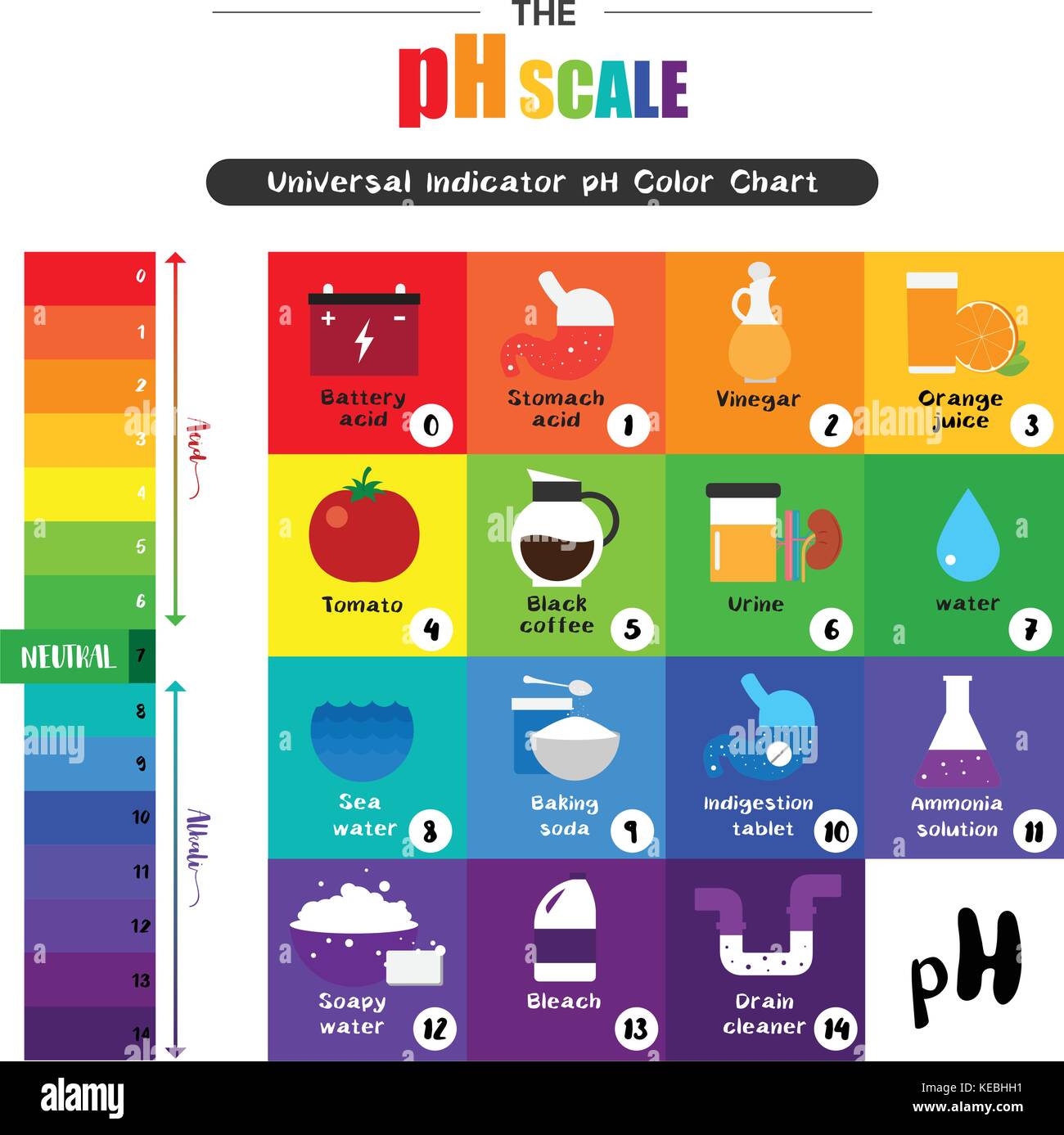
What S The Ph Of Salt Water . Salts derived from a weak base and a weak acid may be acidic or basic. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. A ph of 7 (pure water) is neutral; Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. However, excess carbon dioxide in the water can affect this delicate balance. A ph higher than 7 is basic. Ph is measured on a 14 point scale. Salts derived from a strong acid and a strong base give neutral solutions. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. When pure water is dropped into a solution of universal indicator, the.
from www.alamy.com
In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. When pure water is dropped into a solution of universal indicator, the. A ph higher than 7 is basic. A ph of 7 (pure water) is neutral; Ph is measured on a 14 point scale. Salts derived from a weak base and a weak acid may be acidic or basic. Salts derived from a strong acid and a strong base give neutral solutions. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. However, excess carbon dioxide in the water can affect this delicate balance.
The pH scale Universal Indicator pH Color Chart diagram acidic alkaline
What S The Ph Of Salt Water The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. A ph higher than 7 is basic. Salts derived from a strong acid and a strong base give neutral solutions. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. However, excess carbon dioxide in the water can affect this delicate balance. When pure water is dropped into a solution of universal indicator, the. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Ph is measured on a 14 point scale. Salts derived from a weak base and a weak acid may be acidic or basic. A ph of 7 (pure water) is neutral; The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14.
