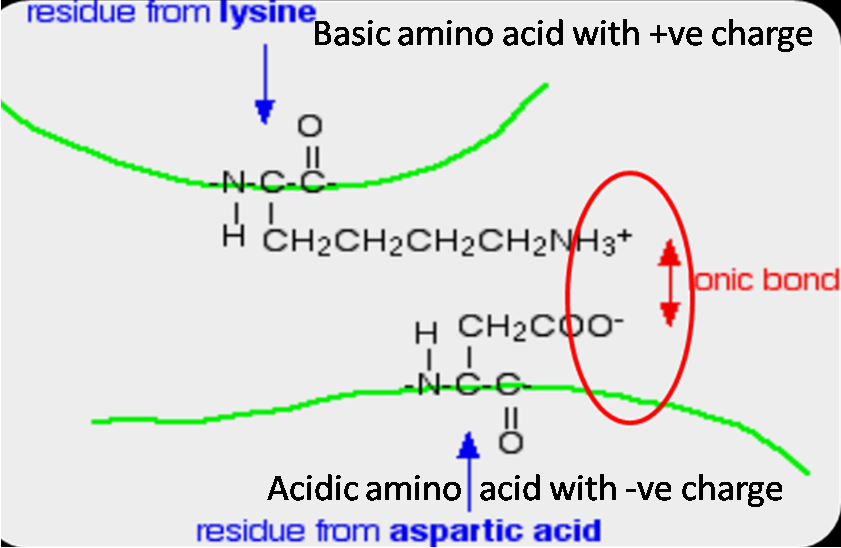
Amino Acids Can Participate In Hydrogen Bonding . Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. The following 3d jmol image allows you to highlight the carbonyl. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Acidic side chains, such as.
from www.biologyexams4u.com
The following 3d jmol image allows you to highlight the carbonyl. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Acidic side chains, such as.
What are the 6 Major Chemical Bonds or Interactions In Proteins?
Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Acidic side chains, such as. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. The following 3d jmol image allows you to highlight the carbonyl.
From www.numerade.com
SOLVED Using the two amino acids below Draw in the appropriate Amino Acids Can Participate In Hydrogen Bonding Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Acidic side chains, such as. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom. Amino Acids Can Participate In Hydrogen Bonding.
From saylordotorg.github.io
Proteins Amino Acids Can Participate In Hydrogen Bonding The following 3d jmol image allows you to highlight the carbonyl. Acidic side chains, such as. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. The hydrogen is covalently attached to one of. Amino Acids Can Participate In Hydrogen Bonding.
From quizlet.com
Which of the following pairs of amino acids can form hydroge Quizlet Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Hydrogen bonding forms between. Amino Acids Can Participate In Hydrogen Bonding.
From philschatz.com
Amines and Amides · Chemistry Amino Acids Can Participate In Hydrogen Bonding In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Polar amino acids have. Amino Acids Can Participate In Hydrogen Bonding.
From slideplayer.com
AMINO ACIDS. ppt download Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Acidic side chains, such as. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other. Amino Acids Can Participate In Hydrogen Bonding.
From www.shutterstock.com
Transient Hydrogen Bonds Protein Formation Amino Stock Vector (Royalty Amino Acids Can Participate In Hydrogen Bonding Acidic side chains, such as. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to. Amino Acids Can Participate In Hydrogen Bonding.
From www.shutterstock.com
Hydrogen Bond Chemistry Lesson Infographic Hydrogen Stock Vector Amino Acids Can Participate In Hydrogen Bonding Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. The following 3d jmol image. Amino Acids Can Participate In Hydrogen Bonding.
From www.numerade.com
Which of the following pairs of amino acids can form hydrogen bonds Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The following 3d jmol image allows you to highlight the carbonyl. Acidic side chains, such as. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Hydrogen bonding forms between a. Amino Acids Can Participate In Hydrogen Bonding.
From socratic.org
What type of interaction is depicted by the dashed line? Socratic Amino Acids Can Participate In Hydrogen Bonding Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Acidic side chains, such as. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical. Amino Acids Can Participate In Hydrogen Bonding.
From www.researchgate.net
Diagram showing how (a) unmodified amino acids can form β sheet Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. The following 3d jmol image. Amino Acids Can Participate In Hydrogen Bonding.
From www.biologyexams4u.com
What are the 6 Major Chemical Bonds or Interactions In Proteins? Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. The following 3d jmol image allows you to highlight the carbonyl. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Hydrogen bonding forms between a highly electronegative. Amino Acids Can Participate In Hydrogen Bonding.
From www.youtube.com
Identifying Hydrogen Bond Donors & Acceptors YouTube Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Acidic. Amino Acids Can Participate In Hydrogen Bonding.
From vigyansarovar.com
Chemical Classification of Amino Acids Vigyan Sarovar Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Hydrogen bonding forms between. Amino Acids Can Participate In Hydrogen Bonding.
From www.slideserve.com
PPT Introduction to Amino Acids of Medical Importance PowerPoint Amino Acids Can Participate In Hydrogen Bonding Acidic side chains, such as. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. The following 3d jmol image allows you to highlight the carbonyl. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Hydrogen bonding forms between a highly electronegative oxygen. Amino Acids Can Participate In Hydrogen Bonding.
From www.weareeaton.com
NonPolar Amino Acids The Building Blocks Of Proteins We Are Eaton Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. The following 3d jmol image allows you to highlight the carbonyl. Acidic side chains, such as. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen. Amino Acids Can Participate In Hydrogen Bonding.
