A Calorimeter Of Heat Capacity 100 J/K . It can analyze the heat exchange between up to 3 objects. Explain the technique of calorimetry. `100g` of water at `40^(@)c` of. the calorimetry calculator can help you solve complex calorimetry problems. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. Calculate and interpret heat and related properties using typical calorimetry data. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.
from www.tec-science.com
Explain the technique of calorimetry. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. `100g` of water at `40^(@)c` of. It can analyze the heat exchange between up to 3 objects. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calculate and interpret heat and related properties using typical calorimetry data. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry.
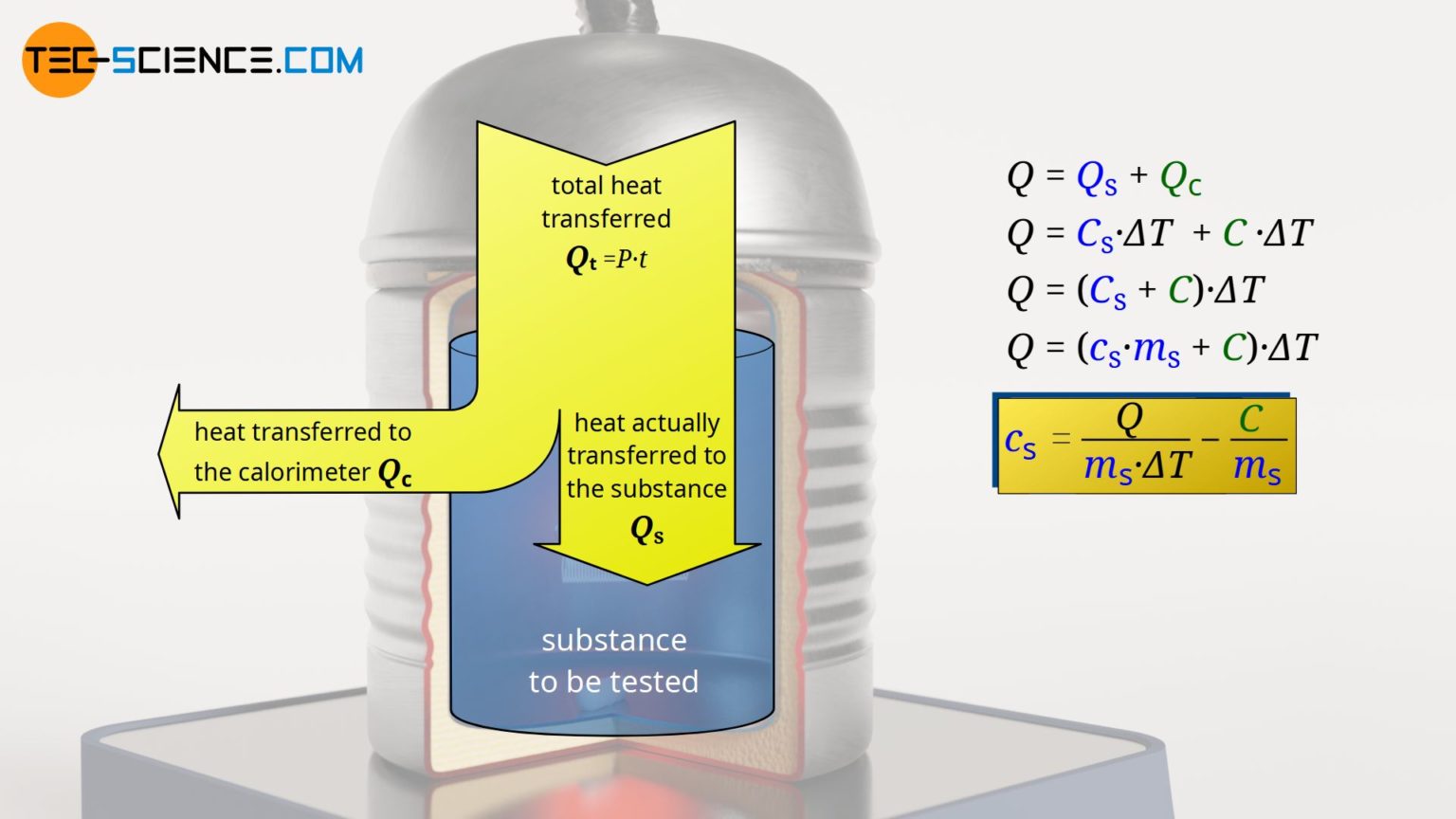
Calorimeter to determine the specific heat capacities of liquids tec
A Calorimeter Of Heat Capacity 100 J/K the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. Calculate and interpret heat and related properties using typical calorimetry data. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. `100g` of water at `40^(@)c` of. Explain the technique of calorimetry. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. It can analyze the heat exchange between up to 3 objects. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. the calorimetry calculator can help you solve complex calorimetry problems. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry.