From www.alevelh2chemistry.com
AcidBase Equilibrium How to calculate pH of a Salt? ALevel H2 What S The Ph Of Salt Water The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. Salts derived from a weak base and a weak acid may be acidic or basic. Salts derived from a strong acid and a strong base give neutral solutions. The average ph of the salt water in the. What S The Ph Of Salt Water.
From treatmywater.com
pH What You need to Know Treat My Water What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. Salts derived from a strong acid and a strong base give neutral solutions. Ph is measured on a 14 point scale. Salts derived from a weak base and a weak acid may be acidic or basic. Ph, which stands for “power of hydrogen”, is a measure of how. What S The Ph Of Salt Water.
From www.bbc.co.uk
What is the pH scale and what does it measure? BBC Bitesize What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. A ph higher than 7 is basic. Ph is measured on a 14 point scale. When pure water is dropped into a solution of universal indicator, the. Salts derived from a strong acid and a strong base give neutral solutions. Salts derived. What S The Ph Of Salt Water.
From sciencenotes.org
The pH Scale of Common Chemicals What S The Ph Of Salt Water Salts derived from a weak base and a weak acid may be acidic or basic. When pure water is dropped into a solution of universal indicator, the. Ph is measured on a 14 point scale. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than. What S The Ph Of Salt Water.
From www.slideserve.com
PPT pH of Salts PowerPoint Presentation, free download ID1994692 What S The Ph Of Salt Water The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. Salts derived from a strong acid and a strong base give neutral solutions. A ph of 7 (pure water) is neutral; A ph higher than 7 is basic. However, excess carbon dioxide in. What S The Ph Of Salt Water.
From blog.havells.com
Right pH Level in Drinking Water How Essential? Havells India Blog What S The Ph Of Salt Water In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. A ph of 7 (pure water) is. What S The Ph Of Salt Water.
From www.alamy.com
The pH scale Universal Indicator pH Color Chart diagram acidic alkaline What S The Ph Of Salt Water When pure water is dropped into a solution of universal indicator, the. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Salts derived from a strong acid and a strong base give neutral solutions. Salts derived from a weak base and a weak acid may be acidic or basic. The average. What S The Ph Of Salt Water.
From www.meritnation.com
What is the pH range of the salts obtained from rocks found in the sea What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Salts derived from a weak base and a weak acid may be acidic or basic. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic.. What S The Ph Of Salt Water.
From www.waterev.com
What is the PH of Distilled Water What is the PH value of water and What S The Ph Of Salt Water Salts derived from a strong acid and a strong base give neutral solutions. A ph of 7 (pure water) is neutral; In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. Salts derived from a weak base and a weak. What S The Ph Of Salt Water.
From aqualife.ca
Discover the power of pH on your health Aqualife What S The Ph Of Salt Water Salts derived from a strong acid and a strong base give neutral solutions. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. However, excess carbon dioxide in the water can affect this delicate balance. Ph is measured on a 14 point scale.. What S The Ph Of Salt Water.
From exofbishk.blob.core.windows.net
What Is The Ph Of Salt Water Solution at Travis Farley blog What S The Ph Of Salt Water Salts derived from a strong acid and a strong base give neutral solutions. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. Salts derived from a weak base and a weak acid may be acidic or basic. Ph, which. What S The Ph Of Salt Water.
From www.mometrix.com
pH Overview (Chemistry Review Video) What S The Ph Of Salt Water When pure water is dropped into a solution of universal indicator, the. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Salts derived from a strong acid and a strong base give neutral solutions. The average ph of the salt water in the oceans is about 8.1 close to the surface,. What S The Ph Of Salt Water.
From materialzonekonner88.z21.web.core.windows.net
Ph Of Salt Solutions Worksheet What S The Ph Of Salt Water In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. A ph of 7 (pure water) is. What S The Ph Of Salt Water.
From ecampusontario.pressbooks.pub
Water Hardness and pH Understanding Ingredients for the Canadian Baker What S The Ph Of Salt Water Salts derived from a strong acid and a strong base give neutral solutions. A ph of 7 (pure water) is neutral; Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to. What S The Ph Of Salt Water.
From www.youtube.com
How to Calculate the pH of a Salt Solution Practice Problems, Shortcut What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. Salts derived from a strong acid and a strong base give neutral solutions. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. In order for any type of salt to affect the ph (potential of hydrogen), it has to. What S The Ph Of Salt Water.
From hubpages.com
What is Universal Indicator and How To Use it What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. A ph of 7 (pure water) is neutral; A ph higher than 7 is basic. When pure water is dropped into a solution of universal indicator, the. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to. What S The Ph Of Salt Water.
From geo.libretexts.org
7.1 Properties of Seawater Geosciences LibreTexts What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. When pure water is dropped into a solution of universal indicator, the. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. However, excess carbon dioxide in the water. What S The Ph Of Salt Water.
From chem.libretexts.org
5.6 The pH Scale Chemistry LibreTexts What S The Ph Of Salt Water Salts derived from a weak base and a weak acid may be acidic or basic. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. A ph of 7 (pure water) is neutral; A ph higher than 7 is basic. In order for any type of salt. What S The Ph Of Salt Water.
