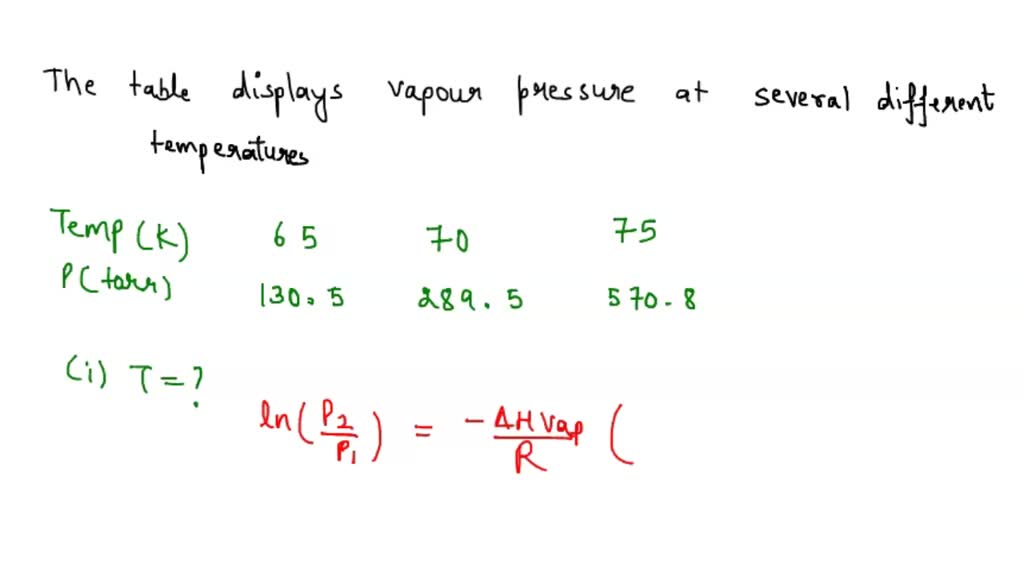Heat Of Vaporisation Nitrogen . (1) where q is the heat supplied during phase transition and m. Learn how nitrogen changes phase with temperature and pressure,. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). In case of liquid to gas phase change, this amount of energy is known as the. Latent heat, l is defined to be: Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. = (1) where q is. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. The latent heat of vaporization can be mathematically expressed as, lv= q m ;
from www.numerade.com
Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. = (1) where q is. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. (1) where q is the heat supplied during phase transition and m. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Latent heat, l is defined to be: Learn how nitrogen changes phase with temperature and pressure,. The latent heat of vaporization can be mathematically expressed as, lv= q m ;
SOLVED The table displays the vapor pressure of nitrogen at several
Heat Of Vaporisation Nitrogen = (1) where q is. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. In case of liquid to gas phase change, this amount of energy is known as the. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). (1) where q is the heat supplied during phase transition and m. Learn how nitrogen changes phase with temperature and pressure,. = (1) where q is. Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat, l is defined to be: The latent heat of vaporization can be mathematically expressed as, lv= q m ;
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. Find the chemical, physical. Heat Of Vaporisation Nitrogen.
From www.periodic-table.org
Nitrogen Latent Heat of Vaporization Heat Of Vaporisation Nitrogen Learn how nitrogen changes phase with temperature and pressure,. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. = (1) where q is. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat, l is defined to be: The latent heat of vaporization. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved The boiling temperature of oxygen at atmospheric Heat Of Vaporisation Nitrogen The latent heat of vaporization can be mathematically expressed as, lv= q m ; Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Latent heat, l is defined to be: The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn the definition,. Heat Of Vaporisation Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporisation Nitrogen = (1) where q is. Latent heat, l is defined to be: In case of liquid to gas phase change, this amount of energy is known as the. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat of vaporization of nitrogen is (n2). Heat Of Vaporisation Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporisation Nitrogen In case of liquid to gas phase change, this amount of energy is known as the. (1) where q is the heat supplied during phase transition and m. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. = (1) where q is. Latent heat, l is defined to be: Learn the definition and formula of latent heat of vaporization,. Heat Of Vaporisation Nitrogen.
From www.fizzics.org
Specific Latent Heat notes and video lesson The Fizzics Organization Heat Of Vaporisation Nitrogen Latent heat, l is defined to be: (1) where q is the heat supplied during phase transition and m. Learn how nitrogen changes phase with temperature and pressure,. = (1) where q is. Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. Find the chemical, physical and. Heat Of Vaporisation Nitrogen.