From exodnulby.blob.core.windows.net
Lab Calorimetry And Specific Heat Summary at Barbara Bailey blog A Calorimeter Of Heat Capacity 100 J/K It can analyze the heat exchange between up to 3 objects. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. the calorimetry calculator can help you solve complex calorimetry problems. compare heat flow from hot to cold objects. A Calorimeter Of Heat Capacity 100 J/K.
From www.youtube.com
2.2A Calorimetry, Heat Capacity & Specific Heat Capacity YouTube A Calorimeter Of Heat Capacity 100 J/K Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the calorimetry calculator can help you solve complex calorimetry problems. `100g` of water at `40^(@)c` of. Explain the technique of calorimetry. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. a calorimeter of heat capacity `100 j//k` is at. A Calorimeter Of Heat Capacity 100 J/K.
From studyadvertiser.z21.web.core.windows.net
How To Use A Calorimeter Stepbystep A Calorimeter Of Heat Capacity 100 J/K specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Explain the technique of calorimetry. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the calorimetry calculator can help you solve complex calorimetry problems. compare heat flow from. A Calorimeter Of Heat Capacity 100 J/K.
From www.chegg.com
Solved HEAT CAPACITY OF THE CALORIMETER Now, calculate the A Calorimeter Of Heat Capacity 100 J/K compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. a calorimeter is a device used to measure the amount of heat involved in a. A Calorimeter Of Heat Capacity 100 J/K.
From dxowceosf.blob.core.windows.net
Calorimetry All Formulas at Spencer McSwain blog A Calorimeter Of Heat Capacity 100 J/K Calculate and interpret heat and related properties using typical calorimetry data. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. It can analyze the heat exchange between. A Calorimeter Of Heat Capacity 100 J/K.
From www.studypool.com
SOLUTION Calorimetry Enthalpy of Reaction and Heat Capacity of a A Calorimeter Of Heat Capacity 100 J/K Explain the technique of calorimetry. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. the calorimetry calculator can help you solve complex calorimetry problems. `100g` of water at `40^(@)c` of. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by. A Calorimeter Of Heat Capacity 100 J/K.
From revisionug.com
What is Calorimetry? A Calorimeter Of Heat Capacity 100 J/K specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Calculate and interpret heat and related properties using typical calorimetry data. `100g` of water at `40^(@)c` of. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. Calculate the. A Calorimeter Of Heat Capacity 100 J/K.
From www.youtube.com
CH5 Q5 Calculating the Heat Capacity of a Calorimeter YouTube A Calorimeter Of Heat Capacity 100 J/K the calorimetry calculator can help you solve complex calorimetry problems. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. Explain the technique of calorimetry. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. It can analyze the heat exchange between up to 3 objects. . A Calorimeter Of Heat Capacity 100 J/K.
From www.chegg.com
Solved CHEMISTRY DETERMINING HEAT CAPACITY OF A CALORIMETER A Calorimeter Of Heat Capacity 100 J/K specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. It can analyze. A Calorimeter Of Heat Capacity 100 J/K.
From deon-has-edwards.blogspot.com
Heat Capacity of Calorimeter DeonhasEdwards A Calorimeter Of Heat Capacity 100 J/K Explain the technique of calorimetry. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. a calorimeter of heat. A Calorimeter Of Heat Capacity 100 J/K.