From www.youtube.com
pH of Salts (लवणों का pH) pHofsalts salts YouTube What S The Ph Of Salt Water Salts derived from a strong acid and a strong base give neutral solutions. A ph of 7 (pure water) is neutral; When pure water is dropped into a solution of universal indicator, the. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms. What S The Ph Of Salt Water.
From mywaterearth.com
Safe PH Level For Drinking Water MyWaterEarth&Sky What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. When pure water is dropped into a solution of universal indicator, the. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. Ph is measured. What S The Ph Of Salt Water.
From zanyplantworld.com
PH Water Levels For The Plants Definition, Benefits, And Effects What S The Ph Of Salt Water The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. A ph of 7 (pure water) is neutral; Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. The average ph of the salt water in the oceans is. What S The Ph Of Salt Water.
From skfelixer.com
The pH Value Of Purified Water — All You Need To Know SKF Elixer What S The Ph Of Salt Water The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Salts derived from a strong acid and a strong base give neutral solutions. A ph. What S The Ph Of Salt Water.
From watertechadvice.com
pH of Water Everything You Need To Know What S The Ph Of Salt Water When pure water is dropped into a solution of universal indicator, the. Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Salts derived from a strong acid and a strong base give neutral solutions. However, excess carbon dioxide in the water can affect this delicate balance. In order for any type. What S The Ph Of Salt Water.
From www.intec-america.com
Things You Must Know About pH Control and Drinking Water Treatment What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. Ph is measured on a 14 point scale. A ph higher than 7 is basic. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. Salts derived from a strong acid. What S The Ph Of Salt Water.
From study.com
The AcidBase Properties of Water Video & Lesson Transcript What S The Ph Of Salt Water In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to release or bind the hydrogen atoms from the water. Salts derived from a strong acid and a strong base give neutral solutions. The ph scale measures how strongly acidic or alkaline a solution is using a set of values. What S The Ph Of Salt Water.
From inspectapedia.com
WHO pH for drinking water, too high pH or too low pH acidic What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. When pure water is dropped into a solution of universal indicator, the. A ph of 7 (pure water) is neutral; A ph higher than 7 is basic. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with water to. What S The Ph Of Salt Water.
From www.youtube.com
CHEMISTRY 201 Calculating the pH of a salt solution YouTube What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. A ph of 7 (pure water) is neutral; Ph is measured on a 14 point scale. A ph higher than 7 is basic. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph. What S The Ph Of Salt Water.
From www.premierpond.com
Guide to Pond pH Levels Premier Ponds What S The Ph Of Salt Water However, excess carbon dioxide in the water can affect this delicate balance. A ph of 7 (pure water) is neutral; Ph is measured on a 14 point scale. When pure water is dropped into a solution of universal indicator, the. The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0. What S The Ph Of Salt Water.
From nittygrittyscience.com
Section 2 Acids, Bases, and Salts Nitty Gritty Science What S The Ph Of Salt Water The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. However, excess carbon dioxide in the water can affect this delicate balance. A ph higher than 7 is basic. A ph of 7 (pure water) is neutral; The average ph of the salt water in the oceans. What S The Ph Of Salt Water.
From www.pmel.noaa.gov
The pH scale with some common examples What S The Ph Of Salt Water Salts derived from a weak base and a weak acid may be acidic or basic. Salts derived from a strong acid and a strong base give neutral solutions. Ph is measured on a 14 point scale. When pure water is dropped into a solution of universal indicator, the. Ph, which stands for “power of hydrogen”, is a measure of how. What S The Ph Of Salt Water.
From general.chemistrysteps.com
Acidity of a Salt Solution Chemistry Steps What S The Ph Of Salt Water The ph scale measures how strongly acidic or alkaline a solution is using a set of values from ph 0 to ph 14. Ph is measured on a 14 point scale. A ph of 7 (pure water) is neutral; The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is. What S The Ph Of Salt Water.
From www.teachoo.com
Salts and it's Properties (with Examples) Acids, Bases and Salt What S The Ph Of Salt Water A ph of 7 (pure water) is neutral; Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. In order for any type of salt. What S The Ph Of Salt Water.
From netsolwater.com
What are the pH and TDS in the water? How can it be controlled? Netsol What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being acidic. Salts derived from a strong acid and a strong base give neutral solutions. When pure. What S The Ph Of Salt Water.
From mungfali.com
Water PH Test Chart What S The Ph Of Salt Water A ph of 7 (pure water) is neutral; Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Ph is measured on a 14 point scale. The average ph of the salt water in the oceans is about 8.1 close to the surface, which means it is generally alkaline rather than being. What S The Ph Of Salt Water.
From www.medicalnewstoday.com
What to know about the pH of water What S The Ph Of Salt Water Ph, which stands for “power of hydrogen”, is a measure of how acidic or alkaline a solution is. Ph is measured on a 14 point scale. Salts derived from a weak base and a weak acid may be acidic or basic. In order for any type of salt to affect the ph (potential of hydrogen), it has to react with. What S The Ph Of Salt Water.