From www.numerade.com
The latent heat of vaporization of liquid nitrogen is about 200 . kJ Heat Of Vaporisation Nitrogen Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). In case of liquid to gas phase change, this amount of energy is known. Heat Of Vaporisation Nitrogen.
From galvinconanstuart.blogspot.com
Nitrogen Phase Diagram Pressure Temperature General Wiring Diagram Heat Of Vaporisation Nitrogen Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. In case of liquid to gas phase change, this amount of energy is known as the. (1) where q is the heat supplied during phase transition. Heat Of Vaporisation Nitrogen.
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporisation Nitrogen (1) where q is the heat supplied during phase transition and m. Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. = (1) where q is. Latent heat, l is defined to be: The latent heat of vaporization can be mathematically expressed as, lv= q m ;. Heat Of Vaporisation Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporisation Nitrogen Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat, l is defined to be: The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Find the chemical, physical and thermal properties of nitrogen, including its boiling. Heat Of Vaporisation Nitrogen.
From golearning5.blogspot.com
The boiling temperature of nitrogen at atmospheric pressure at sea Heat Of Vaporisation Nitrogen Latent heat, l is defined to be: (1) where q is the heat supplied during phase transition and m. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). In case of liquid to gas phase change, this amount of energy is known as the. Latent heat of vaporization of nitrogen is (n2). Heat Of Vaporisation Nitrogen.
From www.researchgate.net
Saturation curve for nitrogen. Download Scientific Diagram Heat Of Vaporisation Nitrogen The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). = (1) where q is. Learn how nitrogen changes phase with temperature and pressure,. The latent heat of vaporization can be mathematically expressed as, lv= q m ; Latent heat, l is defined to be: Find the chemical, physical and thermal properties of. Heat Of Vaporisation Nitrogen.
From www.researchgate.net
A schematic diagram of the nitrogen reactive electron beam evaporation Heat Of Vaporisation Nitrogen Learn how nitrogen changes phase with temperature and pressure,. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat of vaporization of nitrogen is (n2). Heat Of Vaporisation Nitrogen.
From www.numerade.com
SOLVED The table displays the vapor pressure of nitrogen at several Heat Of Vaporisation Nitrogen The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). (1) where q is the heat supplied during phase transition and m. Latent heat, l is defined to be: The latent heat of vaporization can be mathematically expressed as, lv= q m ; In case of liquid to gas phase change, this amount. Heat Of Vaporisation Nitrogen.
From www.researchgate.net
Oxygen, nitrogen and argon vapor pressure curves. Download Scientific Heat Of Vaporisation Nitrogen The latent heat of vaporization can be mathematically expressed as, lv= q m ; Learn how nitrogen changes phase with temperature and pressure,. (1) where q is the heat supplied during phase transition and m. Latent heat, l is defined to be: Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and. Heat Of Vaporisation Nitrogen.
From exolhbtzn.blob.core.windows.net
The Condensing Of Vapor at Carlos Lee blog Heat Of Vaporisation Nitrogen = (1) where q is. Latent heat, l is defined to be: The latent heat of vaporization can be mathematically expressed as, lv= q m ; Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. (1) where q is the heat supplied during phase transition and m.. Heat Of Vaporisation Nitrogen.
From olympiapublishers.com
Latent Heat Of Vaporization Of Liquid Nitrogen In J/g Heat Of Vaporisation Nitrogen The latent heat of vaporization can be mathematically expressed as, lv= q m ; Latent heat, l is defined to be: Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn how nitrogen changes phase with temperature and pressure,. In case of. Heat Of Vaporisation Nitrogen.
From www.researchgate.net
Saturation curve for nitrogen. Download Scientific Diagram Heat Of Vaporisation Nitrogen (1) where q is the heat supplied during phase transition and m. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn how nitrogen changes phase with temperature and pressure,. Latent heat, l is defined to be: Learn the definition and formula of latent heat of vaporization, the input energy required to. Heat Of Vaporisation Nitrogen.
From www.youtube.com
Virtual Lab Heat of Vaporization of Liquid Nitrogen YouTube Heat Of Vaporisation Nitrogen Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat, l is defined to be: Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Learn how nitrogen changes phase with temperature and pressure,. The. Heat Of Vaporisation Nitrogen.
From www.chemix-chemistry-software.com
Nitrogen phase diagram Heat Of Vaporisation Nitrogen The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). In case of liquid to gas phase change, this amount of energy is known as the. Learn how nitrogen changes phase with temperature and pressure,. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the. Heat Of Vaporisation Nitrogen.