From www.toppr.com
A calorimeter of heat capacity 100 J/K is room temperature of 30°C. 100 A Calorimeter Of Heat Capacity 100 J/K Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. It can analyze the heat exchange between up to 3 objects. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. a calorimeter of heat capacity 100 j/k. A Calorimeter Of Heat Capacity 100 J/K.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec A Calorimeter Of Heat Capacity 100 J/K It can analyze the heat exchange between up to 3 objects. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. the calorimetry calculator can help you solve complex calorimetry problems. Explain the technique of calorimetry. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical. A Calorimeter Of Heat Capacity 100 J/K.
From www.toppr.com
partly Hence temperature of mixture will remain 100°C. stration 14 A Calorimeter Of Heat Capacity 100 J/K a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. Calculate and interpret heat and related properties using typical calorimetry data. `100g` of water at `40^(@)c` of. compare heat flow from. A Calorimeter Of Heat Capacity 100 J/K.
From askfilo.com
In a constant pressure calorimeter with heat capacity of 453 Jk−1,200 mL A Calorimeter Of Heat Capacity 100 J/K `100g` of water at `40^(@)c` of. Calculate and interpret heat and related properties using typical calorimetry data. the calorimetry calculator can help you solve complex calorimetry problems. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. compare heat flow from hot. A Calorimeter Of Heat Capacity 100 J/K.
From exodnulby.blob.core.windows.net
Lab Calorimetry And Specific Heat Summary at Barbara Bailey blog A Calorimeter Of Heat Capacity 100 J/K a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. Calculate the molar heat of enthalpy for a reactions using coffee cup. A Calorimeter Of Heat Capacity 100 J/K.
From haipernews.com
How To Calculate Heat Capacity From Calorimeter Haiper A Calorimeter Of Heat Capacity 100 J/K a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. It can. A Calorimeter Of Heat Capacity 100 J/K.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec A Calorimeter Of Heat Capacity 100 J/K a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. `100g` of water at `40^(@)c` of. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. specific heat. A Calorimeter Of Heat Capacity 100 J/K.
From studylib.net
Electrical Determination of the Heat Capacity of a Calorimeter in A Calorimeter Of Heat Capacity 100 J/K a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. `100g` of water at `40^(@)c` of. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Calculate heat, temperature change, and specific heat after. A Calorimeter Of Heat Capacity 100 J/K.
From courses.lumenlearning.com
Calorimetry Chemistry for Majors A Calorimeter Of Heat Capacity 100 J/K compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. `100g` of water at `40^(@)c` of. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the combustion of 1 mole of glucose $\ce{c6h12o6}$. A Calorimeter Of Heat Capacity 100 J/K.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec A Calorimeter Of Heat Capacity 100 J/K specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. Explain the technique of calorimetry. Calculate. A Calorimeter Of Heat Capacity 100 J/K.
From www.toppr.com
A calorimeter of heat capacity 100 J/K is room temperature of 30°C. 100 A Calorimeter Of Heat Capacity 100 J/K the calorimetry calculator can help you solve complex calorimetry problems. specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. Calculate and interpret heat and related properties using typical calorimetry data. It can analyze the heat exchange between up to 3 objects. Calculate. A Calorimeter Of Heat Capacity 100 J/K.
From louis.pressbooks.pub
Calorimetry (9.2) General Chemistry A Calorimeter Of Heat Capacity 100 J/K compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. specific heat is the amount of thermal energy you need to supply to a sample. A Calorimeter Of Heat Capacity 100 J/K.
From exonegtdm.blob.core.windows.net
A Level Chemistry Calorimetry Practical Aqa at Richelle Doty blog A Calorimeter Of Heat Capacity 100 J/K a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. `100g` of water at `40^(@)c` of. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. the calorimetry calculator can help you solve complex calorimetry problems. specific heat is the amount of thermal. A Calorimeter Of Heat Capacity 100 J/K.
From www.animalia-life.club
Calorimeter Diagram A Calorimeter Of Heat Capacity 100 J/K specific heat is the amount of thermal energy you need to supply to a sample weighing 1 kg to increase its temperature by 1 k. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. a. A Calorimeter Of Heat Capacity 100 J/K.
From www.studypool.com
SOLUTION Bs physics practical heat capacity of a calorimeter Studypool A Calorimeter Of Heat Capacity 100 J/K Calculate and interpret heat and related properties using typical calorimetry data. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. the calorimetry calculator can help you solve complex calorimetry problems. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3. A Calorimeter Of Heat Capacity 100 J/K.