From www.science-revision.co.uk
Hydrogen bonding Amino Acids Can Participate In Hydrogen Bonding In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Acidic side chains, such as. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom. Amino Acids Can Participate In Hydrogen Bonding.
From courses.lumenlearning.com
Amino Acids Structure Nutrition Amino Acids Can Participate In Hydrogen Bonding Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Acidic side chains, such as. The following 3d jmol image allows you to highlight the carbonyl. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen. Amino Acids Can Participate In Hydrogen Bonding.
From www.chegg.com
Solved Which of the following amino acids is MOST likely to Amino Acids Can Participate In Hydrogen Bonding The following 3d jmol image allows you to highlight the carbonyl. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar. Amino Acids Can Participate In Hydrogen Bonding.
From www.cryst.bbk.ac.uk
Hydrogen Bonds Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Acidic. Amino Acids Can Participate In Hydrogen Bonding.
From stock.adobe.com
Vetor de hydrogen bond. chemistry lesson. Infographic. hydrogen bonds Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Acidic side chains, such as. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the. Amino Acids Can Participate In Hydrogen Bonding.
From www.numerade.com
SOLVED Using the two amino acids below Draw in the appropriate Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. The following 3d jmol image allows you to highlight the carbonyl. Polar amino acids have side chains that contain electronegative atoms like oxygen or. Amino Acids Can Participate In Hydrogen Bonding.
From quizlet.com
Which of the following pairs of amino acids can form hydroge Quizlet Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Polar amino acids have side chains that contain. Amino Acids Can Participate In Hydrogen Bonding.
From donate-faqs.com
Which Amino Acids Can Donate A Hydrogen Bond Amino Acids Can Participate In Hydrogen Bonding Acidic side chains, such as. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Amino acids are distinguished by their side chains, which can be categorized as acidic or. Amino Acids Can Participate In Hydrogen Bonding.
From donate-faqs.com
Which Amino Acids Can Donate A Hydrogen Bond Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. The following 3d jmol image allows you to highlight the carbonyl. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Polar amino acids have side chains that contain electronegative. Amino Acids Can Participate In Hydrogen Bonding.
From philschatz.com
Organic Compounds Essential to Human Functioning · Anatomy and Physiology Amino Acids Can Participate In Hydrogen Bonding The following 3d jmol image allows you to highlight the carbonyl. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Amino acids are distinguished by their side chains, which can be categorized as. Amino Acids Can Participate In Hydrogen Bonding.
From www.animalia-life.club
Hydrogen Bonding In Dnapairs Amino Acids Can Participate In Hydrogen Bonding Acidic side chains, such as. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to. Amino Acids Can Participate In Hydrogen Bonding.
From www.gkseries.com
All of the below mentioned amino acids can participate in hydrogen Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Acidic side chains, such as. The following 3d jmol image allows you to highlight the carbonyl. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. In proteins, the hydrogen bonding interactions of the backbone. Amino Acids Can Participate In Hydrogen Bonding.
From www.biologyexams4u.com
What are the 6 Major Chemical Bonds or Interactions In Proteins? Amino Acids Can Participate In Hydrogen Bonding The following 3d jmol image allows you to highlight the carbonyl. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino. Amino Acids Can Participate In Hydrogen Bonding.
From www.chegg.com
Solved Which of the following amino acids whose side chain Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Amino. Amino Acids Can Participate In Hydrogen Bonding.
From book.bionumbers.org
» What is the energy of a hydrogen bond? Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. Acidic side chains, such as. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. The hydrogen. Amino Acids Can Participate In Hydrogen Bonding.
From microbiologynotes.org
Amino acids physical, chemical properties and peptide bond Amino Acids Can Participate In Hydrogen Bonding The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. Acidic side. Amino Acids Can Participate In Hydrogen Bonding.
From www.semanticscholar.org
Figure 3 from Recognition of nucleic acid bases and basepairs by Amino Acids Can Participate In Hydrogen Bonding Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The following 3d jmol image allows you to highlight the carbonyl. Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to. Amino Acids Can Participate In Hydrogen Bonding.
From www.chegg.com
Solved The amino acid shown below can participate in Amino Acids Can Participate In Hydrogen Bonding In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The following 3d jmol. Amino Acids Can Participate In Hydrogen Bonding.
From donate-faqs.com
Which Amino Acids Can Donate A Hydrogen Bond Amino Acids Can Participate In Hydrogen Bonding Hydrogen bonding forms between a highly electronegative oxygen atom or a nitrogen atom and a hydrogen atom attached to another oxygen atom or a nitrogen atom, such as those found in polar amino acid side chains. In proteins, the hydrogen bonding interactions of the backbone amide groups is critical to defining the structure. The following 3d jmol image allows you. Amino Acids Can Participate In Hydrogen Bonding.
From www.biologyexams4u.com
What are the 6 Major Chemical Bonds or Interactions In Proteins? Amino Acids Can Participate In Hydrogen Bonding Polar amino acids have side chains that contain electronegative atoms like oxygen or nitrogen, leading to uneven electron distribution. Amino acids are distinguished by their side chains, which can be categorized as acidic or basic. The hydrogen is covalently attached to one of the atoms (called the hydrogen bond donor) and interacts with the other (the hydrogen bond. In proteins,. Amino Acids Can Participate In Hydrogen Bonding.