From studylib.net
HEAT OF VAPORIZATION OF NITROGEN ©By Lee Marek Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. In case of liquid to gas phase change, this amount of energy is known as the. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. The aim of the experiment was to determine the latent. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporisation Nitrogen (1) where q is the heat supplied during phase transition and m. In case of liquid to gas phase change, this amount of energy is known as the. Learn how nitrogen changes phase with temperature and pressure,. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Learn the definition, formula and examples of heat of vaporization, also called enthalpy. Heat Of Vaporisation Nitrogen.
From www.slideserve.com
PPT Thermochemistry PowerPoint Presentation, free download ID2841621 Heat Of Vaporisation Nitrogen Learn how nitrogen changes phase with temperature and pressure,. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. = (1) where q is. The latent heat of vaporization can be mathematically expressed as, lv= q. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved The boiling temperature of nitrogen at atmospheric Heat Of Vaporisation Nitrogen Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Learn how nitrogen changes phase with temperature and pressure,. In case of liquid to gas phase change, this amount of energy is known as the. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which. Heat Of Vaporisation Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporisation Nitrogen In case of liquid to gas phase change, this amount of energy is known as the. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. (1) where q is the heat supplied during phase transition and m. Learn the definition, formula and examples of heat of vaporization, also called enthalpy. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporisation Nitrogen The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). = (1) where q is. The latent heat of vaporization can be mathematically expressed as, lv= q m ; (1) where q is the heat supplied during phase transition and m. Learn the definition and formula of latent heat of vaporization, the input. Heat Of Vaporisation Nitrogen.
From www.toppr.com
Liquid nitrogen has a boiling point of 77.3 K and a latent heat of Heat Of Vaporisation Nitrogen The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). (1) where q is the heat supplied during phase transition and m. In case of liquid to gas phase change, this amount of energy is known as the. = (1) where q is. Learn the definition and formula of latent heat of vaporization,. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved The boiling temperature of nitrogen at atmospheric Heat Of Vaporisation Nitrogen (1) where q is the heat supplied during phase transition and m. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. The latent heat of vaporization can be mathematically expressed as, lv= q m ; In case of liquid to gas phase change, this amount. Heat Of Vaporisation Nitrogen.
From democracyunlimited.web.fc2.com
heat of vaporization of liquid nitrogen Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. = (1) where q is. Learn the. Heat Of Vaporisation Nitrogen.
From safetydata.netlify.app
What the boiling point of nitrogen in celsius safetydata Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Latent heat, l is defined to be: The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. Find the chemical, physical. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved The boiling temperature of nitrogen at atmospheric Heat Of Vaporisation Nitrogen (1) where q is the heat supplied during phase transition and m. = (1) where q is. Latent heat, l is defined to be: Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn how nitrogen changes phase with temperature and pressure,.. Heat Of Vaporisation Nitrogen.
From classes.oc.edu
THE LATENT HEAT OF VAPORIZATION OF NITROGEN Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. (1) where q is the heat supplied during phase transition and m. Latent heat, l is defined to be: = (1) where q is. The aim of the experiment was to determine the latent heat of vaporisation of liquid nitrogen (ln2). Learn the definition, formula and examples of heat of. Heat Of Vaporisation Nitrogen.
From www.britannica.com
How the Nitrogen Cycle Works Britannica Heat Of Vaporisation Nitrogen Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. Learn the definition, formula and examples of heat of vaporization, also called enthalpy of vaporization, which is the heat required to vaporize a liquid. In case of liquid to gas phase change, this amount of energy is known as the. Learn the definition and formula of latent heat of vaporization,. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved 2. The boiling temperature of nitrogen at atmospheric Heat Of Vaporisation Nitrogen Latent heat, l is defined to be: Learn the definition and formula of latent heat of vaporization, the input energy required to change liquid to vapor at constant temperature. Latent heat of vaporization of nitrogen is (n2) 5.56 kj/mol. (1) where q is the heat supplied during phase transition and m. = (1) where q is. Learn the definition, formula. Heat Of Vaporisation Nitrogen.
From www.chegg.com
Solved Latent Heat of Vaporization of Liquid Nitrogen Heat Of Vaporisation Nitrogen In case of liquid to gas phase change, this amount of energy is known as the. = (1) where q is. Find the chemical, physical and thermal properties of nitrogen, including its boiling point, density, viscosity, specific heat and more. The latent heat of vaporization can be mathematically expressed as, lv= q m ; Latent heat, l is defined to. Heat Of Vaporisation Nitrogen.