From www.scienceabc.com
Molar Heat Capacity Definition, Formula, Equation, Calculation A Calorimeter Of Heat Capacity 100 J/K the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calculate and interpret heat and related properties using typical calorimetry data. Explain the technique of calorimetry. `100g` of water at `40^(@)c` of. specific heat is. A Calorimeter Of Heat Capacity 100 J/K.
From haipernews.com
How To Calculate Heat Capacity Of A Calorimeter Haiper A Calorimeter Of Heat Capacity 100 J/K a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. `100g` of water at `40^(@)c` of. Calculate and interpret heat and related properties using typical calorimetry data. the calorimetry calculator can help you solve complex calorimetry problems. Explain the technique. A Calorimeter Of Heat Capacity 100 J/K.
From www.doubtnut.com
A calorimeter of heat capacity 100 J//K is at room temperature of 30^( A Calorimeter Of Heat Capacity 100 J/K a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. `100g` of water at `40^(@)c` of. Calculate and interpret heat and related properties using typical calorimetry data. the calorimetry calculator can help you solve complex calorimetry problems. specific heat is the amount of thermal energy you need to supply to a sample weighing. A Calorimeter Of Heat Capacity 100 J/K.
From www.slideserve.com
PPT Unit 13 Thermochemistry PowerPoint Presentation, free download A Calorimeter Of Heat Capacity 100 J/K It can analyze the heat exchange between up to 3 objects. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. Calculate heat, temperature change, and. A Calorimeter Of Heat Capacity 100 J/K.
From www.slideserve.com
PPT CHEM 1011 PowerPoint Presentation, free download ID317228 A Calorimeter Of Heat Capacity 100 J/K Calculate heat, temperature change, and specific heat after thermal equilibrium is reached between two substances in a calorimeter. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. Explain the technique of calorimetry. specific heat is the amount. A Calorimeter Of Heat Capacity 100 J/K.
From users.highland.edu
Calorimetry A Calorimeter Of Heat Capacity 100 J/K Calculate and interpret heat and related properties using typical calorimetry data. Explain the technique of calorimetry. It can analyze the heat exchange between up to 3 objects. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. the calorimetry calculator can help you solve complex calorimetry problems. the combustion of 1. A Calorimeter Of Heat Capacity 100 J/K.
From www.slideserve.com
PPT Specific Heat Capacity PowerPoint Presentation, free download A Calorimeter Of Heat Capacity 100 J/K the calorimetry calculator can help you solve complex calorimetry problems. a calorimeter of heat capacity `100 j//k` is at room temperature of `30^(@)c`. the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. Calculate and interpret heat and related properties using typical calorimetry data. Calculate the molar heat of enthalpy for a reactions using. A Calorimeter Of Heat Capacity 100 J/K.
From bceweb.org
Heat Capacity Chart A Visual Reference of Charts Chart Master A Calorimeter Of Heat Capacity 100 J/K the calorimetry calculator can help you solve complex calorimetry problems. Calculate the molar heat of enthalpy for a reactions using coffee cup calorimetry. It can analyze the heat exchange between up to 3 objects. a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. a calorimeter of heat capacity `100 j//k` is at. A Calorimeter Of Heat Capacity 100 J/K.
From www.youtube.com
CHEMISTRY 101 Calculating Heat Capacity of a Bomb Calorimeter YouTube A Calorimeter Of Heat Capacity 100 J/K the combustion of 1 mole of glucose $\ce{c6h12o6}$ releases $\pu{2.82\times10^3 kj}$ of heat. the calorimetry calculator can help you solve complex calorimetry problems. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. specific heat is the amount of thermal energy you need to supply to a sample weighing 1. A Calorimeter Of Heat Capacity 100 J/K.
From dxowceosf.blob.core.windows.net
Calorimetry All Formulas at Spencer McSwain blog A Calorimeter Of Heat Capacity 100 J/K a calorimeter of heat capacity 100 j/k is at room temperature of 30 c. a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. compare heat flow from hot to cold objects in an ideal calorimeter versus a real calorimeter. Calculate heat, temperature change, and specific heat after. A Calorimeter Of Heat Capacity 100 J/K.