 Update List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and. List Of Off Patent Drugs.
From www.fiercepharma.com
Copycats wanted Pfizer, AZ and more under threat as China solicits List Of Off Patent Drugs List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage. List Of Off Patent Drugs.
From www.pharmacompass.com
USFDA's list of offpatent drugs that need generic alternatives Radio List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities.. List Of Off Patent Drugs.
From www.sfgate.com
How much for medicine? Depends on where you buy / Bay Area survey shows List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Drugpatentwatch ® estimated upcoming generic entry dates. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for. List Of Off Patent Drugs.
From www.pharmacompass.com
FDA’s list of offpatent drugs suggests higher approvals of first List Of Off Patent Drugs Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the. List Of Off Patent Drugs.
From www.nber.org
The Digest NBER List Of Off Patent Drugs The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Drugpatentwatch ® estimated upcoming generic entry dates. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list. List Of Off Patent Drugs.
From www.slideshare.net
Drug Patents Expiring in 2011 Sample by closer2patents List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. The opoe list includes approved new drug applications (ndas) for. List Of Off Patent Drugs.
From www.pfizer.com
Patents Let’s Protect What’s Next Pfizer List Of Off Patent Drugs Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new. List Of Off Patent Drugs.
From www.scribd.com
Ayurveda & Patent Drugs List PDF List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Explore the list maintained by fda of approved new drug. List Of Off Patent Drugs.
From www.slideshare.net
Drug+patent+expiration+table List Of Off Patent Drugs The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a list. List Of Off Patent Drugs.
From www.infogrades.com
Pharmaceutical Drug Patent Expiration List for 2017 [Infographic] List Of Off Patent Drugs Drugpatentwatch ® estimated upcoming generic entry dates. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for. List Of Off Patent Drugs.
From www.moneycontrol.com
NLEM 2022 Govt releases new list of 384 essential medicines; 34 drugs List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. Drugpatentwatch ® estimated upcoming generic entry dates. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. Explore the list. List Of Off Patent Drugs.
From mavink.com
Printable Top 200 Drug Classification List List Of Off Patent Drugs The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. List of off‐patent, off‐exclusivity. List Of Off Patent Drugs.
From www.researchgate.net
List of drugs classified in classes 1 and 3 of BCS. Download Table List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. Explore the list maintained. List Of Off Patent Drugs.
From www.pharmacompass.com
FDA's List of OffPatent, OffExclusivity Drugs with No Approved List Of Off Patent Drugs The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list maintained by fda of approved new drug. List Of Off Patent Drugs.
From www.lachmanconsultants.com
OffPatent Drugs with No Competition Lists Updated Lachman Consultants List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage. List Of Off Patent Drugs.
From www.researchgate.net
List of drugs patent under FDDS 24 Download Table List Of Off Patent Drugs List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. To improve transparency and encourage. List Of Off Patent Drugs.
From www.pharmacompass.com
Radio Data Compilation Market Intel PharmaFlow List Of Off Patent Drugs Drugpatentwatch ® estimated upcoming generic entry dates. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The fda maintains a. List Of Off Patent Drugs.
From old.sermitsiaq.ag
Printable Medication Cheat Sheet For Nurses List Of Off Patent Drugs Drugpatentwatch ® estimated upcoming generic entry dates. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for. List Of Off Patent Drugs.
From www.pharmacompass.com
Updated FDA List Of OffPatent, OffExclusivity Drugs Without An List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. To. List Of Off Patent Drugs.
From www.pharmacompass.com
FDA’s June 2023 list of offpatent, offexclusivity drugs sees steep List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Explore the list maintained by fda of approved new drug application (nda) drug products that are no longer protected by patents or exclusivities. The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or.. List Of Off Patent Drugs.
From info.dicksondata.com
[Infographic] Major Drugs Going OffPatent in 2018 List Of Off Patent Drugs The opoe list includes approved new drug applications (ndas) for drug products that are not protected by patents or. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. To improve transparency and. List Of Off Patent Drugs.
From blog.ipleaders.in
Pharmaceutical drugs and patent protection with reference to COVID19 List Of Off Patent Drugs To improve transparency and encourage the development and submission of abbreviated new drug applications (andas) for drugs with. Drugpatentwatch ® estimated upcoming generic entry dates. List of off‐patent, off‐exclusivity drugs without an approved generic to improve transparency and. The fda maintains a list of approved new drug application (nda) drug products that are no longer protected by. To improve transparency. List Of Off Patent Drugs.